Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Histamine H3 receptor
Ligand
BDBM50401001
Substrate
n/a
Meas. Tech.
ChEMBL_879382 (CHEMBL2208780)
Ki
7.7±n/a nM
Citation
 Wager, TT; Pettersen, BA; Schmidt, AW; Spracklin, DK; Mente, S; Butler, TW; Howard, H; Lettiere, DJ; Rubitski, DM; Wong, DF; Nedza, FM; Nelson, FR; Rollema, H; Raggon, JW; Aubrecht, J; Freeman, JK; Marcek, JM; Cianfrogna, J; Cook, KW; James, LC; Chatman, LA; Iredale, PA; Banker, MJ; Homiski, ML; Munzner, JB; Chandrasekaran, RY Discovery of two clinical histamine H(3) receptor antagonists: trans-N-ethyl-3-fluoro-3-[3-fluoro-4-(pyrrolidinylmethyl)phenyl]cyclobutanecarboxamide (PF-03654746) and trans-3-fluoro-3-[3-fluoro-4-(pyrrolidin-1-ylmethyl)phenyl]-N-(2-methylpropyl)cyclobutanecarboxamide (PF-03654764). J Med Chem 54:7602-20 (2011) [PubMed] Article
Wager, TT; Pettersen, BA; Schmidt, AW; Spracklin, DK; Mente, S; Butler, TW; Howard, H; Lettiere, DJ; Rubitski, DM; Wong, DF; Nedza, FM; Nelson, FR; Rollema, H; Raggon, JW; Aubrecht, J; Freeman, JK; Marcek, JM; Cianfrogna, J; Cook, KW; James, LC; Chatman, LA; Iredale, PA; Banker, MJ; Homiski, ML; Munzner, JB; Chandrasekaran, RY Discovery of two clinical histamine H(3) receptor antagonists: trans-N-ethyl-3-fluoro-3-[3-fluoro-4-(pyrrolidinylmethyl)phenyl]cyclobutanecarboxamide (PF-03654746) and trans-3-fluoro-3-[3-fluoro-4-(pyrrolidin-1-ylmethyl)phenyl]-N-(2-methylpropyl)cyclobutanecarboxamide (PF-03654764). J Med Chem 54:7602-20 (2011) [PubMed] Article More Info.:
Target
Name:
Histamine H3 receptor
Synonyms:
HH3R | HRH3_RAT | Hrh3
Type:
G Protein-Coupled Receptor (GPCR)
Mol. Mass.:
48607.98
Organism:
Rattus norvegicus (rat)
Description:
n/a
Residue:
445
Sequence:
MERAPPDGLMNASGTLAGEAAAAGGARGFSAAWTAVLAALMALLIVATVLGNALVMLAFVADSSLRTQNNFFLLNLAISDFLVGAFCIPLYVPYVLTGRWTFGRGLCKLWLVVDYLLCASSVFNIVLISYDRFLSVTRAVSYRAQQGDTRRAVRKMALVWVLAFLLYGPAILSWEYLSGGSSIPEGHCYAEFFYNWYFLITASTLEFFTPFLSVTFFNLSIYLNIQRRTRLRLDGGREAGPEPPPDAQPSPPPAPPSCWGCWPKGHGEAMPLHRYGVGEAGPGVEAGEAALGGGSGGGAAASPTSSSGSSSRGTERPRSLKRGSKPSASSASLEKRMKMVSQSITQRFRLSRDKKVAKSLAIIVSIFGLCWAPYTLLMIIRAACHGRCIPDYWYETSFWLLWANSAVNPVLYPLCHYSFRRAFTKLLCPQKLKVQPHGSLEQCWK
Inhibitor
Name:
BDBM50401001
Synonyms:
CHEMBL2206288
Type:
Small organic molecule
Emp. Form.:
C20H30N2O
Mol. Mass.:
314.465
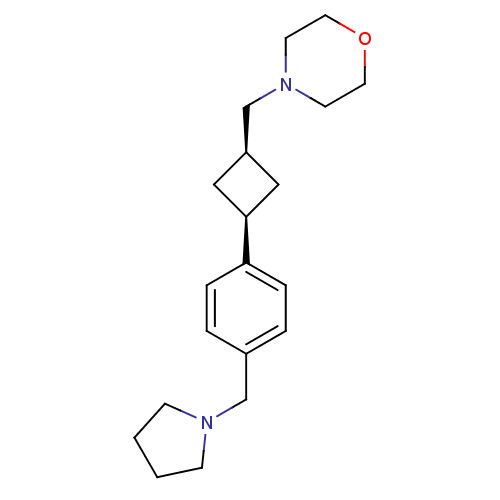
SMILES:
C([C@H]1C[C@H](C1)c1ccc(CN2CCCC2)cc1)N1CCOCC1 |r,wU:3.5,1.0,(12.44,-17.63,;13.93,-17.23,;14.7,-15.89,;16.03,-16.66,;15.26,-18,;17.52,-16.26,;18.61,-17.35,;20.1,-16.95,;20.5,-15.47,;21.98,-15.07,;23.07,-16.16,;22.83,-17.68,;24.2,-18.38,;25.29,-17.29,;24.59,-15.92,;19.41,-14.38,;17.92,-14.78,;11.35,-16.54,;11.75,-15.05,;10.66,-13.96,;9.18,-14.36,;8.78,-15.85,;9.87,-16.93,)|