Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Apolipoprotein(a)
Ligand
BDBM544455
Substrate
n/a
Meas. Tech.
In Vitro Apo(a) Binding Assay
IC50
<0.314±n/a nM
Citation
 Blanco, CL; Buezo, ND; Perez, JA; Sanz Gil, GC; Soler, JP Pyrrolidine compounds US Patent US11286249 Publication Date 3/29/2022
Blanco, CL; Buezo, ND; Perez, JA; Sanz Gil, GC; Soler, JP Pyrrolidine compounds US Patent US11286249 Publication Date 3/29/2022 More Info.:
Target
Name:
Apolipoprotein(a)
Synonyms:
APOA_HUMAN | Apo(a) | LPA | Lp(a)
Type:
Protein
Mol. Mass.:
226526.93
Organism:
Human
Description:
P08519
Residue:
2040
Sequence:
MEHKEVVLLLLLFLKSAAPEQSHVVQDCYHGDGQSYRGTYSTTVTGRTCQAWSSMTPHQHNRTTENYPNAGLIMNYCRNPDAVAAPYCYTRDPGVRWEYCNLTQCSDAEGTAVAPPTVTPVPSLEAPSEQAPTEQRPGVQECYHGNGQSYRGTYSTTVTGRTCQAWSSMTPHSHSRTPEYYPNAGLIMNYCRNPDAVAAPYCYTRDPGVRWEYCNLTQCSDAEGTAVAPPTVTPVPSLEAPSEQAPTEQRPGVQECYHGNGQSYRGTYSTTVTGRTCQAWSSMTPHSHSRTPEYYPNAGLIMNYCRNPDAVAAPYCYTRDPGVRWEYCNLTQCSDAEGTAVAPPTVTPVPSLEAPSEQAPTEQRPGVQECYHGNGQSYRGTYSTTVTGRTCQAWSSMTPHSHSRTPEYYPNAGLIMNYCRNPDAVAAPYCYTRDPGVRWEYCNLTQCSDAEGTAVAPPTVTPVPSLEAPSEQAPTEQRPGVQECYHGNGQSYRGTYSTTVTGRTCQAWSSMTPHSHSRTPEYYPNAGLIMNYCRNPDAVAAPYCYTRDPGVRWEYCNLTQCSDAEGTAVAPPTVTPVPSLEAPSEQAPTEQRPGVQECYHGNGQSYRGTYSTTVTGRTCQAWSSMTPHSHSRTPEYYPNAGLIMNYCRNPDAVAAPYCYTRDPGVRWEYCNLTQCSDAEGTAVAPPTVTPVPSLEAPSEQAPTEQRPGVQECYHGNGQSYRGTYSTTVTGRTCQAWSSMTPHSHSRTPEYYPNAGLIMNYCRNPDAVAAPYCYTRDPGVRWEYCNLTQCSDAEGTAVAPPTVTPVPSLEAPSEQAPTEQRPGVQECYHGNGQSYRGTYSTTVTGRTCQAWSSMTPHSHSRTPEYYPNAGLIMNYCRNPDPVAAPYCYTRDPSVRWEYCNLTQCSDAEGTAVAPPTITPIPSLEAPSEQAPTEQRPGVQECYHGNGQSYQGTYFITVTGRTCQAWSSMTPHSHSRTPAYYPNAGLIKNYCRNPDPVAAPWCYTTDPSVRWEYCNLTRCSDAEWTAFVPPNVILAPSLEAFFEQALTEETPGVQDCYYHYGQSYRGTYSTTVTGRTCQAWSSMTPHQHSRTPENYPNAGLTRNYCRNPDAEIRPWCYTMDPSVRWEYCNLTQCLVTESSVLATLTVVPDPSTEASSEEAPTEQSPGVQDCYHGDGQSYRGSFSTTVTGRTCQSWSSMTPHWHQRTTEYYPNGGLTRNYCRNPDAEISPWCYTMDPNVRWEYCNLTQCPVTESSVLATSTAVSEQAPTEQSPTVQDCYHGDGQSYRGSFSTTVTGRTCQSWSSMTPHWHQRTTEYYPNGGLTRNYCRNPDAEIRPWCYTMDPSVRWEYCNLTQCPVMESTLLTTPTVVPVPSTELPSEEAPTENSTGVQDCYRGDGQSYRGTLSTTITGRTCQSWSSMTPHWHRRIPLYYPNAGLTRNYCRNPDAEIRPWCYTMDPSVRWEYCNLTRCPVTESSVLTTPTVAPVPSTEAPSEQAPPEKSPVVQDCYHGDGRSYRGISSTTVTGRTCQSWSSMIPHWHQRTPENYPNAGLTENYCRNPDSGKQPWCYTTDPCVRWEYCNLTQCSETESGVLETPTVVPVPSMEAHSEAAPTEQTPVVRQCYHGNGQSYRGTFSTTVTGRTCQSWSSMTPHRHQRTPENYPNDGLTMNYCRNPDADTGPWCFTMDPSIRWEYCNLTRCSDTEGTVVAPPTVIQVPSLGPPSEQDCMFGNGKGYRGKKATTVTGTPCQEWAAQEPHRHSTFIPGTNKWAGLEKNYCRNPDGDINGPWCYTMNPRKLFDYCDIPLCASSSFDCGKPQVEPKKCPGSIVGGCVAHPHSWPWQVSLRTRFGKHFCGGTLISPEWVLTAAHCLKKSSRPSSYKVILGAHQEVNLESHVQEIEVSRLFLEPTQADIALLKLSRPAVITDKVMPACLPSPDYMVTARTECYITGWGETQGTFGTGLLKEAQLLVIENEVCNHYKYICAEHLARGTDSCQGDSGGPLVCFEKDKYILQGVTSWGLGCARPNKPGVYARVSRFVTWIEGMMRNN
Inhibitor
Name:
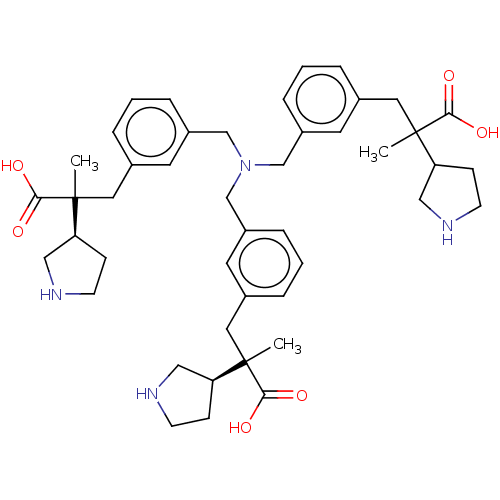
BDBM544455
Synonyms:
3-[3-[[bis[[3-[2-carboxy-2-[(3R)-pyrrolidin-3-yl]propyl]phenyl]methyl]amino]methyl]phenyl]-2-methyl-2-[(3R)-pyrrolidin-3-yl]propanoic acid; tetrahydrochloride | US11286249, Example 6
Type:
Small organic molecule
Emp. Form.:
C45H60N4O6
Mol. Mass.:
752.9811
SMILES:
CC(Cc1cccc(CN(Cc2cccc(CC(C)([C@H]3CCNC3)C(O)=O)c2)Cc2cccc(CC(C)([C@H]3CCNC3)C(O)=O)c2)c1)(C1CCNC1)C(O)=O |r|