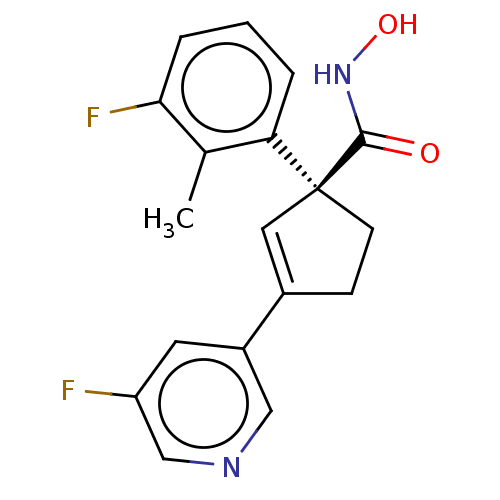
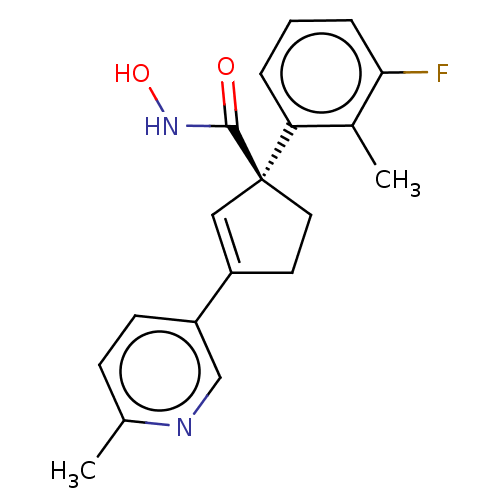
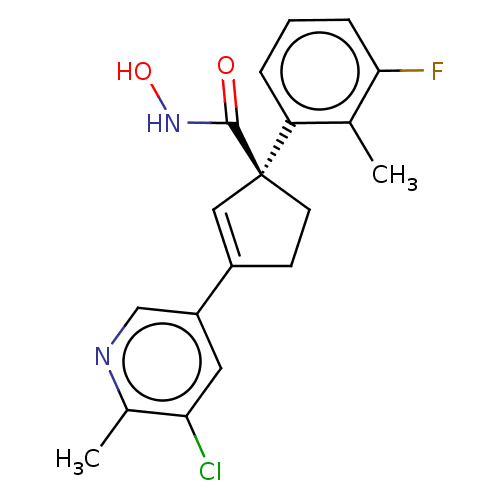
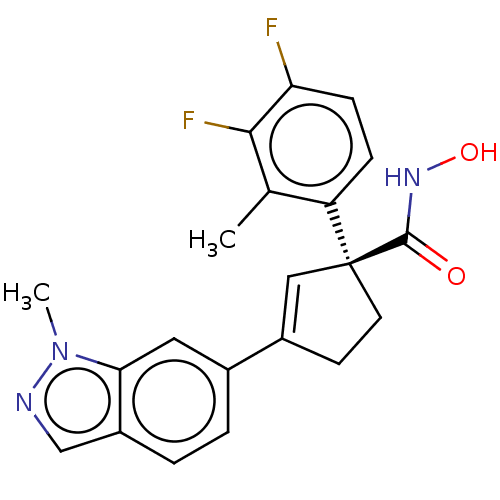
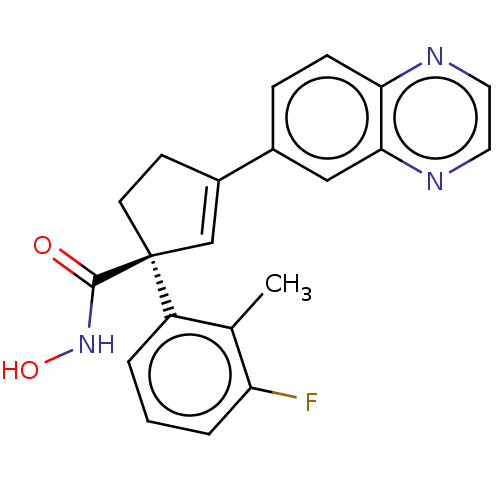
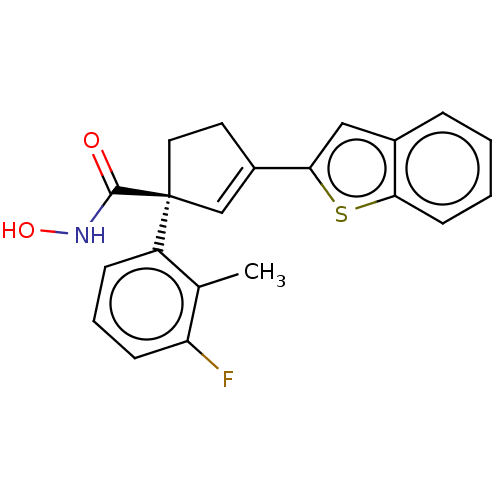
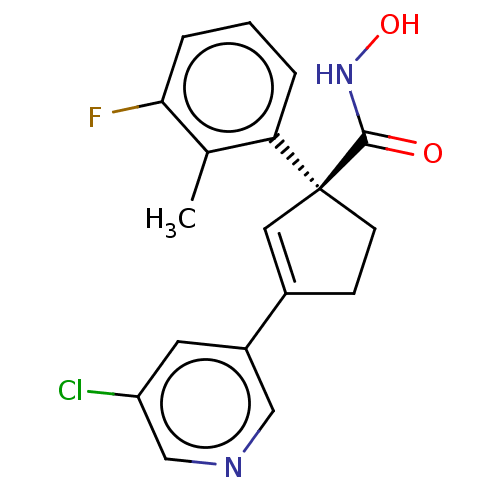
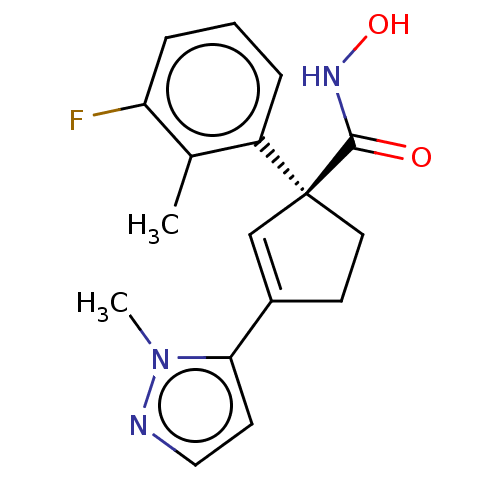
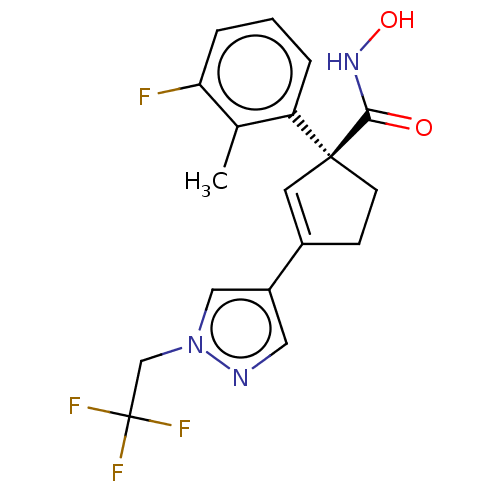
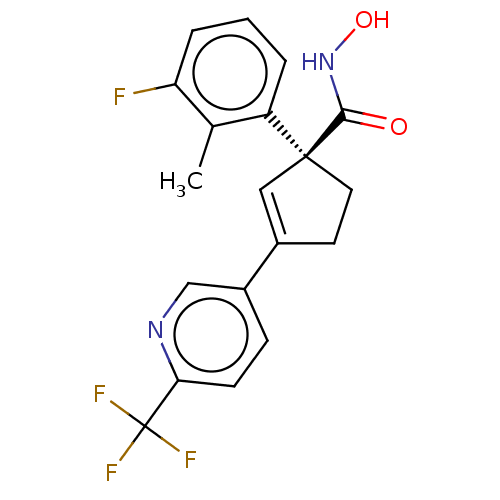
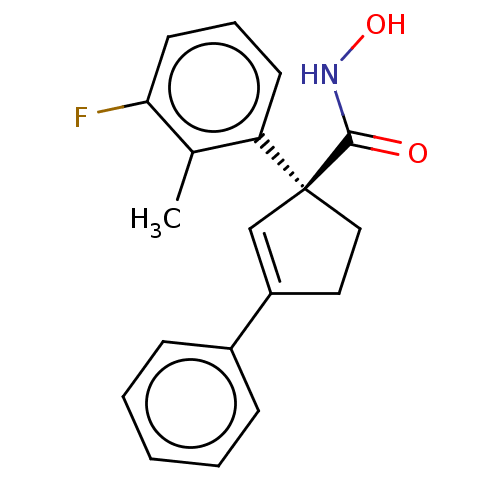
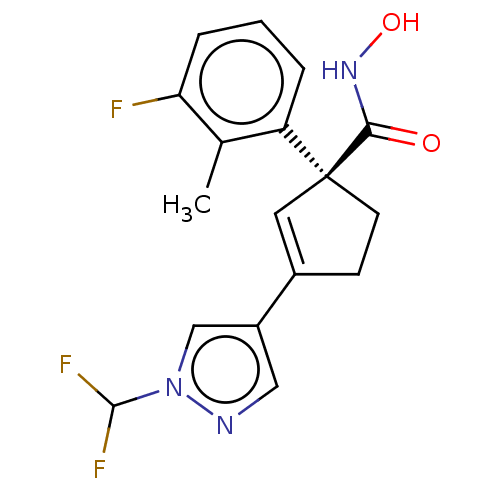
