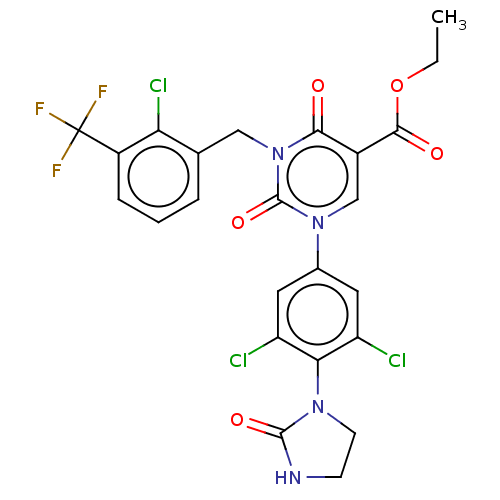
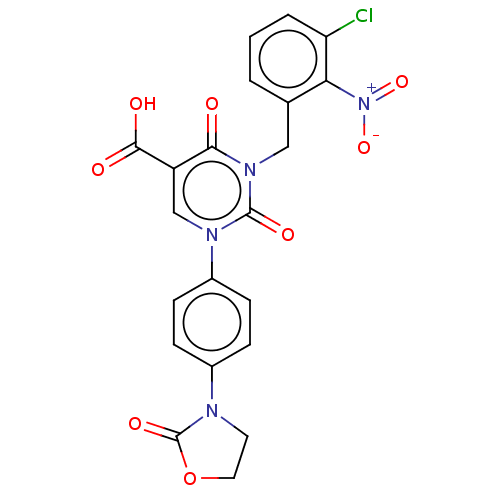
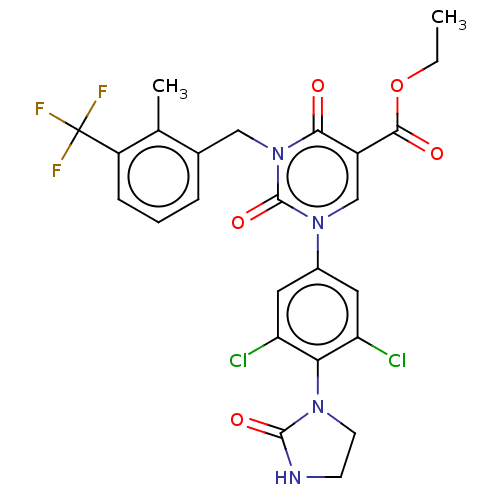
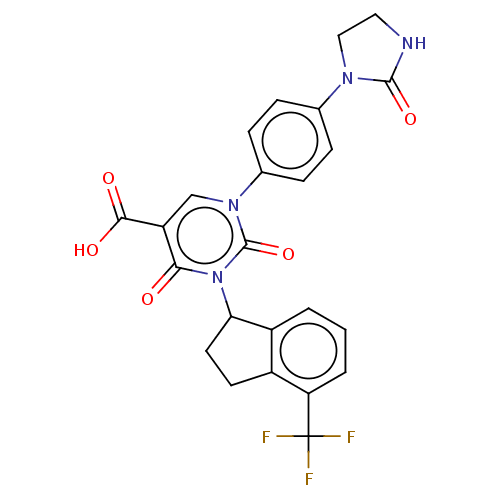
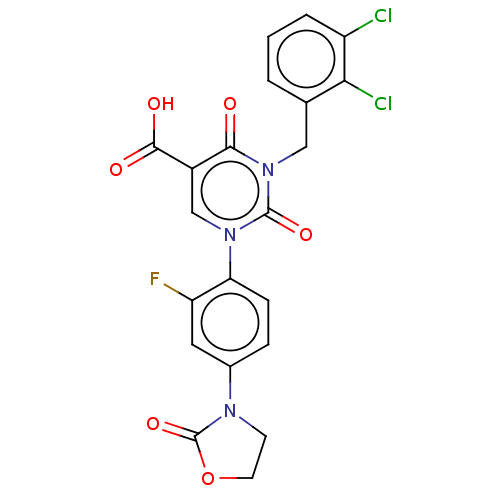
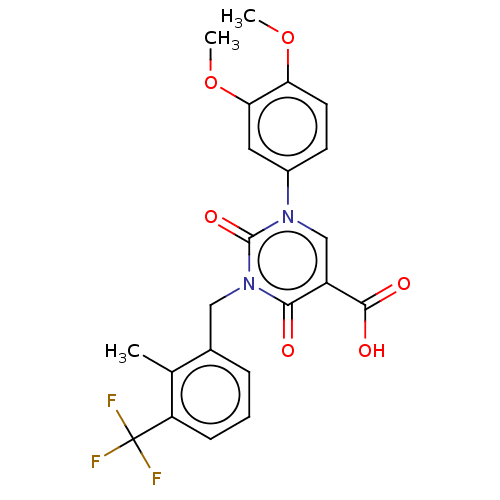
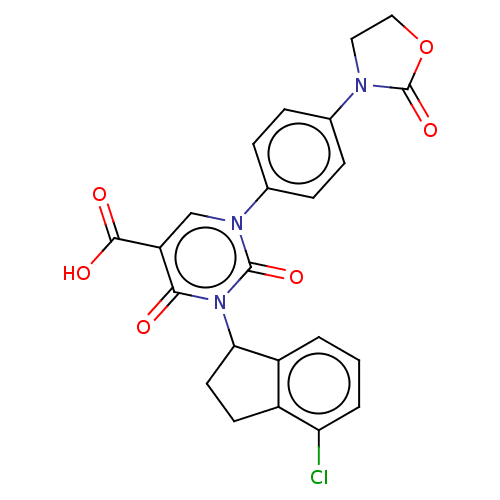
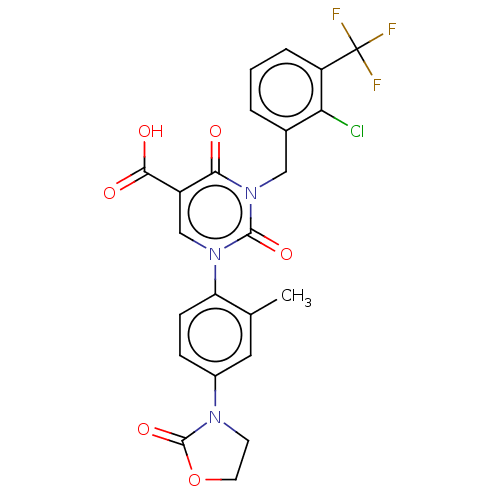
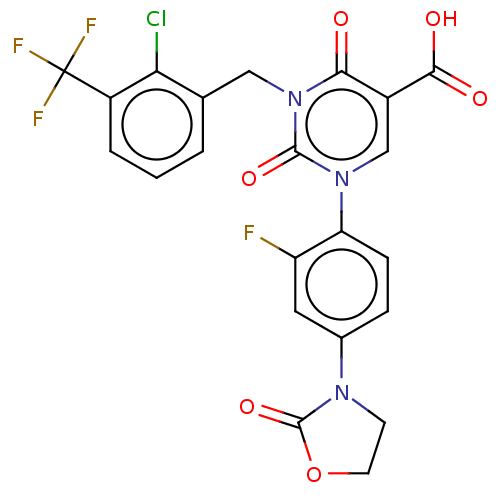
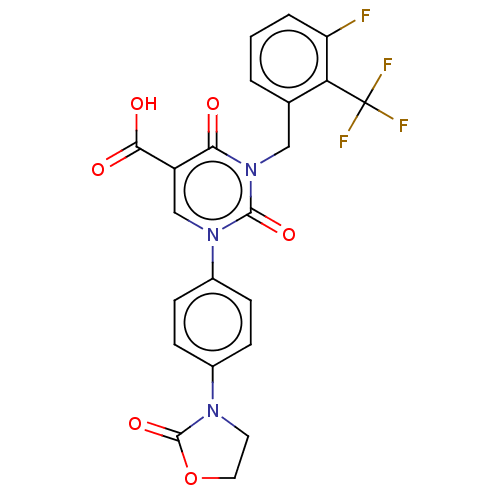
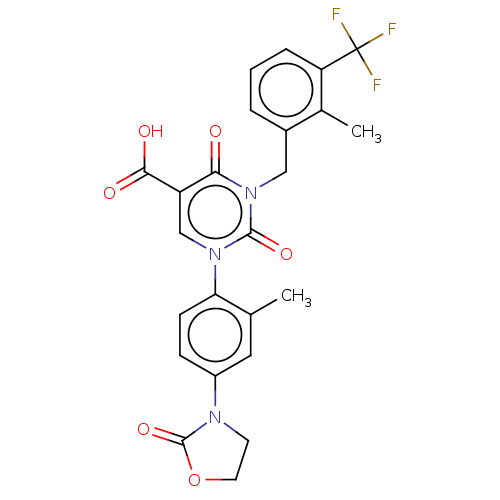
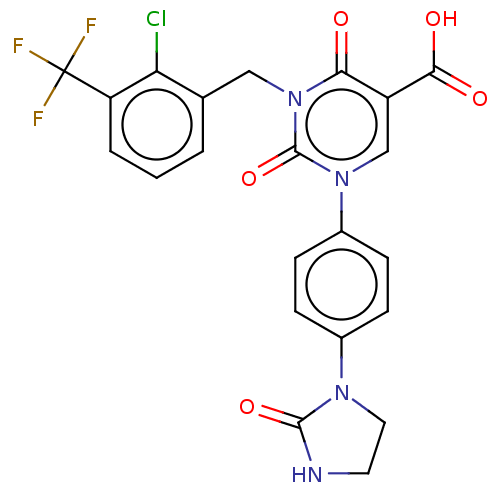
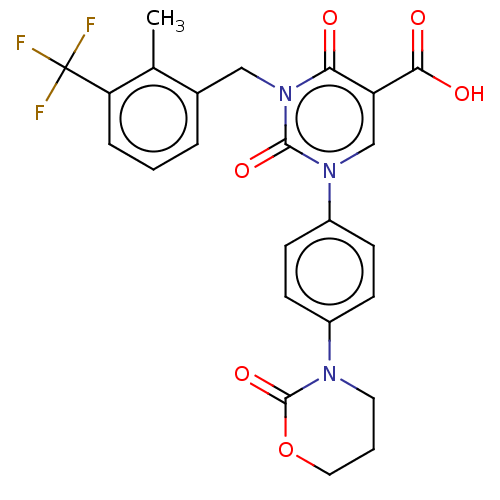
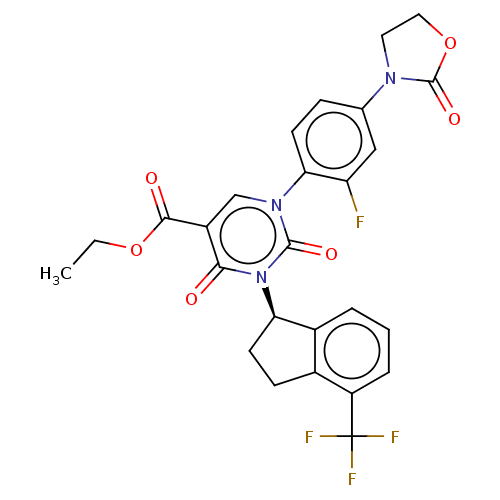
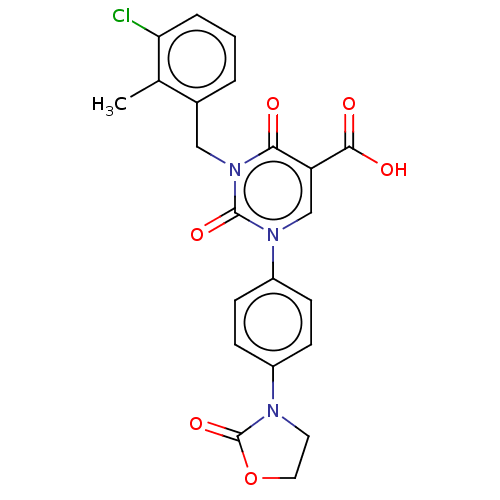
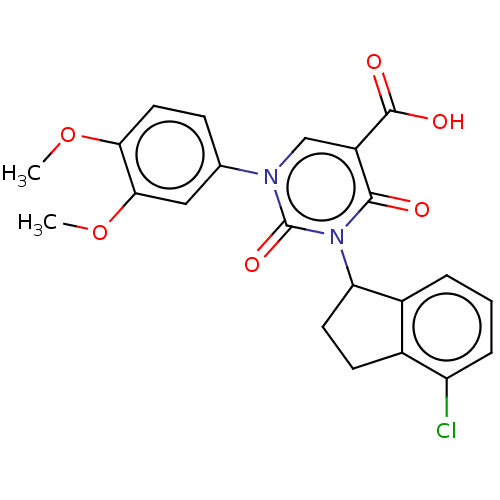
