Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cytochrome P450 2C9
Ligand
BDBM291702
Substrate
n/a
Meas. Tech.
ChEMBL_2249036 (CHEMBL5163246)
IC50
>50000±n/a nM
Citation
 Huang, Y; Sendzik, M; Zhang, J; Gao, Z; Sun, Y; Wang, L; Gu, J; Zhao, K; Yu, Z; Zhang, L; Zhang, Q; Blanz, J; Chen, Z; Dubost, V; Fang, D; Feng, L; Fu, X; Kiffe, M; Li, L; Luo, F; Luo, X; Mi, Y; Mistry, P; Pearson, D; Piaia, A; Scheufler, C; Terranova, R; Weiss, A; Zeng, J; Zhang, H; Zhang, J; Zhao, M; Dillon, MP; Jeay, S; Qi, W; Moggs, J; Pissot-Soldermann, C; Li, E; Atadja, P; Lingel, A; Oyang, C Discovery of the Clinical Candidate MAK683: An EED-Directed, Allosteric, and Selective PRC2 Inhibitor for the Treatment of Advanced Malignancies. J Med Chem 65:5317-5333 (2022) [PubMed]
Huang, Y; Sendzik, M; Zhang, J; Gao, Z; Sun, Y; Wang, L; Gu, J; Zhao, K; Yu, Z; Zhang, L; Zhang, Q; Blanz, J; Chen, Z; Dubost, V; Fang, D; Feng, L; Fu, X; Kiffe, M; Li, L; Luo, F; Luo, X; Mi, Y; Mistry, P; Pearson, D; Piaia, A; Scheufler, C; Terranova, R; Weiss, A; Zeng, J; Zhang, H; Zhang, J; Zhao, M; Dillon, MP; Jeay, S; Qi, W; Moggs, J; Pissot-Soldermann, C; Li, E; Atadja, P; Lingel, A; Oyang, C Discovery of the Clinical Candidate MAK683: An EED-Directed, Allosteric, and Selective PRC2 Inhibitor for the Treatment of Advanced Malignancies. J Med Chem 65:5317-5333 (2022) [PubMed] More Info.:
Target
Name:
Cytochrome P450 2C9
Synonyms:
(R)-limonene 6-monooxygenase | (S)-limonene 6-monooxygenase | CP2C9_HUMAN | CYP2C10 | CYP2C9 | CYPIIC9 | Cytochrome P450 2C9 (CYP2C9 ) | Cytochrome P450 2C9 (CYP2C9) | P-450MP | P450 MP-4/MP-8 | P450 PB-1 | S-mephenytoin 4-hydroxylase
Type:
Enzyme
Mol. Mass.:
55636.33
Organism:
Homo sapiens (Human)
Description:
P11712
Residue:
490
Sequence:
MDSLVVLVLCLSCLLLLSLWRQSSGRGKLPPGPTPLPVIGNILQIGIKDISKSLTNLSKVYGPVFTLYFGLKPIVVLHGYEAVKEALIDLGEEFSGRGIFPLAERANRGFGIVFSNGKKWKEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICSIIFHKRFDYKDQQFLNLMEKLNENIKILSSPWIQICNNFSPIIDYFPGTHNKLLKNVAFMKSYILEKVKEHQESMDMNNPQDFIDCFLMKMEKEKHNQPSEFTIESLENTAVDLFGAGTETTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRSHMPYTDAVVHEVQRYIDLLPTSLPHAVTCDIKFRNYLIPKGTTILISLTSVLHDNKEFPNPEMFDPHHFLDEGGNFKKSKYFMPFSAGKRICVGEALAGMELFLFLTSILQNFNLKSLVDPKNLDTTPVVNGFASVPPFYQLCFIPV
Inhibitor
Name:
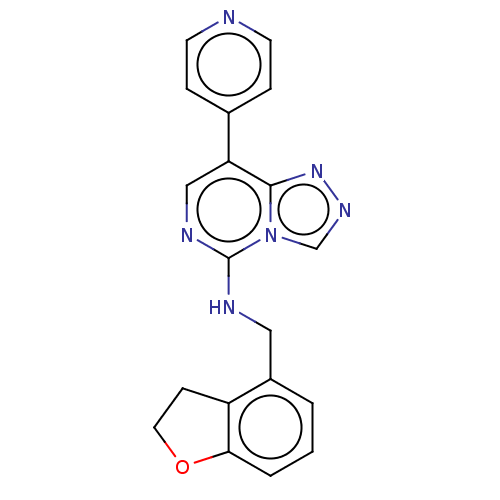
BDBM291702
Synonyms:
N-((2,3-dihydrobenzofuran-4-yl)methyl)-8-(pyridin-4-yl)-[1,2,4]triazolo[4,3-c]pyrimidin-5-amine | US11207325, Example 17 | US9580437, Example 17
Type:
Small organic molecule
Emp. Form.:
C19H16N6O
Mol. Mass.:
344.3699
SMILES:
C(Nc1ncc(-c2ccncc2)c2nncn12)c1cccc2OCCc12