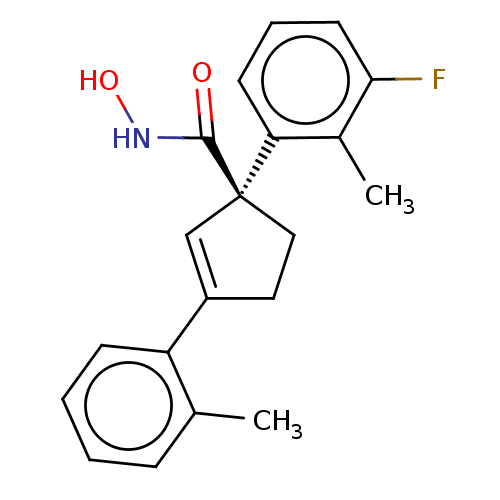
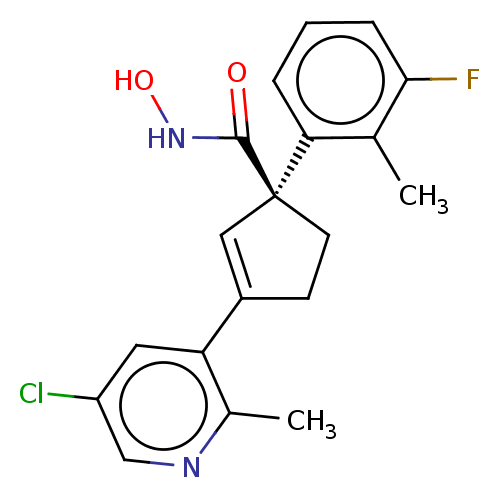
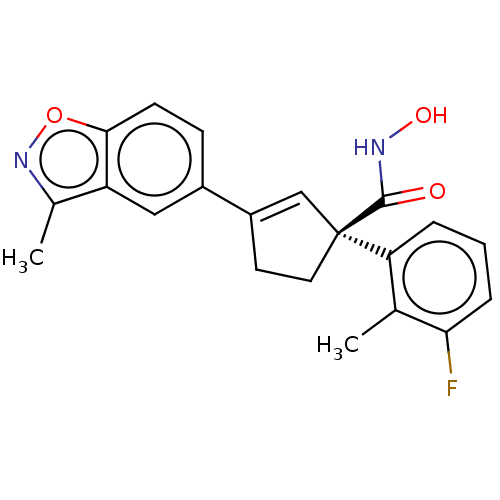
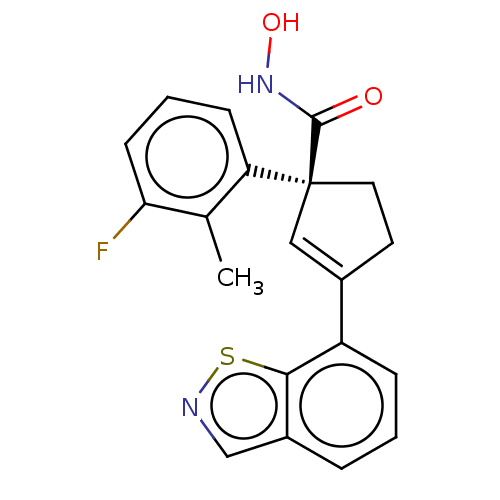
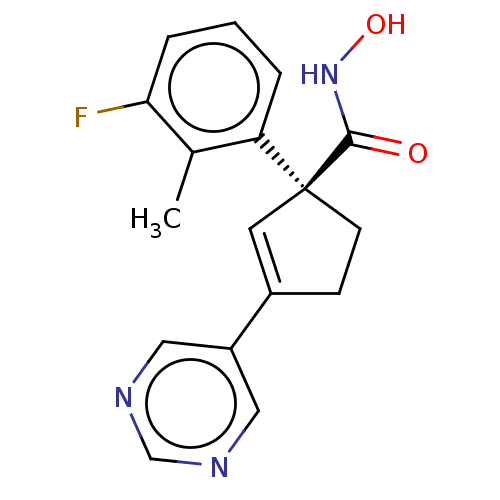
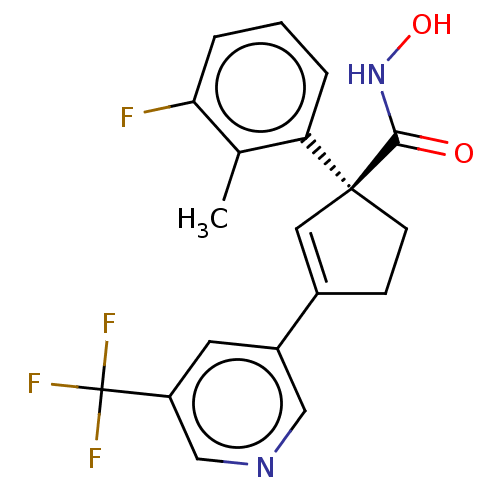
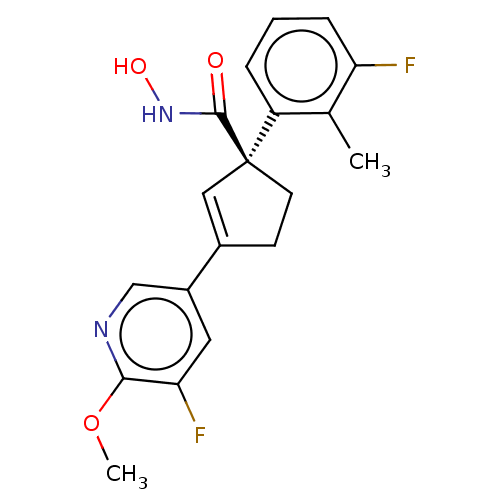
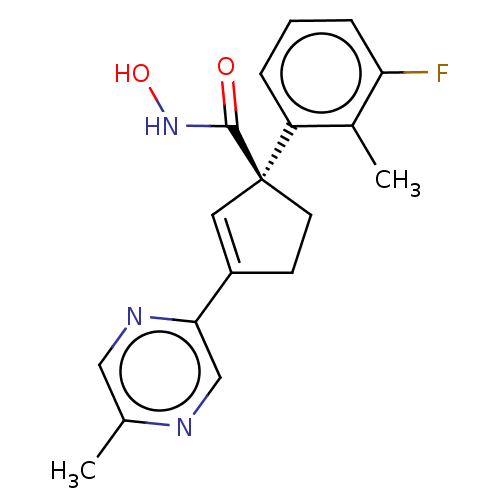
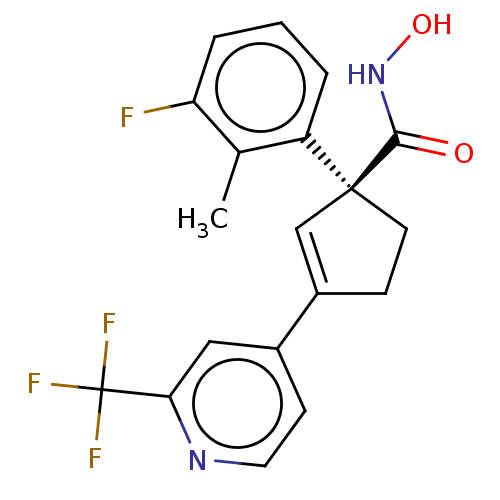
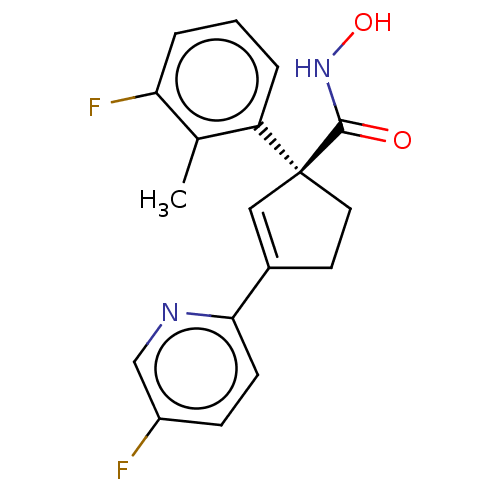
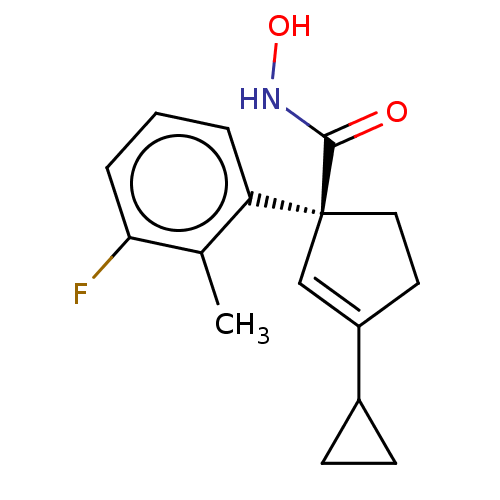