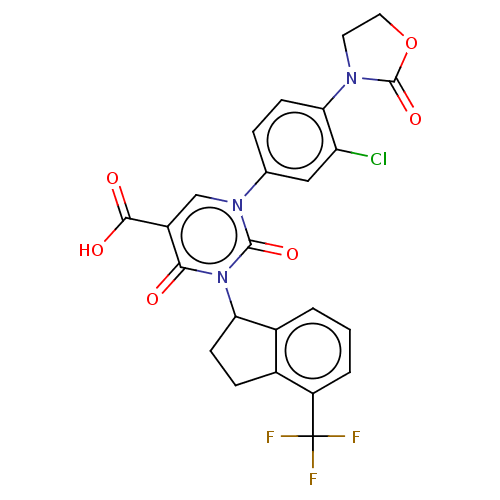
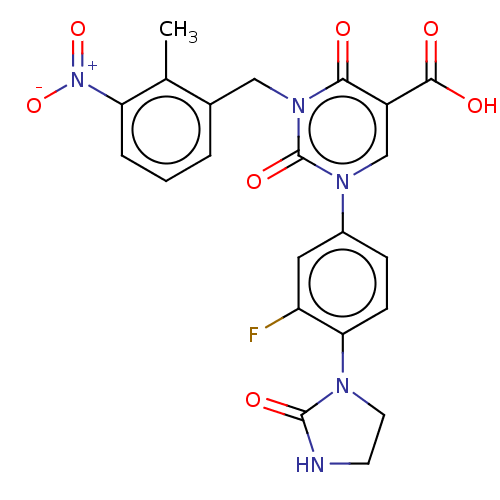
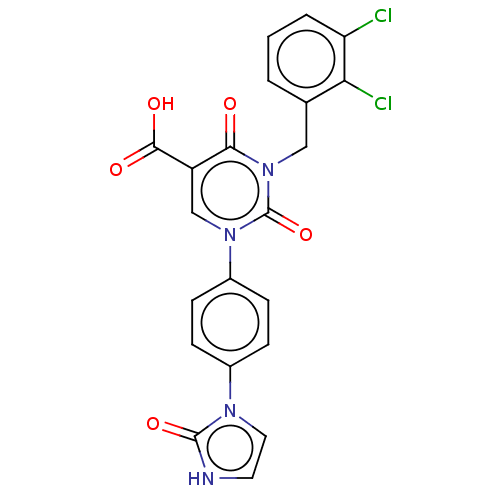
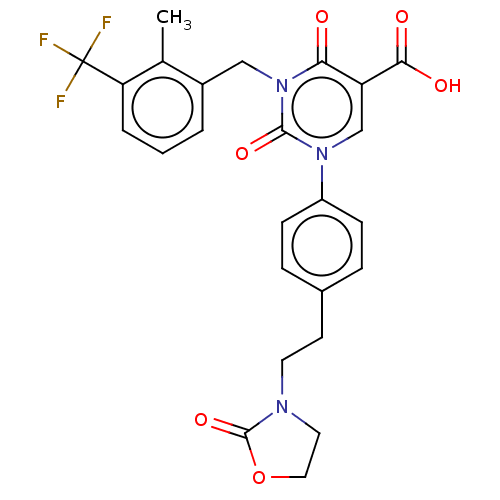
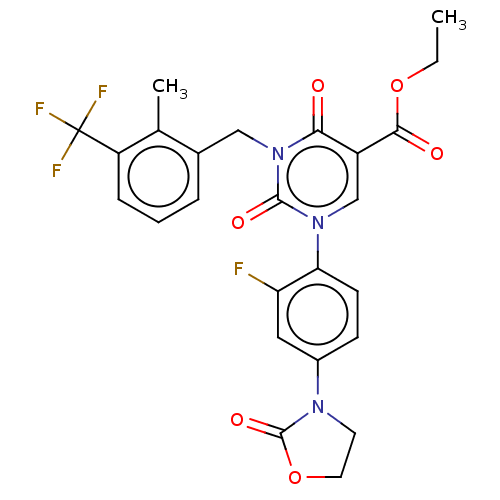
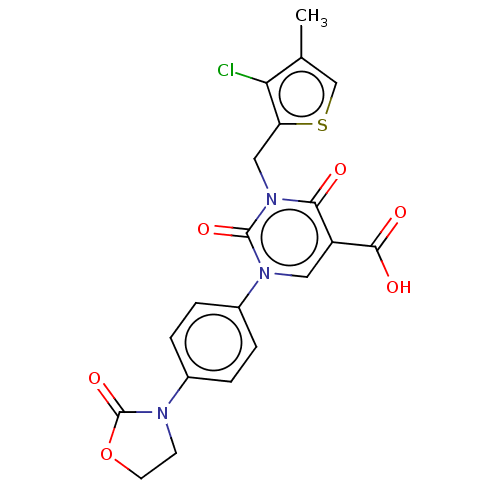
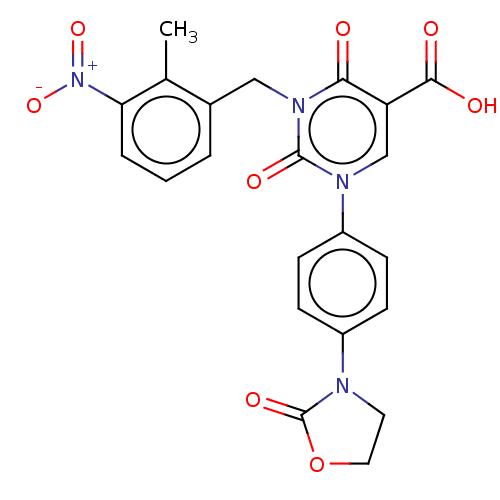
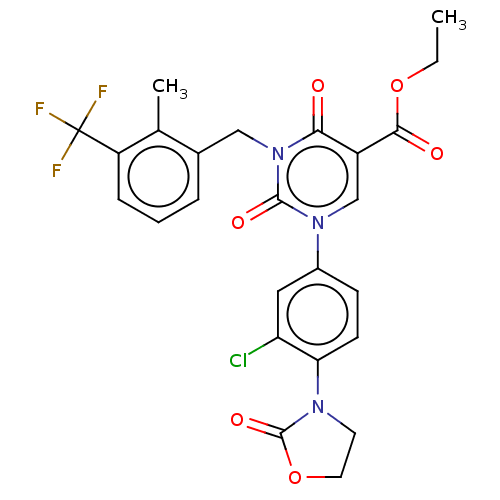
Report error Found 148 Enz. Inhib. hit(s) with all data for entry = 1743
Affinity DataIC50: 10nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 11nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 12nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 13nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 13nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 16nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 16nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 17nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 17nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 18nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 18nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 19nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 21nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 21nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 22nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 24nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 24nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 28nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 31nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 31nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 31nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 37nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 38nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 38nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 43nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 45nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 49nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 54nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 54nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 54nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 55nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 56nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 56nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 58nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 58nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 59nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 60nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 60nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 61nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 62nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 63nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 64nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 66nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 67nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 68nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 71nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 72nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 74nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 74nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
Affinity DataIC50: 76nMAssay Description:The enzyme source used is recombinant human chymase (expressed in HEK293 cells) or chymase purified from hamsters' tongues. The substrate used fo...More data for this Ligand-Target Pair
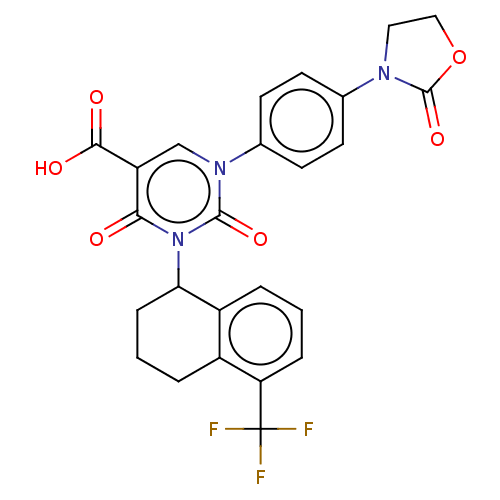
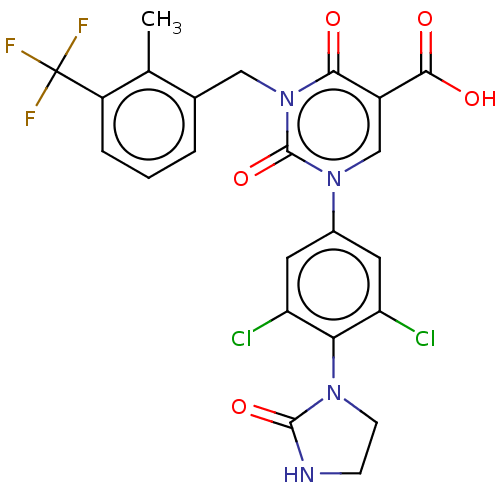
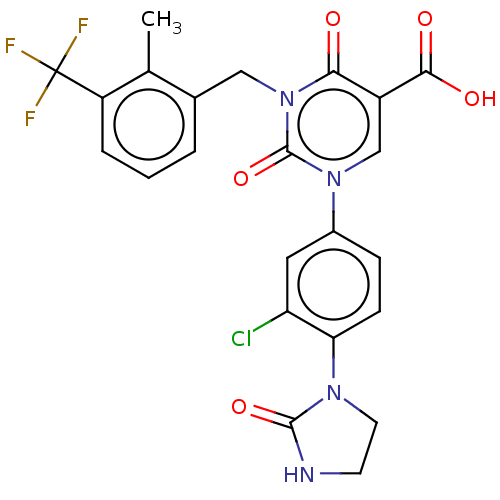
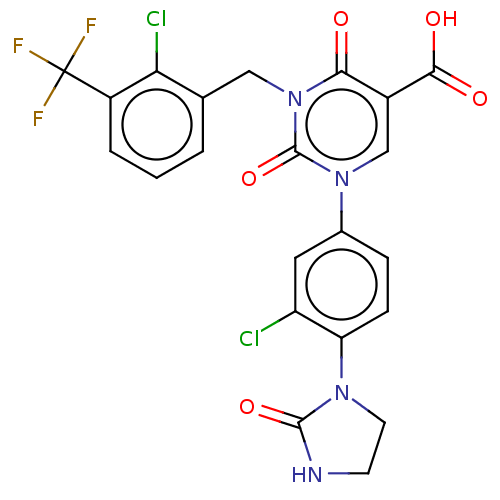
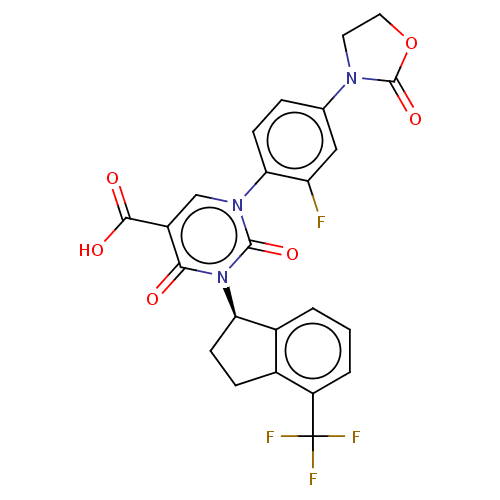
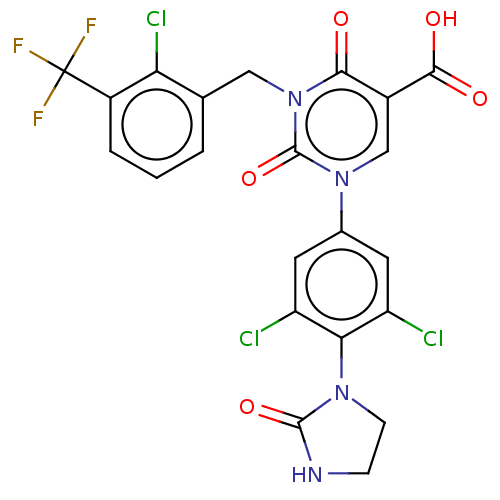
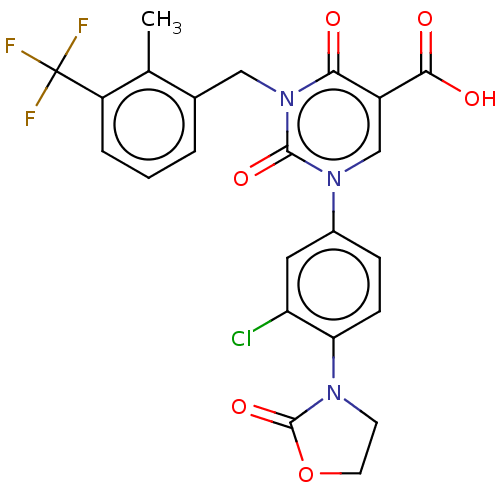
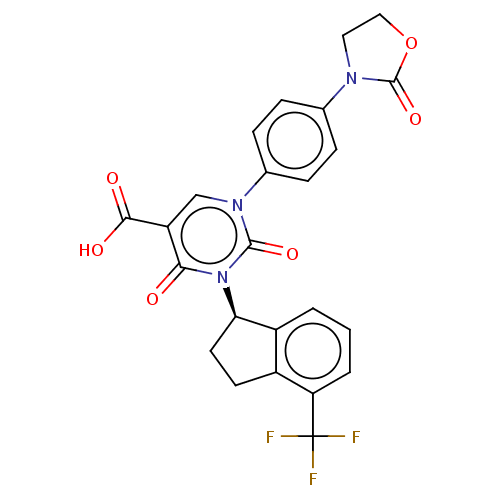
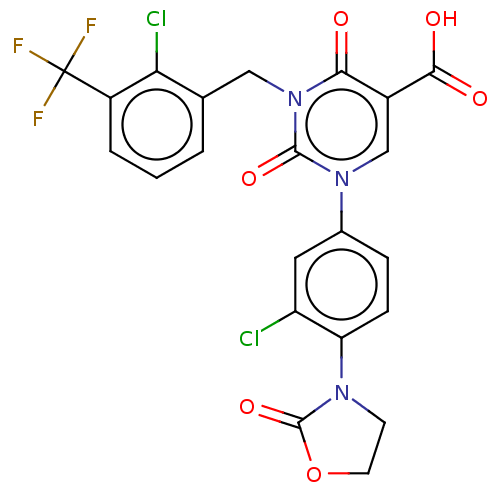
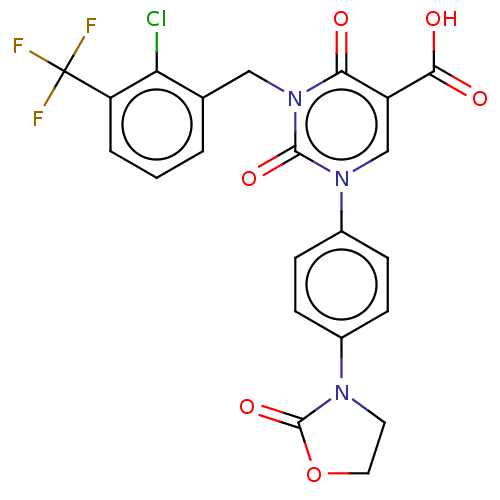
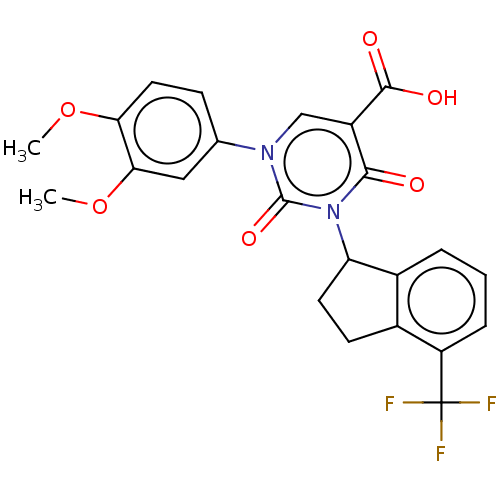
