Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Prothrombin
Ligand
BDBM12751
Substrate
BDBM13573
Meas. Tech.
Enzyme Assay and Determination of the Inhibition Constants.
pH
7±n/a
Temperature
295.15±n/a K
Ki
300±n/a nM
Citation
 Lam, PY; Clark, CG; Li, R; Pinto, DJ; Orwat, MJ; Galemmo, RA; Fevig, JM; Teleha, CA; Alexander, RS; Smallwood, AM; Rossi, KA; Wright, MR; Bai, SA; He, K; Luettgen, JM; Wong, PC; Knabb, RM; Wexler, RR Structure-based design of novel guanidine/benzamidine mimics: potent and orally bioavailable factor Xa inhibitors as novel anticoagulants. J Med Chem 46:4405-18 (2003) [PubMed] Article
Lam, PY; Clark, CG; Li, R; Pinto, DJ; Orwat, MJ; Galemmo, RA; Fevig, JM; Teleha, CA; Alexander, RS; Smallwood, AM; Rossi, KA; Wright, MR; Bai, SA; He, K; Luettgen, JM; Wong, PC; Knabb, RM; Wexler, RR Structure-based design of novel guanidine/benzamidine mimics: potent and orally bioavailable factor Xa inhibitors as novel anticoagulants. J Med Chem 46:4405-18 (2003) [PubMed] Article More Info.:
Target
Name:
Prothrombin
Synonyms:
Activation peptide fragment 1 | Activation peptide fragment 2 | Coagulation factor II | F2 | Prothrombin precursor | THRB_HUMAN | Thrombin heavy chain | Thrombin light chain
Type:
Protein
Mol. Mass.:
70029.57
Organism:
Homo sapiens (Human)
Description:
P00734
Residue:
622
Sequence:
MAHVRGLQLPGCLALAALCSLVHSQHVFLAPQQARSLLQRVRRANTFLEEVRKGNLERECVEETCSYEEAFEALESSTATDVFWAKYTACETARTPRDKLAACLEGNCAEGLGTNYRGHVNITRSGIECQLWRSRYPHKPEINSTTHPGADLQENFCRNPDSSTTGPWCYTTDPTVRRQECSIPVCGQDQVTVAMTPRSEGSSVNLSPPLEQCVPDRGQQYQGRLAVTTHGLPCLAWASAQAKALSKHQDFNSAVQLVENFCRNPDGDEEGVWCYVAGKPGDFGYCDLNYCEEAVEEETGDGLDEDSDRAIEGRTATSEYQTFFNPRTFGSGEADCGLRPLFEKKSLEDKTERELLESYIDGRIVEGSDAEIGMSPWQVMLFRKSPQELLCGASLISDRWVLTAAHCLLYPPWDKNFTENDLLVRIGKHSRTRYERNIEKISMLEKIYIHPRYNWRENLDRDIALMKLKKPVAFSDYIHPVCLPDRETAASLLQAGYKGRVTGWGNLKETWTANVGKGQPSVLQVVNLPIVERPVCKDSTRIRITDNMFCAGYKPDEGKRGDACEGDSGGPFVMKSPFNNRWYQMGIVSWGEGCDRDGKYGFYTHVFRLKKWIQKVIDQFGE
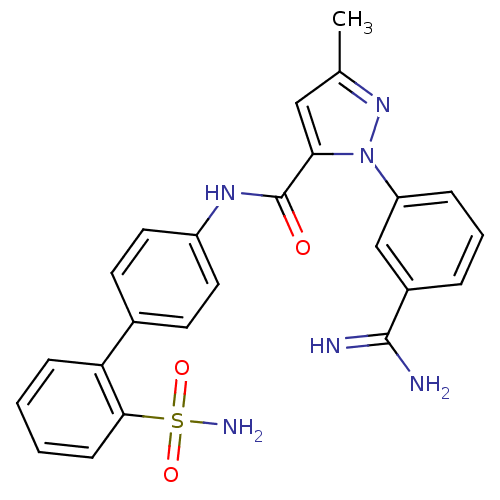
Inhibitor
Name:
BDBM12751
Synonyms:
1-(3-carbamimidoylphenyl)-3-methyl-N-[4-(2-sulfamoylphenyl)phenyl]-1H-pyrazole-5-carboxamide | CHEMBL152854 | CHEMBL37251 | SN-429
Type:
Small organic molecule
Emp. Form.:
C24H22N6O3S
Mol. Mass.:
474.535
SMILES:
Cc1cc(C(=O)Nc2ccc(cc2)-c2ccccc2S(N)(=O)=O)n(n1)-c1cccc(c1)C(N)=N
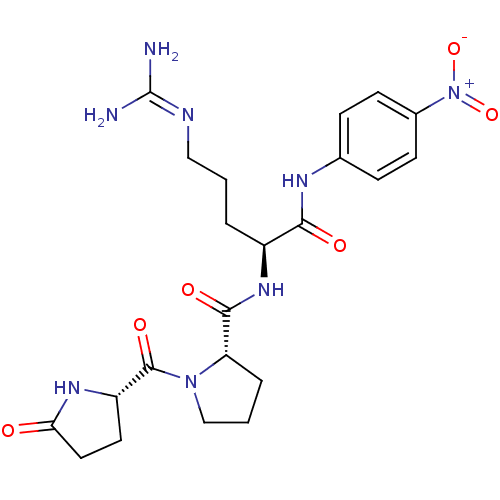
Substrate
Name:
BDBM13573
Synonyms:
(2S)-5-carbamimidamido-N-(4-nitrophenyl)-2-{[(2S)-1-{[(2S)-5-oxopyrrolidin-2-yl]carbonyl}pyrrolidin-2-yl]formamido}pentanamide hydrochloride | Chromogenic Substrate S-2366 | Glu-Pro-Arg-pNA | Hepsin Chromogenic Substrate | L-Pyroglutamyl-L-prolyl-L-arginine-p-Nitroaniline | L-Pyroglutamyl-L-prolyl-L-argininep-Nitroaniline | S-2366
Type:
Small organic molecule
Emp. Form.:
C22H30N8O6
Mol. Mass.:
502.5236
SMILES:
[#7]\[#6](-[#7])=[#7]/[#6]-[#6]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6]-[#7]-1-[#6](=O)-[#6@@H]-1-[#6]-[#6]-[#6](=O)-[#7]-1)-[#6](=O)-[#7]-c1ccc(cc1)-[#7+](-[#8-])=O |r|