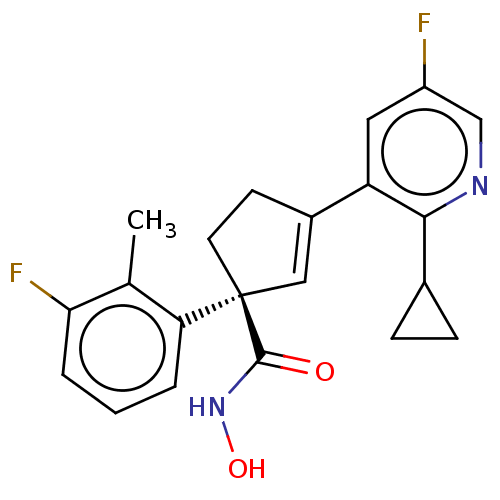
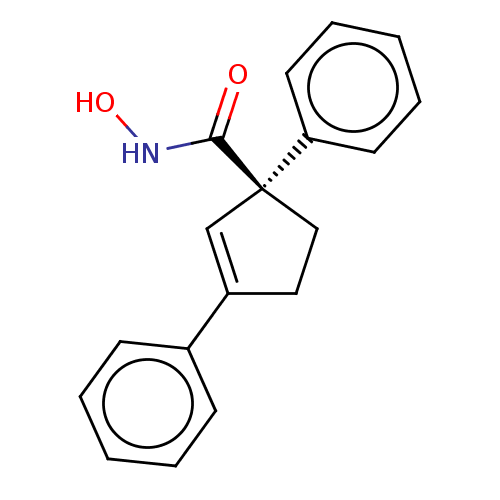
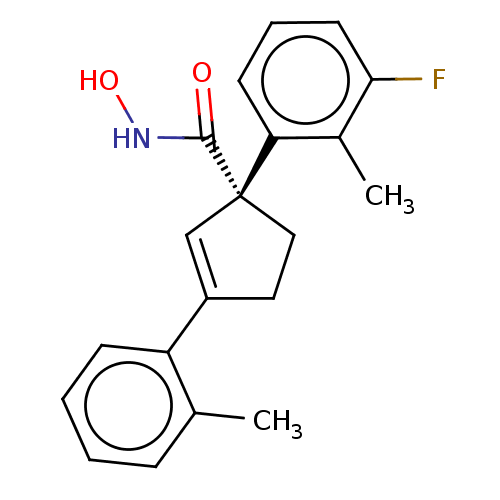
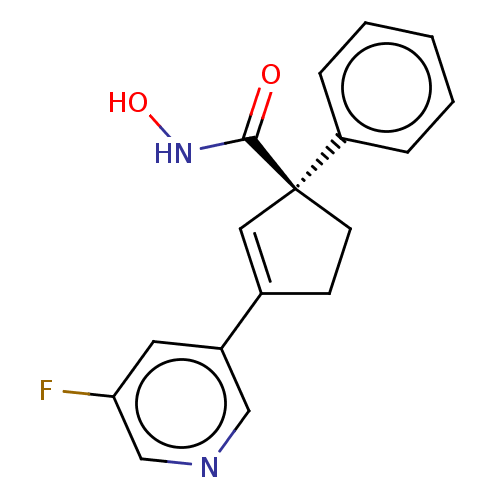
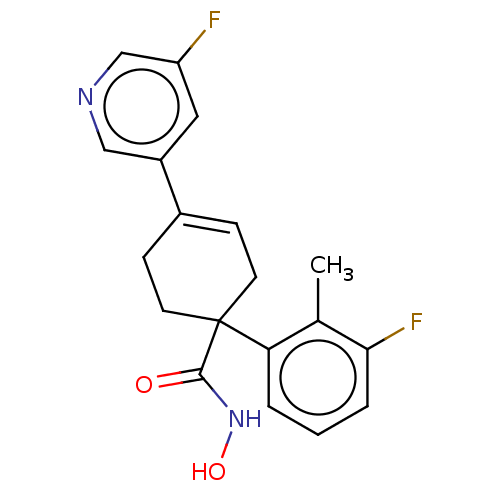
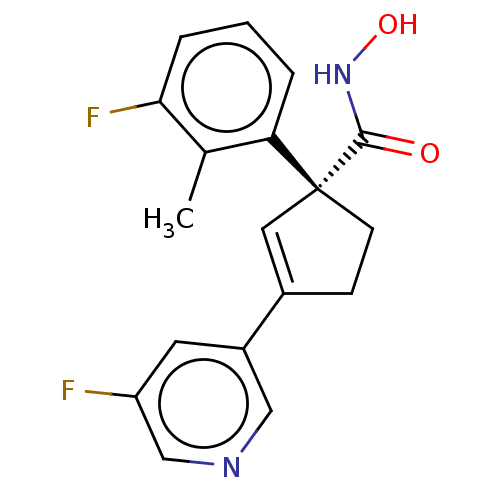
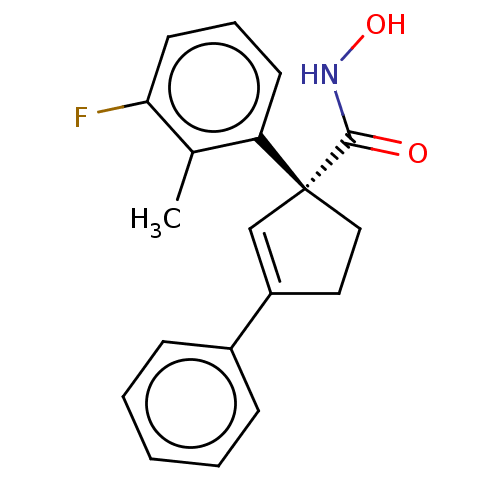
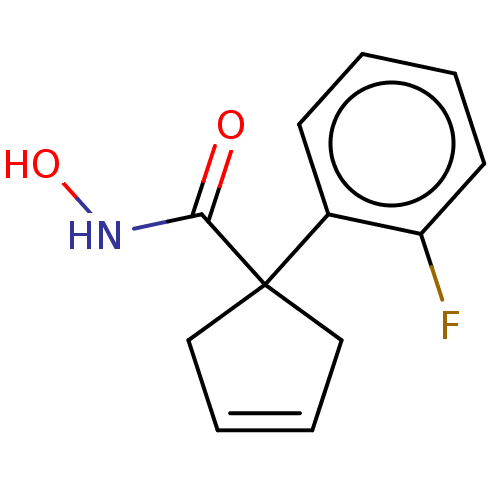
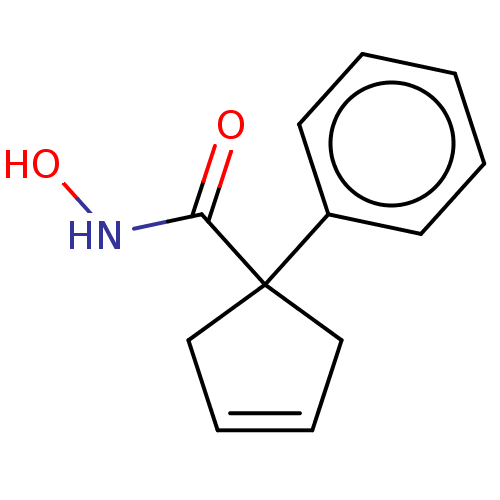
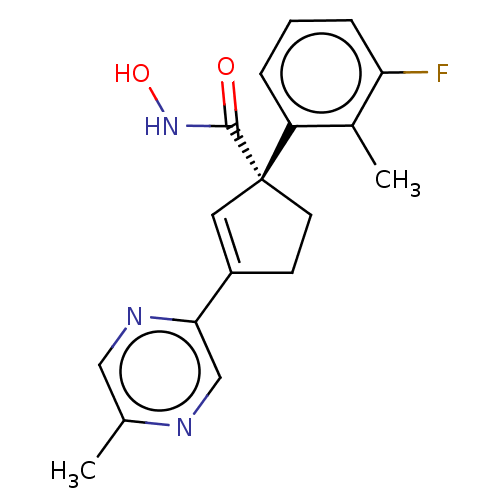
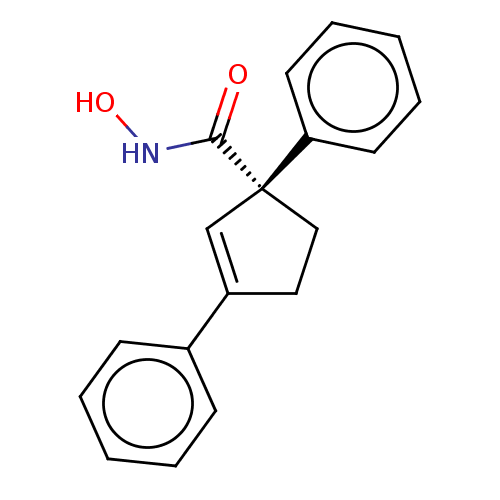
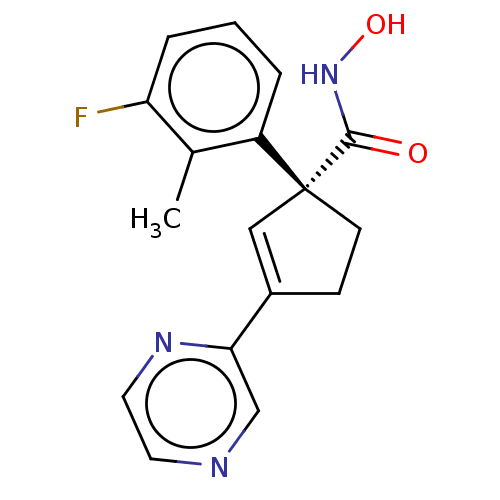
Report error Found 45 Enz. Inhib. hit(s) with all data for entry = 1318
Affinity DataIC50: 10nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 20nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 30nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 30nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 30nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 30nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 30nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 40nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 40nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 40nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 40nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 40nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 40nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 70nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 190nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 230nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 340nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 490nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 550nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 670nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 2.40E+3nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 3.60E+3nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 7.60E+3nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 8.30E+3nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 1.23E+4nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 2.68E+4nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 3.10E+4nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair
Affinity DataIC50: 5.00E+4nMAssay Description:2 μl (200×) of each diluted solution and each control (full activity: 100% DMSO alone or full inhibition 1 mM) is stamped into V-bottomed polypr...More data for this Ligand-Target Pair