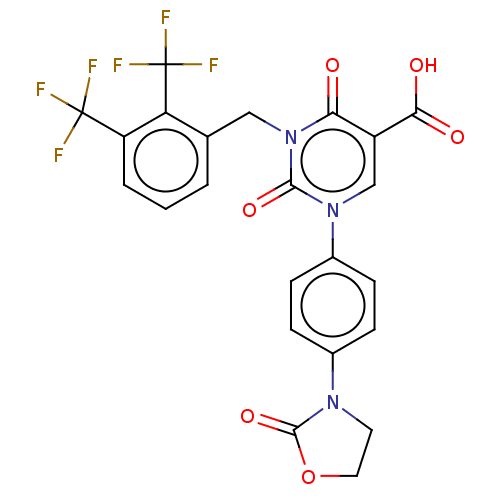
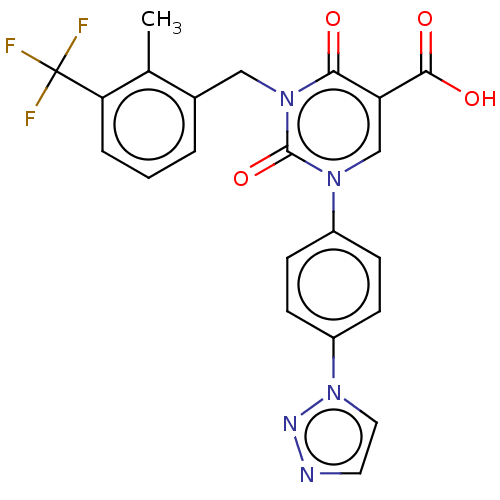
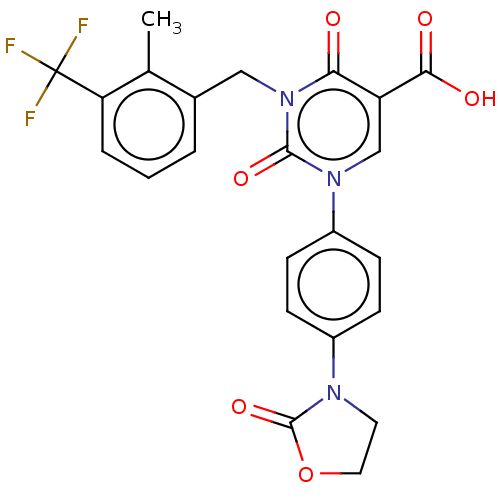
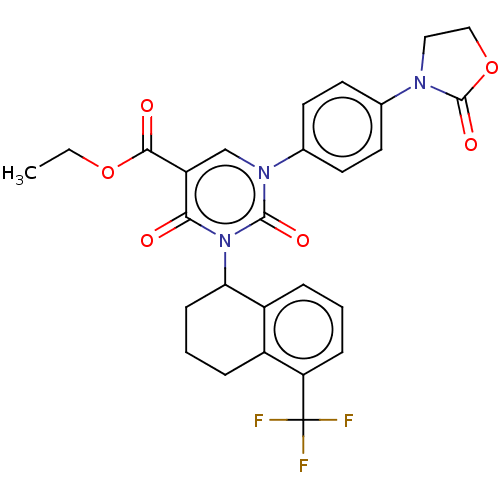
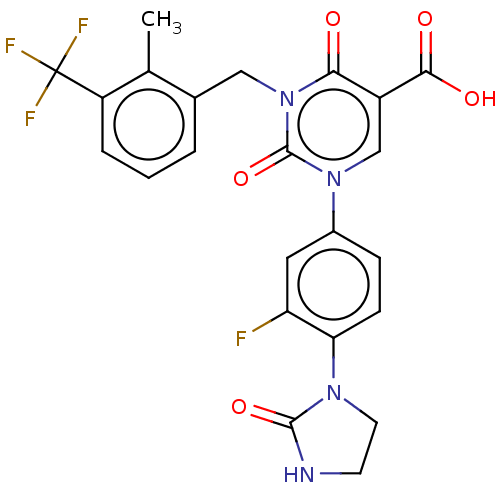
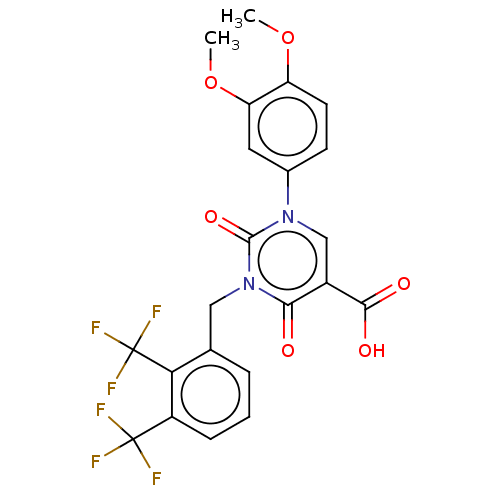
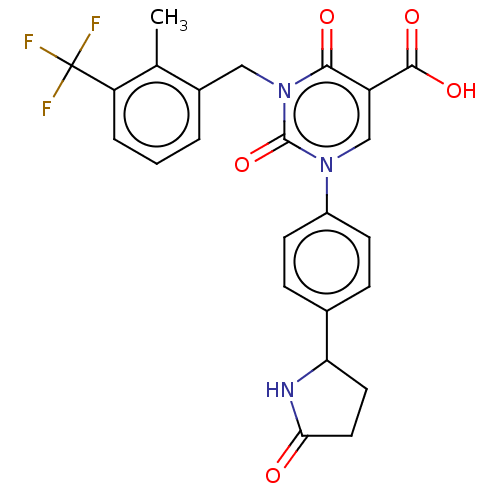
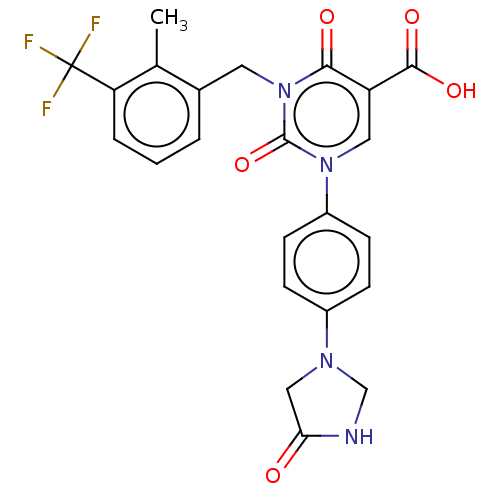
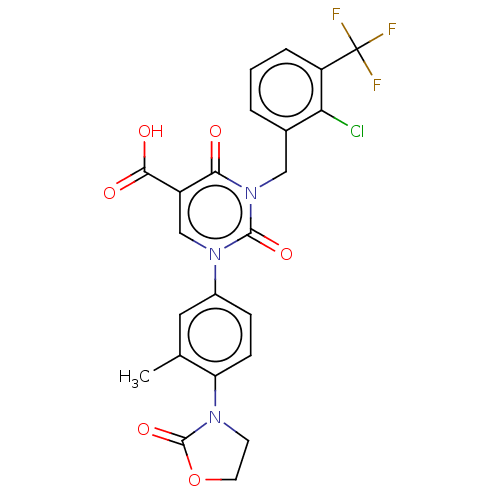
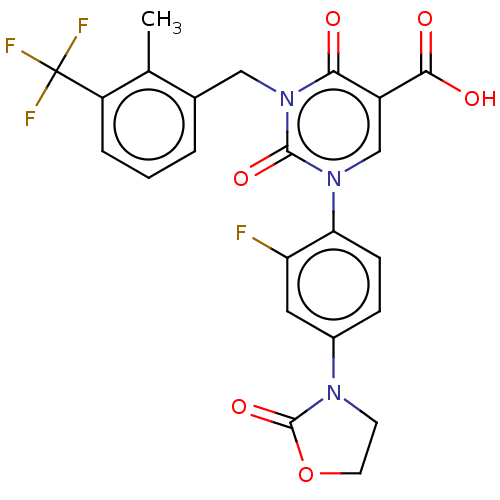
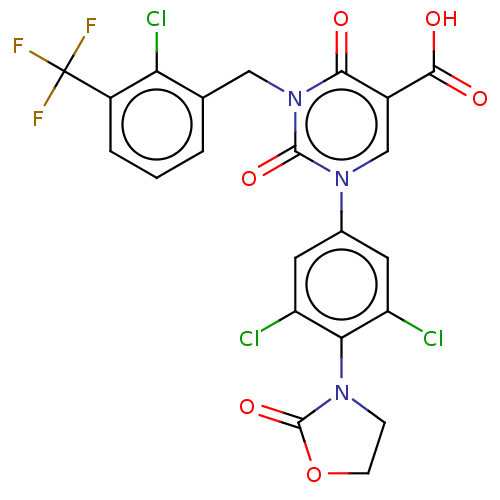
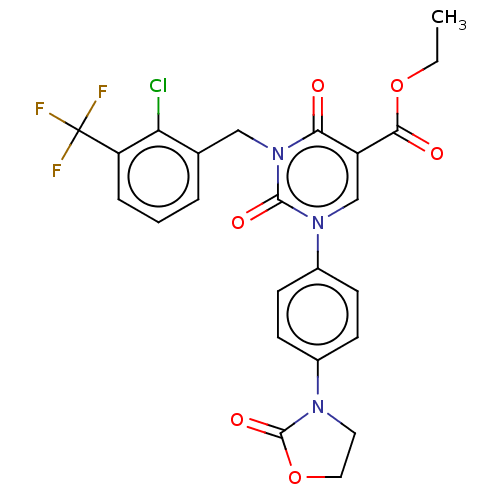
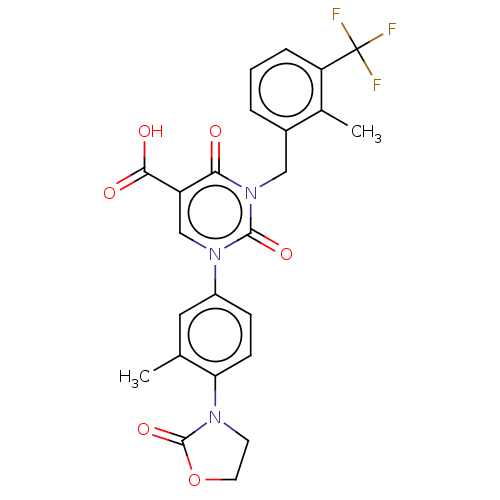
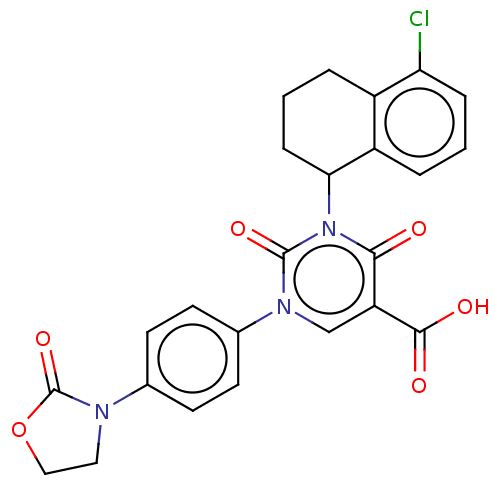
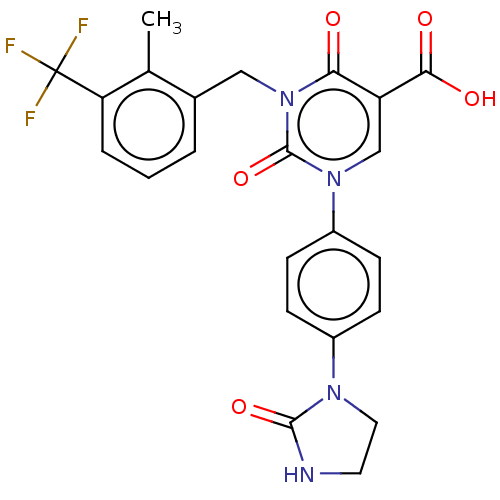
 3D Structure (crystal)
3D Structure (crystal)