Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Chymotrypsinogen A
Ligand
BDBM50098854
Substrate
n/a
Meas. Tech.
ChEMBL_49621 (CHEMBL660459)
Ki
46.8±n/a nM
Citation
 Akahoshi, F; Ashimori, A; Sakashita, H; Yoshimura, T; Imada, T; Nakajima, M; Mitsutomi, N; Kuwahara, S; Ohtsuka, T; Fukaya, C; Miyazaki, M; Nakamura, N Synthesis, structure-activity relationships, and pharmacokinetic profiles of nonpeptidic alpha-keto heterocycles as novel inhibitors of human chymase. J Med Chem 44:1286-96 (2001) [PubMed] Article
Akahoshi, F; Ashimori, A; Sakashita, H; Yoshimura, T; Imada, T; Nakajima, M; Mitsutomi, N; Kuwahara, S; Ohtsuka, T; Fukaya, C; Miyazaki, M; Nakamura, N Synthesis, structure-activity relationships, and pharmacokinetic profiles of nonpeptidic alpha-keto heterocycles as novel inhibitors of human chymase. J Med Chem 44:1286-96 (2001) [PubMed] Article More Info.:
Target
Name:
Chymotrypsinogen A
Synonyms:
Alpha-chymotrypsin | CTRA_BOVIN | Chymotrypsin A | Chymotrypsin A chain A | Chymotrypsin A chain B | Chymotrypsin A chain C | Chymotrypsinogen A | alpha-Chymotrypsin (α-Chymotrypsin)
Type:
Serine protease
Mol. Mass.:
25670.88
Organism:
Bovine
Description:
n/a
Residue:
245
Sequence:
CGVPAIQPVLSGLSRIVNGEEAVPGSWPWQVSLQDKTGFHFCGGSLINENWVVTAAHCGVTTSDVVVAGEFDQGSSSEKIQKLKIAKVFKNSKYNSLTINNDITLLKLSTAASFSQTVSAVCLPSASDDFAAGTTCVTTGWGLTRYTNANTPDRLQQASLPLLSNTNCKKYWGTKIKDAMICAGASGVSSCMGDSGGPLVCKKNGAWTLVGIVSWGSSTCSTSTPGVYARVTALVNWVQQTLAAN
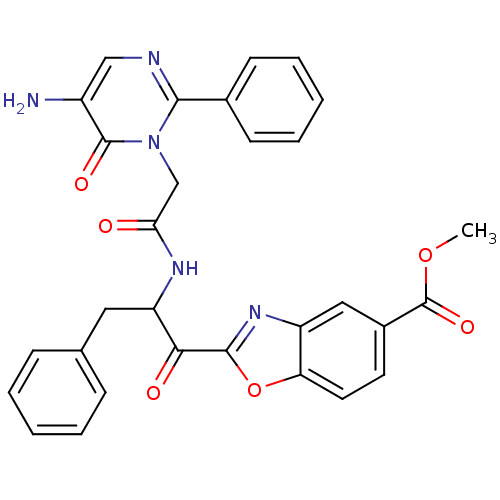
Inhibitor
Name:
BDBM50098854
Synonyms:
2-{2-[2-(5-Amino-6-oxo-2-phenyl-6H-pyrimidin-1-yl)-acetylamino]-3-phenyl-propionyl}-benzooxazole-5-carboxylic acid methyl ester | CHEMBL26297
Type:
Small organic molecule
Emp. Form.:
C30H25N5O6
Mol. Mass.:
551.5494
SMILES:
COC(=O)c1ccc2oc(nc2c1)C(=O)C(Cc1ccccc1)NC(=O)Cn1c(ncc(N)c1=O)-c1ccccc1