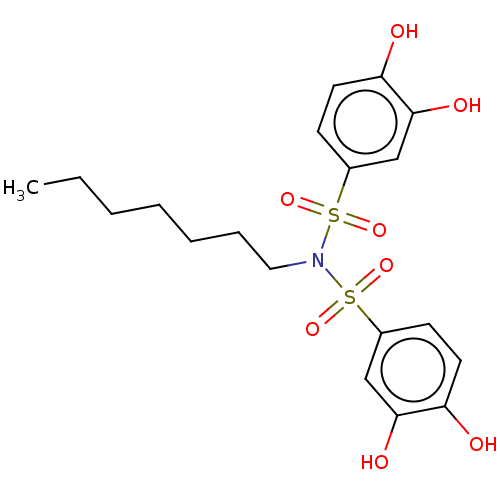
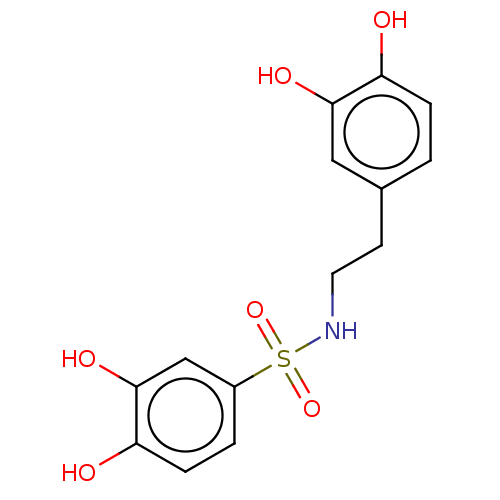
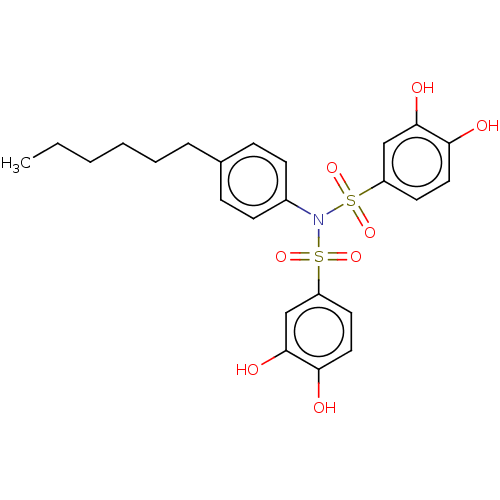
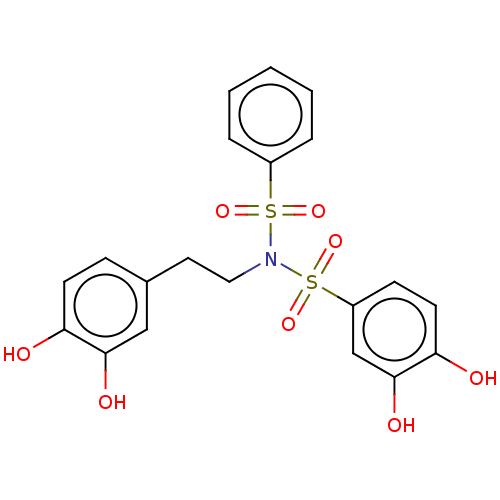
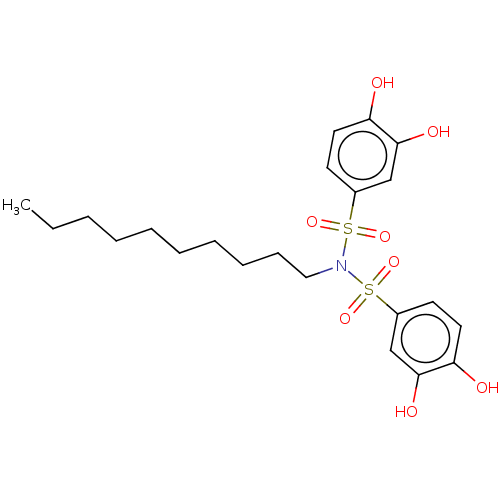
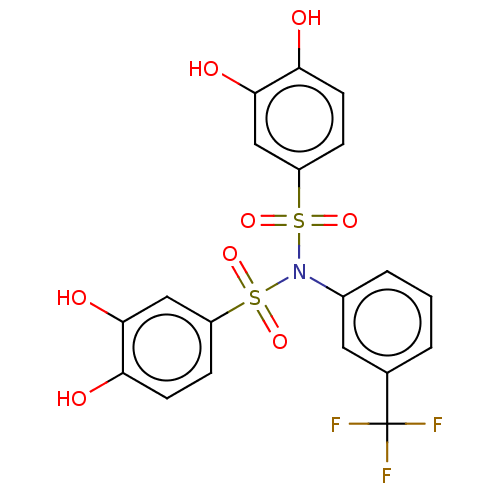
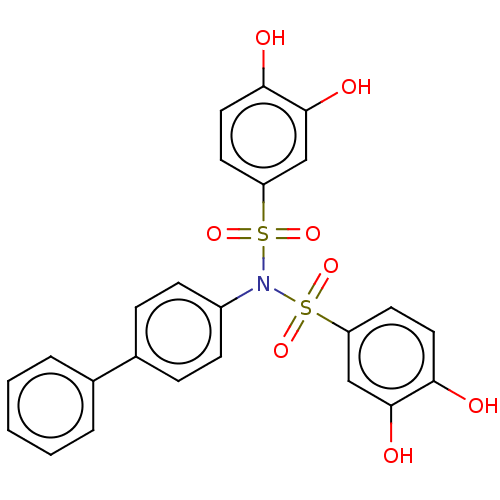
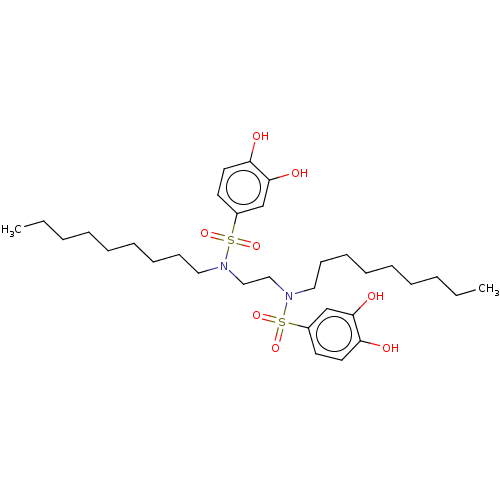
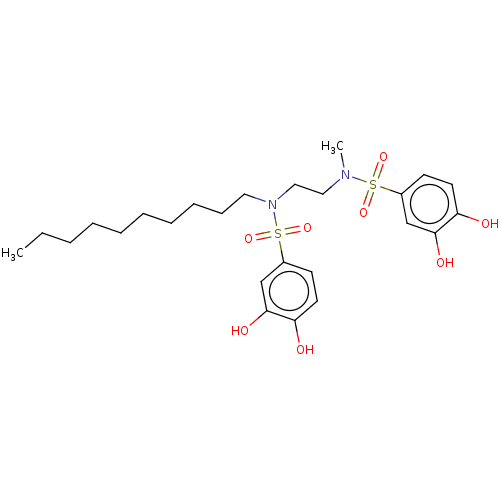
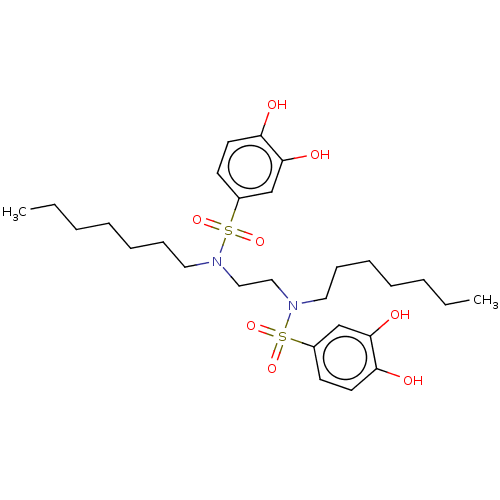
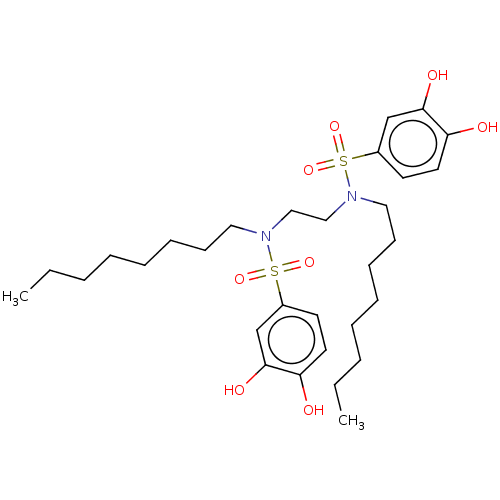
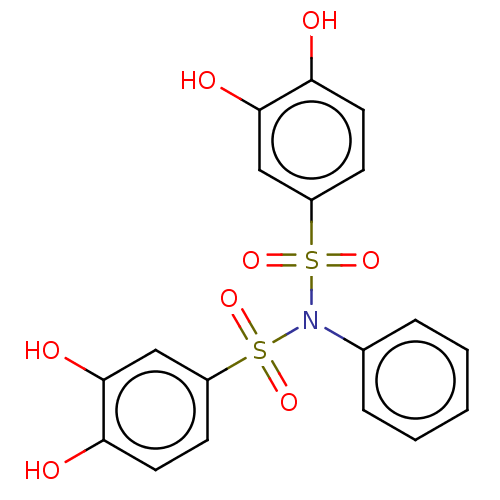
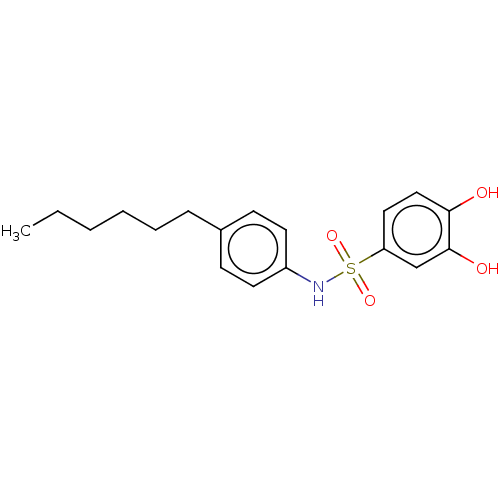
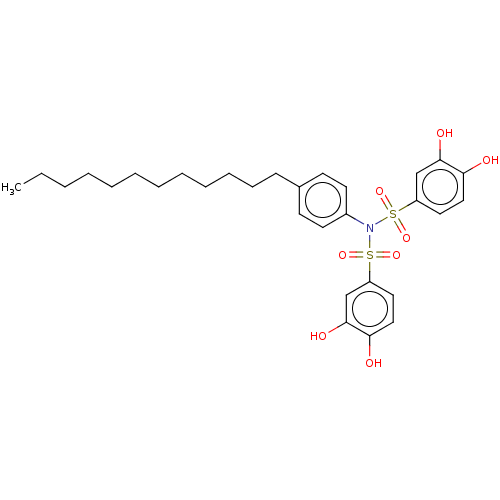
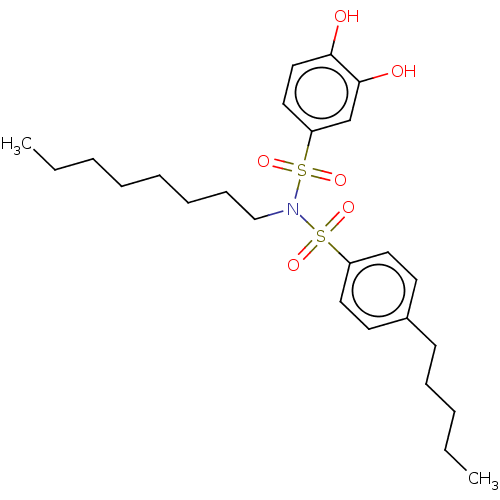
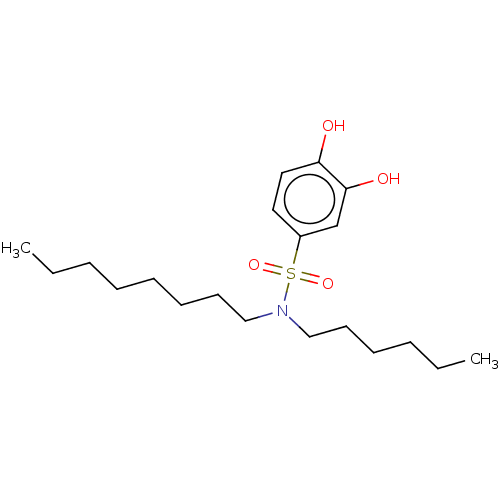
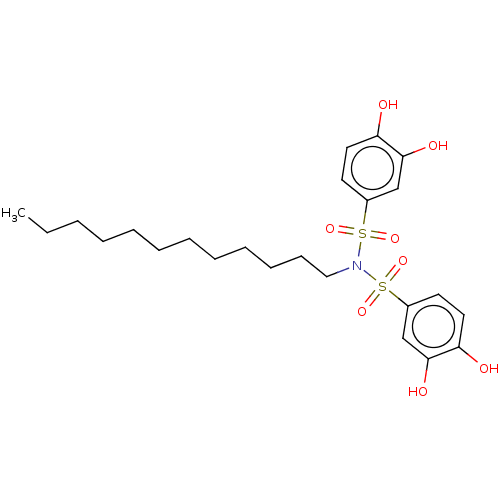
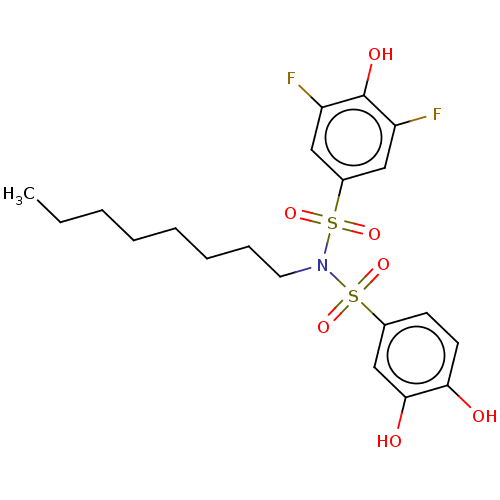
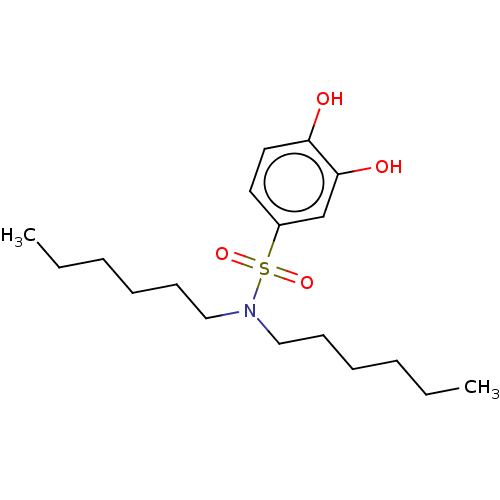
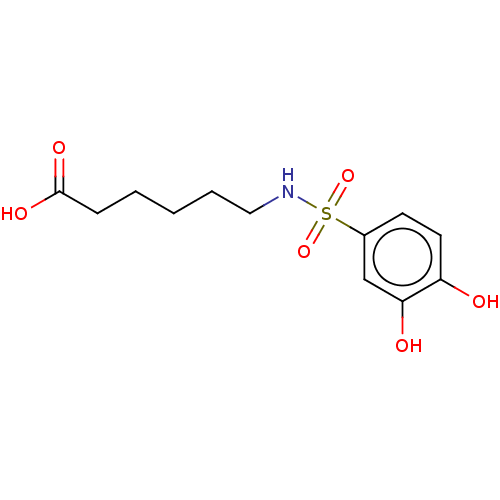
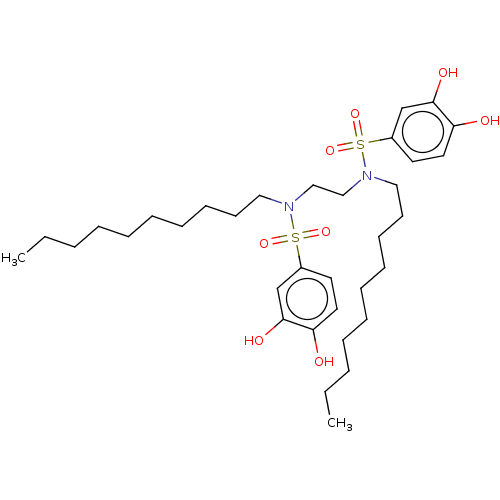
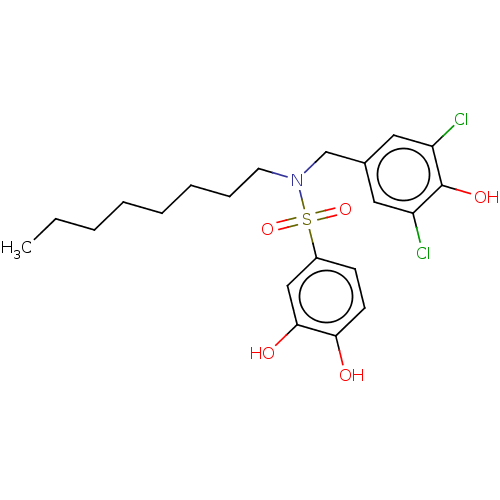
