Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cholinesterase
Ligand
BDBM10988
Substrate
BDBM8978
Meas. Tech.
Cholinesterase Inhibition Assay
IC50
3890±n/a nM
Citation
 Yu, Q; Holloway, HW; Flippen-Anderson, JL; Hoffman, B; Brossi, A; Greig, NH Methyl analogues of the experimental Alzheimer drug phenserine: synthesis and structure/activity relationships for acetyl- and butyrylcholinesterase inhibitory action. J Med Chem 44:4062-71 (2001) [PubMed] Article
Yu, Q; Holloway, HW; Flippen-Anderson, JL; Hoffman, B; Brossi, A; Greig, NH Methyl analogues of the experimental Alzheimer drug phenserine: synthesis and structure/activity relationships for acetyl- and butyrylcholinesterase inhibitory action. J Med Chem 44:4062-71 (2001) [PubMed] Article More Info.:
Target
Name:
Cholinesterase
Synonyms:
Acylcholine acylhydrolase | BCHE | Butyrylcholine esterase (BChE) | Butyrylcholinesterase (BChE) | Butyrylcholinesterase (BuChE) | CHE1 | CHLE_HUMAN | Choline esterase II | Cholinesterases | Cholinesterases; ACHE & BCHE | Pseudocholinesterase
Type:
Homotetramer
Mol. Mass.:
68422.27
Organism:
Homo sapiens (Human)
Description:
P06276
Residue:
602
Sequence:
MHSKVTIICIRFLFWFLLLCMLIGKSHTEDDIIIATKNGKVRGMNLTVFGGTVTAFLGIPYAQPPLGRLRFKKPQSLTKWSDIWNATKYANSCCQNIDQSFPGFHGSEMWNPNTDLSEDCLYLNVWIPAPKPKNATVLIWIYGGGFQTGTSSLHVYDGKFLARVERVIVVSMNYRVGALGFLALPGNPEAPGNMGLFDQQLALQWVQKNIAAFGGNPKSVTLFGESAGAASVSLHLLSPGSHSLFTRAILQSGSFNAPWAVTSLYEARNRTLNLAKLTGCSRENETEIIKCLRNKDPQEILLNEAFVVPYGTPLSVNFGPTVDGDFLTDMPDILLELGQFKKTQILVGVNKDEGTAFLVYGAPGFSKDNNSIITRKEFQEGLKIFFPGVSEFGKESILFHYTDWVDDQRPENYREALGDVVGDYNFICPALEFTKKFSEWGNNAFFYYFEHRSSKLPWPEWMGVMHGYEIEFVFGLPLERRDNYTKAEEILSRSIVKRWANFAKYGNPNETQNNSTSWPVFKSTEQKYLTLNTESTRIMTKLRAQQCRFWTSFFPKVLEMTGNIDEAEWEWKAGFHRWNNYMMDWKNQFNDYTSKKESCVGL
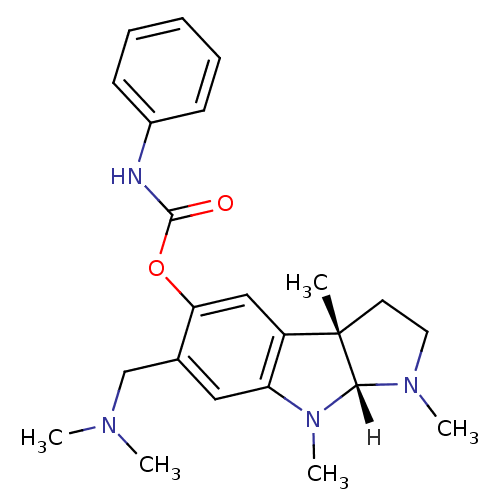
Inhibitor
Name:
BDBM10988
Synonyms:
(3aS,8aR)-6-[(dimethylamino)methyl]-1,3a,8-trimethyl-1,2,3,3a,8,8a-hexahydropyrrolo[2,3-b]indol-5-yl phenylcarbamate | (3aS,8aR)-6-[(dimethylamino)methyl]-1,3a,8-trimethyl-1H,2H,3H,3aH,8H,8aH-pyrrolo[2,3-b]indol-5-yl N-phenylcarbamate | Phenserine analog 32
Type:
Small organic molecule
Emp. Form.:
C23H30N4O2
Mol. Mass.:
394.5099
SMILES:
[H][C@]12N(C)CC[C@@]1(C)c1cc(OC(=O)Nc3ccccc3)c(CN(C)C)cc1N2C |r|
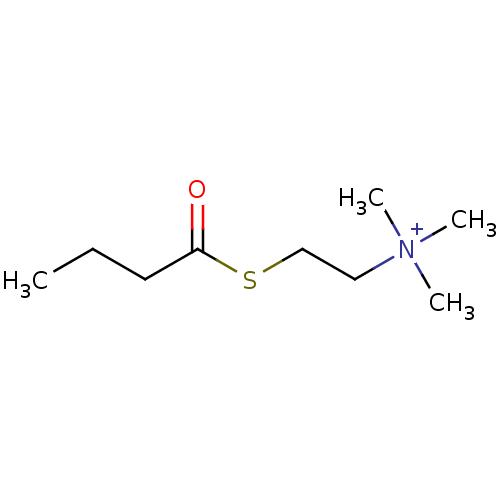
Substrate
Name:
BDBM8978
Synonyms:
(Propylcarbonylthioethyl)trimethylammonium iodide | CHEMBL139908 | CHEMBL148530 | [2-(butanoylsulfanyl)ethyl]trimethylazanium iodide | butyrylthiocholine | butyrylthiocholine chloride
Type:
Small organic molecule
Emp. Form.:
C9H20NOS
Mol. Mass.:
190.326
SMILES:
CCCC(=O)SCC[N+](C)(C)C