Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Dual specificity mitogen-activated protein kinase kinase 1
Ligand
BDBM50391802
Substrate
n/a
Meas. Tech.
ChEMBL_850079 (CHEMBL2149502)
IC50
0.9±n/a nM
Citation
 Rice, KD; Aay, N; Anand, NK; Blazey, CM; Bowles, OJ; Bussenius, J; Costanzo, S; Curtis, JK; Defina, SC; Dubenko, L; Engst, S; Joshi, AA; Kennedy, AR; Kim, AI; Koltun, ES; Lougheed, JC; Manalo, JC; Martini, JF; Nuss, JM; Peto, CJ; Tsang, TH; Yu, P; Johnston, S Novel Carboxamide-Based Allosteric MEK Inhibitors: Discovery and Optimization Efforts toward XL518 (GDC-0973). ACS Med Chem Lett 3:416-421 (2012) [PubMed] Article
Rice, KD; Aay, N; Anand, NK; Blazey, CM; Bowles, OJ; Bussenius, J; Costanzo, S; Curtis, JK; Defina, SC; Dubenko, L; Engst, S; Joshi, AA; Kennedy, AR; Kim, AI; Koltun, ES; Lougheed, JC; Manalo, JC; Martini, JF; Nuss, JM; Peto, CJ; Tsang, TH; Yu, P; Johnston, S Novel Carboxamide-Based Allosteric MEK Inhibitors: Discovery and Optimization Efforts toward XL518 (GDC-0973). ACS Med Chem Lett 3:416-421 (2012) [PubMed] Article More Info.:
Target
Name:
Dual specificity mitogen-activated protein kinase kinase 1
Synonyms:
Dual specificity mitogen-activated protein kinase (MEK) | Dual specificity mitogen-activated protein kinase kinase 1 (MEK) | Dual specificity mitogen-activated protein kinase kinase 1 (MEK1) | Dual specificity mitogen-activated protein kinase kinase 1/Mitogen-activated protein kinase 1/RAF proto-oncogene serine/threonine-protein kinase | Dual specificity mitogen-activated protein kinase kinase MEK1/2 | ERK activator kinase 1 | MAP kinase kinase 1 | MAP2K1 | MAPK/ERK kinase 1 | MAPK/ERK kinase 1 (MEK1) | MEK-1 | MEK1 | MP2K1_HUMAN | Mitogen-activated protein kinase 1 (MEK1) | PRKMK1 | VHL-MAP2K1/MAP2K2
Type:
Other Protein Type
Mol. Mass.:
43439.03
Organism:
Human
Description:
Full-length human MEK-1 was generated by PCR and purified as a fusion protein from Escherichia coli lysates.
Residue:
393
Sequence:
MPKKKPTPIQLNPAPDGSAVNGTSSAETNLEALQKKLEELELDEQQRKRLEAFLTQKQKVGELKDDDFEKISELGAGNGGVVFKVSHKPSGLVMARKLIHLEIKPAIRNQIIRELQVLHECNSPYIVGFYGAFYSDGEISICMEHMDGGSLDQVLKKAGRIPEQILGKVSIAVIKGLTYLREKHKIMHRDVKPSNILVNSRGEIKLCDFGVSGQLIDSMANSFVGTRSYMSPERLQGTHYSVQSDIWSMGLSLVEMAVGRYPIPPPDAKELELMFGCQVEGDAAETPPRPRTPGRPLSSYGMDSRPPMAIFELLDYIVNEPPPKLPSGVFSLEFQDFVNKCLIKNPAERADLKQLMVHAFIKRSDAEEVDFAGWLCSTIGLNQPSTPTHAAGV
Inhibitor
Name:
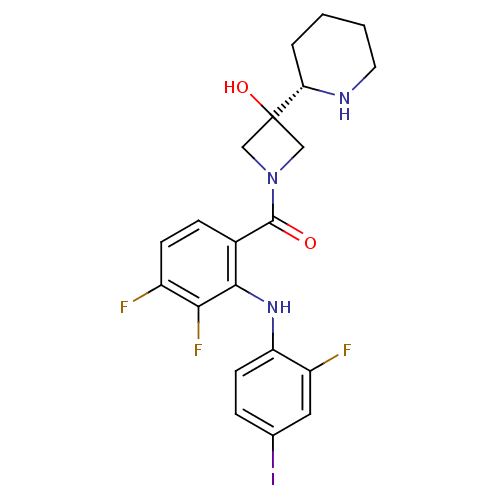
BDBM50391802
Synonyms:
(3,4-difluoro-2-(2-fluoro-4-iodophenylamino)phenyl)(3-hydroxy-3-(piperidin-2-yl)azetidin-1-yl)methanone | CHEMBL2146883 | COBIMETINIB FUMARATE | Cotellic | GDC-0973 | XL518 | cobimetinib
Type:
Small organic molecule
Emp. Form.:
C21H21F3IN3O2
Mol. Mass.:
531.31
SMILES:
OC1(CN(C1)C(=O)c1ccc(F)c(F)c1Nc1ccc(I)cc1F)[C@@H]1CCCCN1 |r|