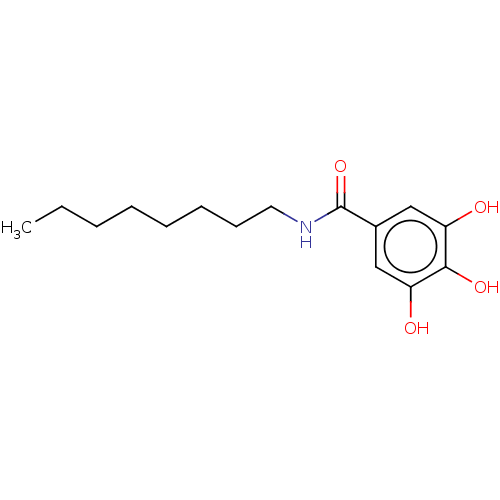
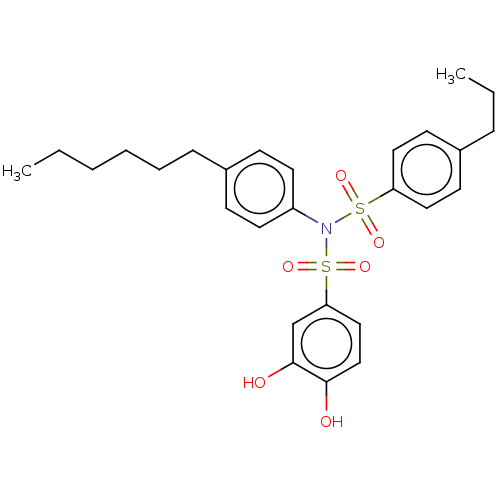
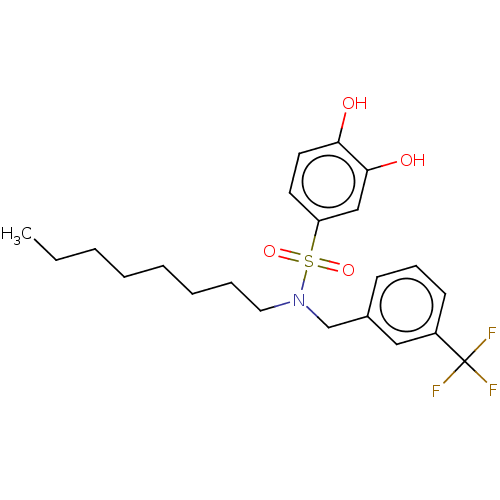
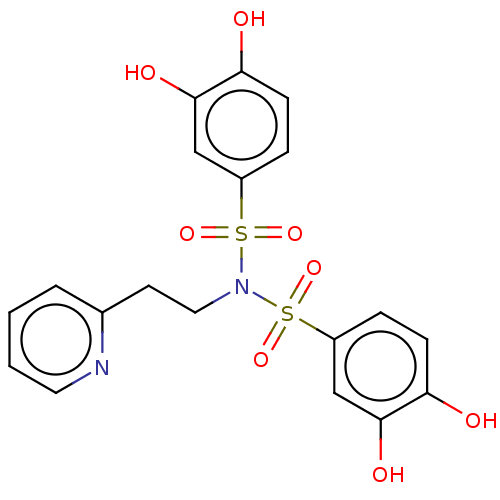
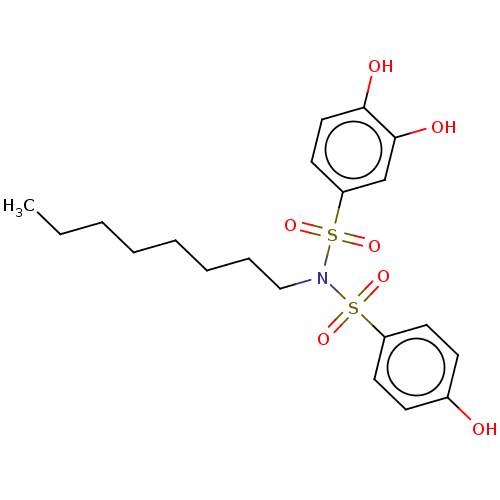
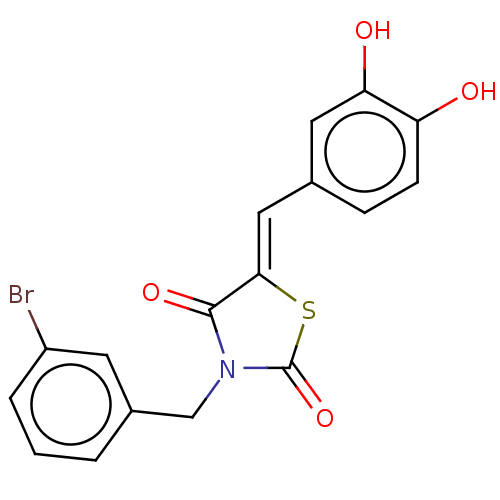
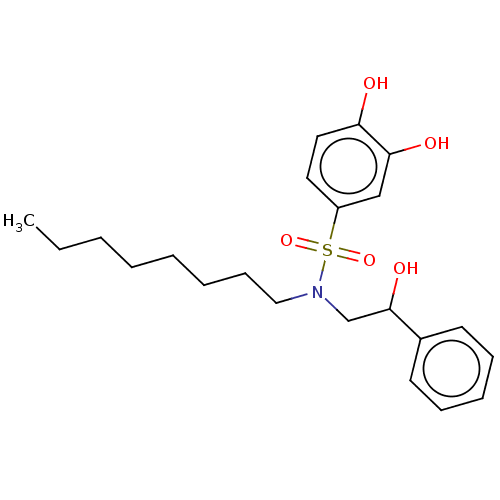
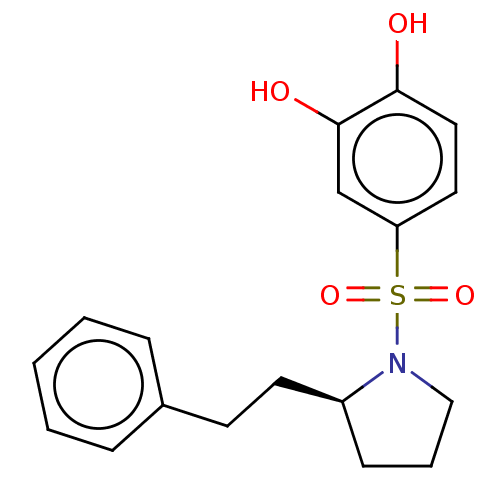
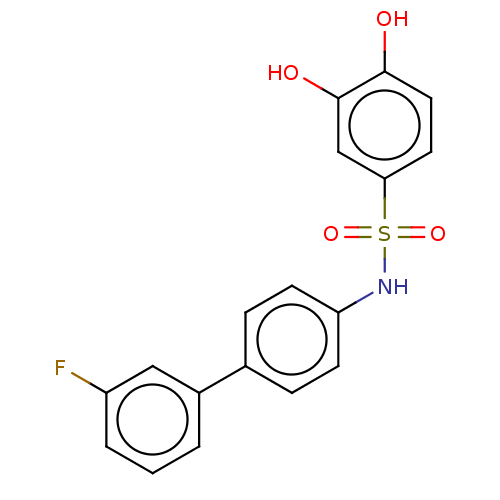
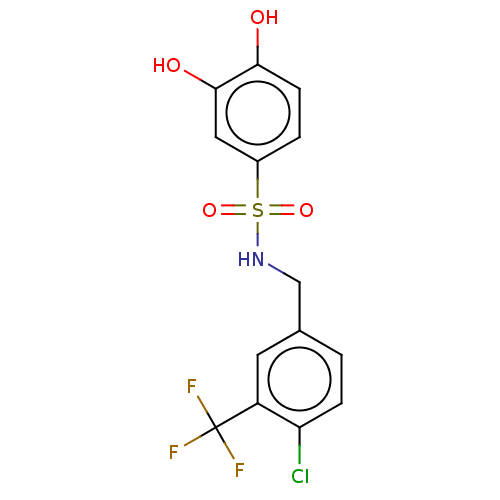
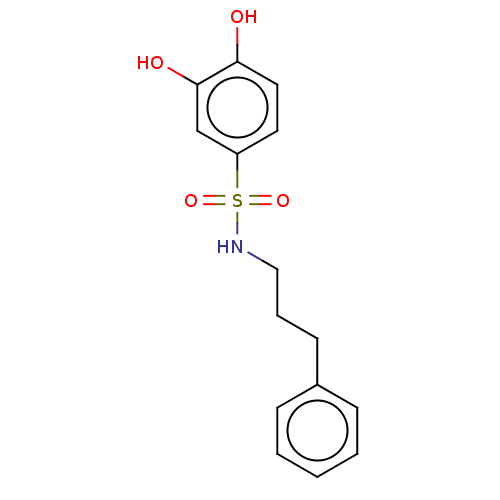
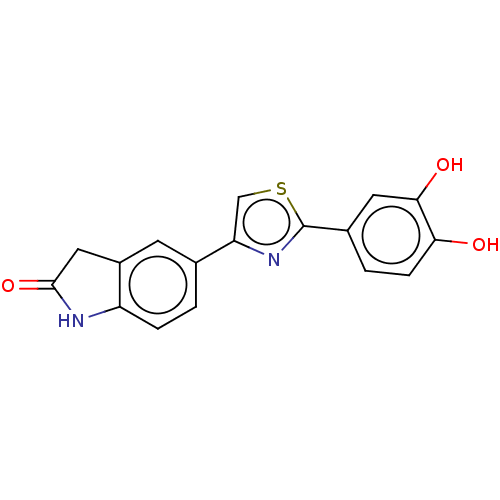
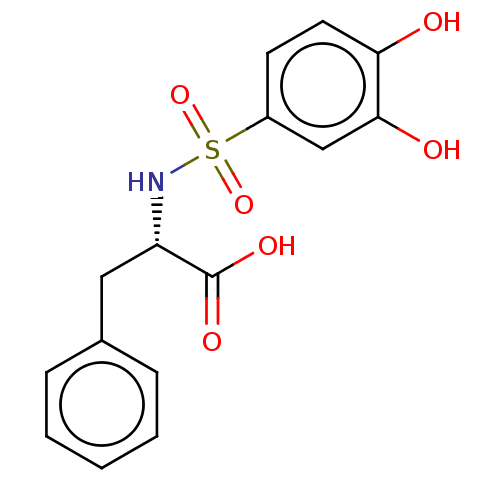
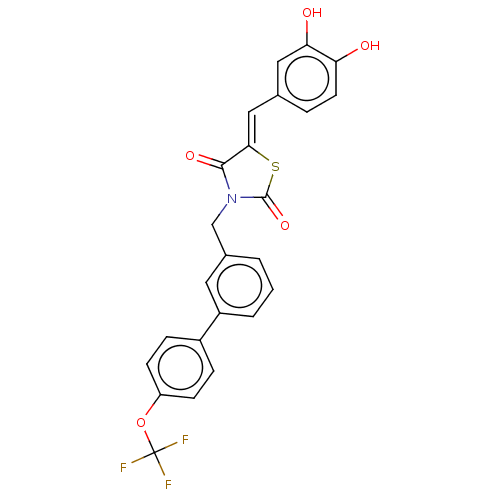
Report error Found 257 Enz. Inhib. hit(s) with all data for entry = 390
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
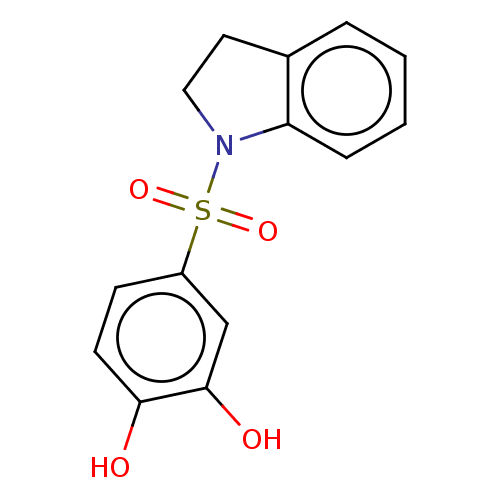
Affinity DataIC50: 33nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
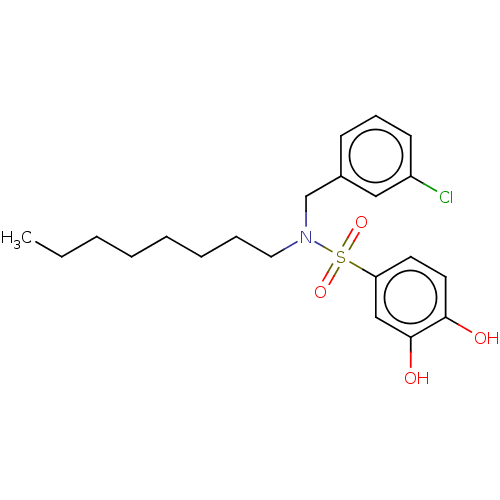
