Reaction Details  Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataCell Reactant:
HIV-1 Protease Mutant (V82A/I84V)
Syringe Reactant:
BDBM577
Meas. Tech.:
Isothermal Titration Calorimetry
Entry Date.:
05/10/04
ΔG°:
-12.2±n/a (kcal/mole)
pH:
5±n/a
Log10Kb:
7.7
Temperature:
298.15±n/a (K)
ΔH° :
-5.6±0.2 (kJ/mole)
ΔHobs :
-5.6±0.2 (kJ/mole)
Corrected for ΔHioniz:
not known
ΔS° :
0.02±n/a (kJ/mole-K)
Comments:
The binding affinities were determined by using ITC displacement experiments. Acetyl pepstatin was selected as the weak inhibitor in the displacement titration because this inhibitor is endothermic and amplifies the signal of a high affinity exothermic inhibitor when displaced
Citation
Cell React
Source:
Plasmid-encoded HIV-1 protease was expressed in E. coli cells.
Purity:
99%
Prep. Method:
HIV-1 protease was purified and refolded from E. coli inclusion bodies.
Name:
HIV-1 Protease Mutant (V82A/I84V)
Synonyms:
n/a
Type:
Protein Complex
Mol. Mass.:
n/a
Description:
n/a
Components:
This complex has 2 components.
Component 1
Name:
HIV-1 Protease Mutant (V82A/I84V) chain A
Synonyms:
HIV-1 Protease Mutant (Q7K/L33I/L63I/V82A/I84V) | HIV-1 Protease Mutant (V82A/I84V) chain B
Type:
Enzyme Subunit
Mol. Mass.:
10754.16
Organism:
Human immunodeficiency virus type 1
Description:
Mutations at selected positions (V82A/I84V) were introduced into HIV-1 protease pseudo wild type (Q7K/L33I/L63I)
Residue:
99
Sequence:
PQITLWKRPLVTIKIGGQLKEALLDTGADDTVIEEMSLPGRWKPKMIGGIGGFIKVRQYDQIIIEICGHKAIGTVLVGPTPANVIGRNLLTQIGCTLNF
Component 2
Name:
HIV-1 Protease Mutant (V82A/I84V) chain A
Synonyms:
HIV-1 Protease Mutant (Q7K/L33I/L63I/V82A/I84V) | HIV-1 Protease Mutant (V82A/I84V) chain B
Type:
Enzyme Subunit
Mol. Mass.:
10754.16
Organism:
Human immunodeficiency virus type 1
Description:
Mutations at selected positions (V82A/I84V) were introduced into HIV-1 protease pseudo wild type (Q7K/L33I/L63I)
Residue:
99
Sequence:
PQITLWKRPLVTIKIGGQLKEALLDTGADDTVIEEMSLPGRWKPKMIGGIGGFIKVRQYDQIIIEICGHKAIGTVLVGPTPANVIGRNLLTQIGCTLNF
Syringe React
Source:
Purified from commercial capsules
Prep. Method:
Further purified by HPLC using a semipreparative C-18 reversed-phase column developed with 0-100% acetonitrile in 0.05% TFA.
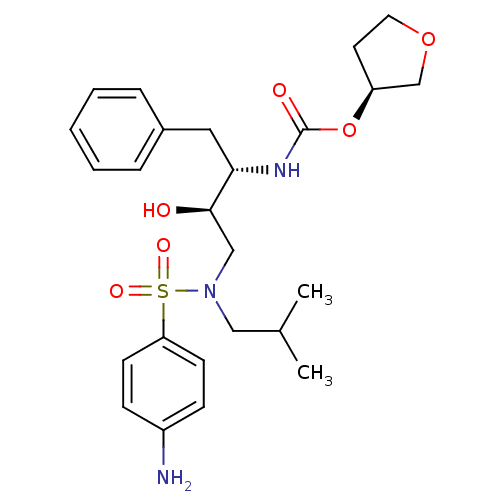
Name:
BDBM577
Synonyms:
(3S)-oxolan-3-yl N-[(2S,3R)-4-[(4-aminobenzene)(2-methylpropyl)sulfonamido]-3-hydroxy-1-phenylbutan-2-yl]carbamate | 141W94 | APV | Agenerase | Amprenavir | BDBM50215393 | CHEMBL116 | VX-478
Type:
Small organic molecule
Emp. Form.:
C25H35N3O6S
Mol. Mass.:
505.627
SMILES:
CC(C)CN(C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)O[C@H]1CCOC1)S(=O)(=O)c1ccc(N)cc1 |r|