Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Phospho-N-acetylmuramoyl-pentapeptide-transferase
Ligand
BDBM50386967
Substrate
n/a
Meas. Tech.
ChEMBL_827497 (CHEMBL2051354)
IC50
8900±n/a nM
Citation
 Okamoto, K; Sakagami, M; Feng, F; Takahashi, F; Uotani, K; Togame, H; Takemoto, H; Ichikawa, S; Matsuda, A Synthesis of pacidamycin analogues via an Ugi-multicomponent reaction. Bioorg Med Chem Lett 22:4810-5 (2012) [PubMed] Article
Okamoto, K; Sakagami, M; Feng, F; Takahashi, F; Uotani, K; Togame, H; Takemoto, H; Ichikawa, S; Matsuda, A Synthesis of pacidamycin analogues via an Ugi-multicomponent reaction. Bioorg Med Chem Lett 22:4810-5 (2012) [PubMed] Article More Info.:
Target
Name:
Phospho-N-acetylmuramoyl-pentapeptide-transferase
Synonyms:
MRAY_STAAU | mraY
Type:
PROTEIN
Mol. Mass.:
35238.55
Organism:
Staphylococcus aureus (strain MRSA252)
Description:
ChEMBL_827497
Residue:
321
Sequence:
MIFVYALLALVITFVLVPVLIPTLKRMKFGQSIREEGPQSHMKKTGTPTMGGLTFLLSIVITSLVAIIFVDQANPIILLLFVTIGFGLIGFIDDYIIVVKKNNQGLTSKQKFLAQIGIAIIFFVLSNVFHLVNFSTSIHIPFTNVAIPLSFAYVIFIVFWQVGFSNAVNLTDGLDGLATGLSIIGFTMYAIMSFVLGETAIGIFCIIMLFALLGFLPYNINPAKVFMGDTGSLALGGIFATISIMLNQELSLIFIGLVFVIETLSVMLQVASFKLTGKRIFKMSPIHHHFELIGWSEWKVVTVFWAVGLISGLIGLWIGVH
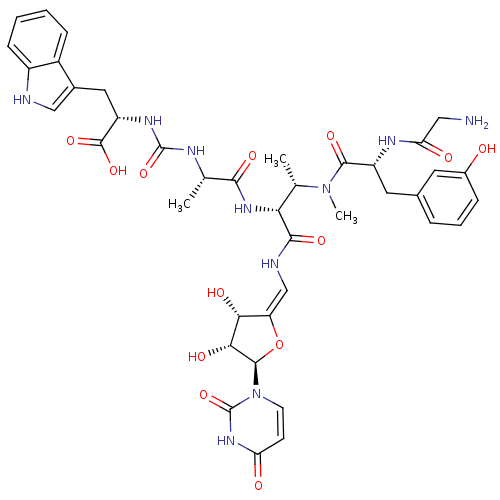
Inhibitor
Name:
BDBM50386967
Synonyms:
CHEMBL2048832
Type:
Small organic molecule
Emp. Form.:
C40H48N10O13
Mol. Mass.:
876.8683
SMILES:
C[C@H](NC(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(O)=O)C(=O)N[C@H]([C@H](C)N(C)C(=O)[C@@H](Cc1cccc(O)c1)NC(=O)CN)C(=O)N\C=C1/O[C@H]([C@H](O)[C@@H]1O)n1ccc(=O)[nH]c1=O |r|