Affinity DataIC50: 35nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
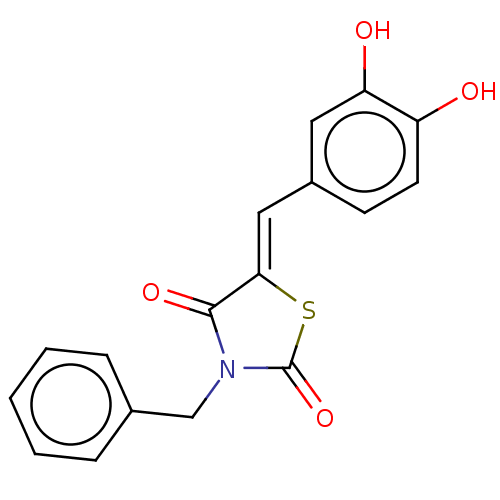
Affinity DataIC50: 51nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
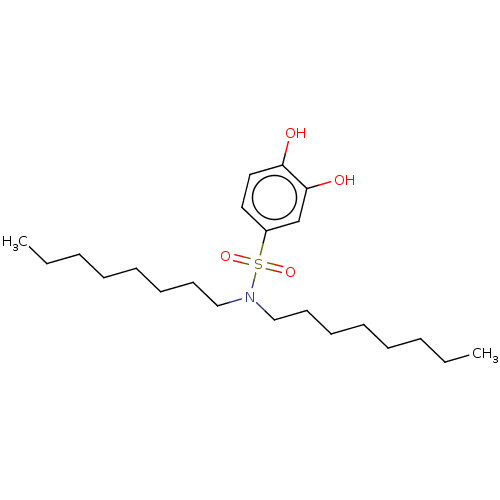
Affinity DataIC50: 120nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 173nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 180nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 250nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 288nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 350nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 420nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 450nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Tissue-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 510nMT: 2°CAssay Description:PAI-1 inhibitor compounds were dissolved in DMSO to a final concentration of (10-50 mM), depending upon solubility. Compounds were then diluted in ph...