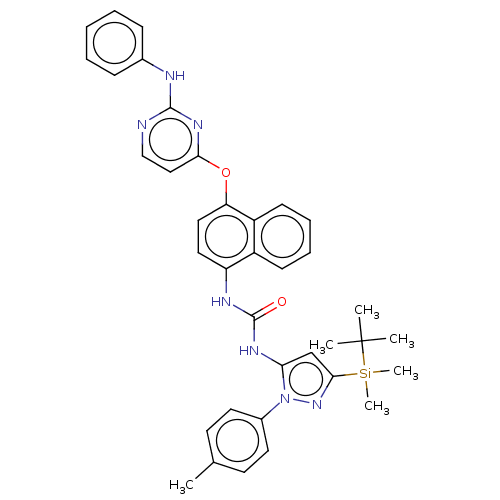
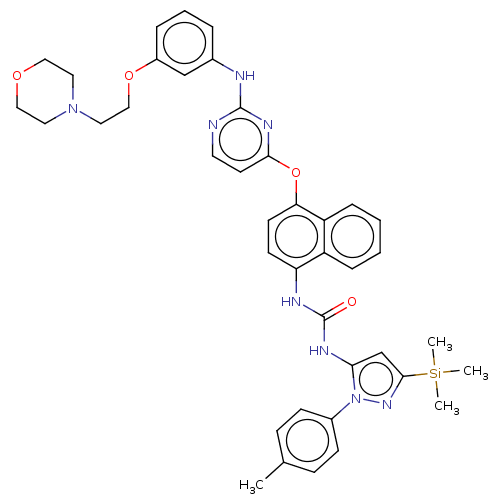
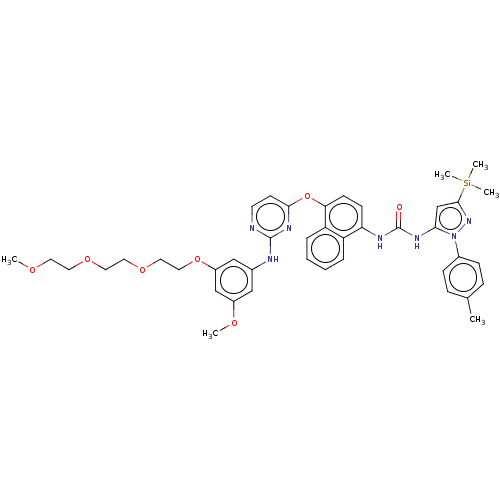
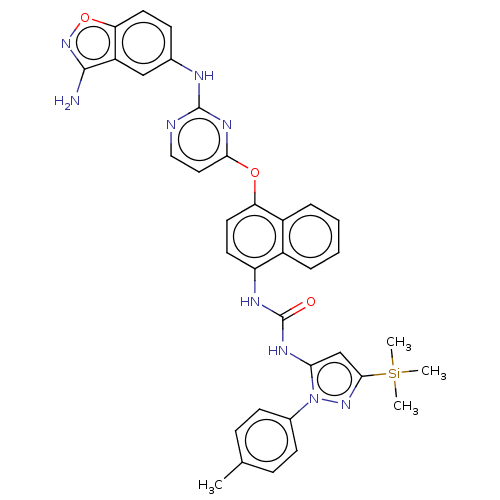
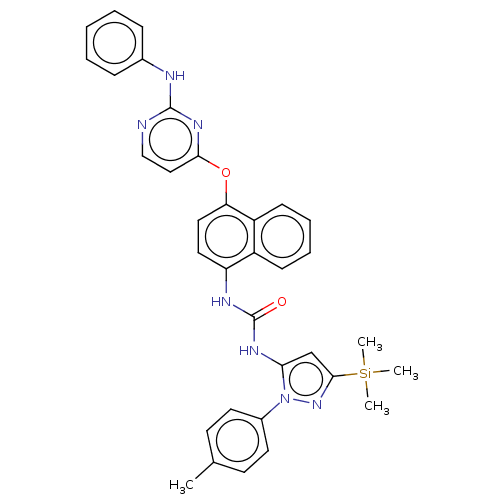
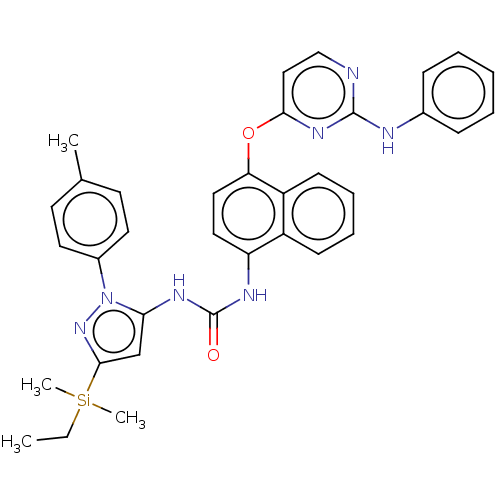
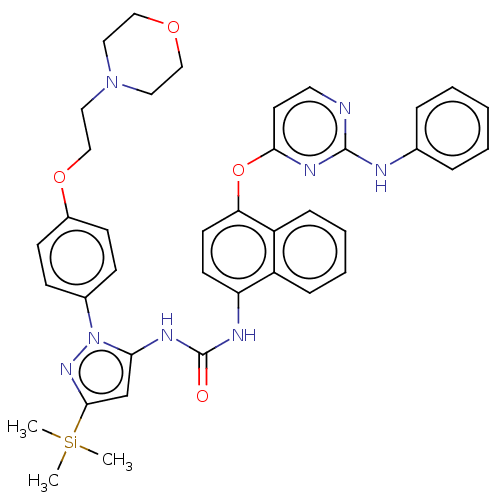
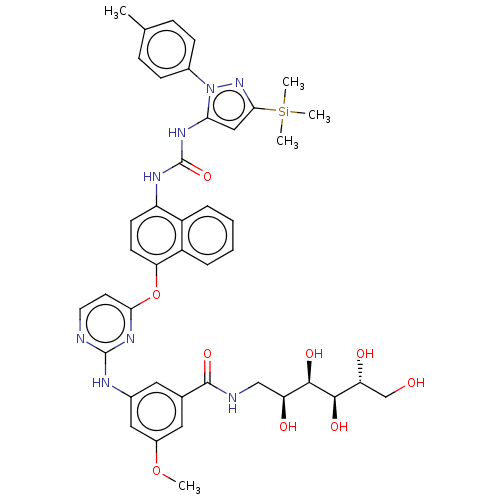
Report error Found 68 Enz. Inhib. hit(s) with all data for entry = 1867
Affinity DataIC50: 5nMAssay Description:The inhibitory activities of compounds of the invention against the GSK 3α enzyme isoform (Invitrogen), are evaluated by determining the level o...More data for this Ligand-Target Pair
Affinity DataIC50: 7nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 10nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 15nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 17nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 17nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 19nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 24nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 25nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 37nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 41nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 45nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 59nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 65nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 77nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 77.5nMAssay Description:The inhibitory activities of compounds of the invention against the GSK 3α enzyme isoform (Invitrogen), are evaluated by determining the level o...More data for this Ligand-Target Pair
Affinity DataIC50: 89nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 93nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 106nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 108nMAssay Description:The inhibitory activities of compounds of the invention against p38MAPKγ (MAPK12: Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 116nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 120nMAssay Description:The inhibitory activities of compounds of the invention against the GSK 3α enzyme isoform (Invitrogen), are evaluated by determining the level o...More data for this Ligand-Target Pair
Affinity DataIC50: 135nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 142nMAssay Description:The inhibitory activities of compounds of the invention against p38MAPKγ (MAPK12: Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 152nMAssay Description:The inhibitory activities of compounds of the invention against p38MAPKγ (MAPK12: Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 170nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 204nMAssay Description:The inhibitory activities of compounds of the invention against p38MAPKγ (MAPK12: Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 227nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 244nMAssay Description:The inhibitory activities of compounds of the invention against the GSK 3α enzyme isoform (Invitrogen), are evaluated by determining the level o...More data for this Ligand-Target Pair
