Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cathepsin B
Ligand
BDBM50335282
Substrate
n/a
Meas. Tech.
ChEMBL_702785 (CHEMBL1655228)
Ki
2.8±n/a nM
Citation
 Frizler, M; Lohr, F; Furtmann, N; Kläs, J; Gütschow, M Structural optimization of azadipeptide nitriles strongly increases association rates and allows the development of selective cathepsin inhibitors. J Med Chem 54:396-400 (2011) [PubMed] Article
Frizler, M; Lohr, F; Furtmann, N; Kläs, J; Gütschow, M Structural optimization of azadipeptide nitriles strongly increases association rates and allows the development of selective cathepsin inhibitors. J Med Chem 54:396-400 (2011) [PubMed] Article More Info.:
Target
Name:
Cathepsin B
Synonyms:
APP secretase | APPS | CATB_HUMAN | CPSB | CTSB | Cathepsin B heavy chain | Cathepsin B light chain | Cathepsin B1
Type:
Enzyme
Mol. Mass.:
37819.69
Organism:
Homo sapiens (Human)
Description:
gi_63102437
Residue:
339
Sequence:
MWQLWASLCCLLVLANARSRPSFHPLSDELVNYVNKRNTTWQAGHNFYNVDMSYLKRLCGTFLGGPKPPQRVMFTEDLKLPASFDAREQWPQCPTIKEIRDQGSCGSCWAFGAVEAISDRICIHTNAHVSVEVSAEDLLTCCGSMCGDGCNGGYPAEAWNFWTRKGLVSGGLYESHVGCRPYSIPPCEHHVNGSRPPCTGEGDTPKCSKICEPGYSPTYKQDKHYGYNSYSVSNSEKDIMAEIYKNGPVEGAFSVYSDFLLYKSGVYQHVTGEMMGGHAIRILGWGVENGTPYWLVANSWNTDWGDNGFFKILRGQDHCGIESEVVAGIPRTDQYWEKI
Inhibitor
Name:
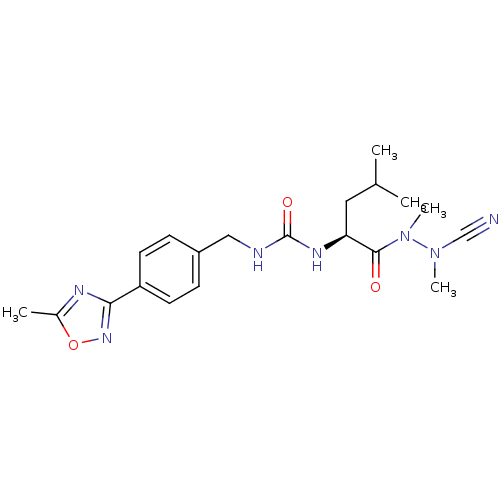
BDBM50335282
Synonyms:
CHEMBL1651356 | N-[4-(5-Methyl-1,2,4-oxadiazol-3-yl)benzylcarbamoyl]-leucyl-methylazaalanine-nitrile | acs.jmedchem.1c00409_ST.405
Type:
Small organic molecule
Emp. Form.:
C20H27N7O3
Mol. Mass.:
413.4735
SMILES:
CC(C)C[C@H](NC(=O)NCc1ccc(cc1)-c1noc(C)n1)C(=O)N(C)N(C)C#N |r|