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 590nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Tissue-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 611nMT: 2°CAssay Description:PAI-1 inhibitor compounds were dissolved in DMSO to a final concentration of (10-50 mM), depending upon solubility. Compounds were then diluted in ph...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 620nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 700nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 910nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 920nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Tissue-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 980nMT: 2°CAssay Description:PAI-1 inhibitor compounds were dissolved in DMSO to a final concentration of (10-50 mM), depending upon solubility. Compounds were then diluted in ph...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Tissue-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 1.02E+3nMT: 2°CAssay Description:PAI-1 inhibitor compounds were dissolved in DMSO to a final concentration of (10-50 mM), depending upon solubility. Compounds were then diluted in ph...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 1.28E+3nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 1.34E+3nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Tissue-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 1.44E+3nMT: 2°CAssay Description:PAI-1 inhibitor compounds were dissolved in DMSO to a final concentration of (10-50 mM), depending upon solubility. Compounds were then diluted in ph...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 1.57E+3nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 1.67E+3nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Tissue-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 1.71E+3nMT: 2°CAssay Description:PAI-1 inhibitor compounds were dissolved in DMSO to a final concentration of (10-50 mM), depending upon solubility. Compounds were then diluted in ph...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 1.82E+3nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 1.98E+3nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 1.98E+3nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Tissue-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 2.21E+3nMT: 2°CAssay Description:PAI-1 inhibitor compounds were dissolved in DMSO to a final concentration of (10-50 mM), depending upon solubility. Compounds were then diluted in ph...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 2.29E+3nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 2.40E+3nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 2.60E+3nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 3.70E+3nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 3.90E+3nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 4.34E+3nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 4.96E+3nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 5.05E+3nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 5.11E+3nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 5.36E+3nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 5.49E+3nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 6.74E+3nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 6.98E+3nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Tissue-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 7.59E+3nMT: 2°CAssay Description:PAI-1 inhibitor compounds were dissolved in DMSO to a final concentration of (10-50 mM), depending upon solubility. Compounds were then diluted in ph...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 9.70E+3nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 1.26E+4nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 1.33E+4nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 1.48E+4nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 1.64E+4nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair
TargetPlasminogen activator inhibitor 1/Urokinase-type plasminogen activator(Human)
University of Michigan
US Patent
University of Michigan
US Patent
Affinity DataIC50: 1.68E+4nMpH: 7.4 T: 2°CAssay Description:To determine the efficacy of various synthesized compounds as PAI-1 inhibitors, a fluorometric plate assay was carried out to measure the half maxima...More data for this Ligand-Target Pair