Affinity DataIC50: 253nMAssay Description:The inhibitory activities of compounds of the invention against p38MAPKγ (MAPK12: Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 269nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 314nMAssay Description:The inhibitory activities of compounds of the invention against p38MAPKγ (MAPK12: Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 320nMAssay Description:The inhibitory activities of compounds of the invention against the GSK 3α enzyme isoform (Invitrogen), are evaluated by determining the level o...More data for this Ligand-Target Pair
Affinity DataIC50: 320nMAssay Description:The inhibitory activities of compounds of the invention against the GSK 3α enzyme isoform (Invitrogen), are evaluated by determining the level o...More data for this Ligand-Target Pair
Affinity DataIC50: 329nMAssay Description:The inhibitory activities of compounds of the invention against p38MAPKγ (MAPK12: Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 345nMAssay Description:The inhibitory activities of compounds of the invention against the GSK 3α enzyme isoform (Invitrogen), are evaluated by determining the level o...More data for this Ligand-Target Pair
Affinity DataIC50: 347nMAssay Description:The inhibitory activities of compounds of the invention against p38MAPKγ (MAPK12: Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 358nMAssay Description:The inhibitory activities of compounds of the invention against p38MAPKγ (MAPK12: Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 360nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 370nMAssay Description:The inhibitory activities of compounds of the invention against the GSK 3α enzyme isoform (Invitrogen), are evaluated by determining the level o...More data for this Ligand-Target Pair
Affinity DataIC50: 380nMAssay Description:The inhibitory activities of compounds of the invention against p38MAPKγ (MAPK12: Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 416nMAssay Description:The inhibitory activities of compounds of the invention against the GSK 3α enzyme isoform (Invitrogen), are evaluated by determining the level o...More data for this Ligand-Target Pair
Affinity DataIC50: 461nMAssay Description:The inhibitory activities of compounds of the invention against the GSK 3α enzyme isoform (Invitrogen), are evaluated by determining the level o...More data for this Ligand-Target Pair
Affinity DataIC50: 498nMAssay Description:The inhibitory activities of compounds of the invention against the GSK 3α enzyme isoform (Invitrogen), are evaluated by determining the level o...More data for this Ligand-Target Pair
Affinity DataIC50: 520nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 541nMAssay Description:The inhibitory activities of compounds of the invention against p38MAPKγ (MAPK12: Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 673nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair
Affinity DataIC50: 690nMAssay Description:The inhibitory activities of compounds of the invention against the GSK 3α enzyme isoform (Invitrogen), are evaluated by determining the level o...More data for this Ligand-Target Pair
Affinity DataIC50: 990nMAssay Description:The inhibitory activities of compounds of the invention against c-Src and Syk enzymes (Invitrogen), are evaluated in a similar fashion to that descri...More data for this Ligand-Target Pair