Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Ribosyldihydronicotinamide dehydrogenase [quinone]
Ligand
BDBM29612
Substrate
BDBM29211
Meas. Tech.
QR2 Inhibition Assay
pH
8±n/a
Temperature
298.15±n/a K
IC50
9900±1800 nM
Citation
 Calamini, B; Santarsiero, BD; Boutin, JA; Mesecar, AD Kinetic, thermodynamic and X-ray structural insights into the interaction of melatonin and analogues with quinone reductase 2. Biochem J 413:81-91 (2008) [PubMed] Article
Calamini, B; Santarsiero, BD; Boutin, JA; Mesecar, AD Kinetic, thermodynamic and X-ray structural insights into the interaction of melatonin and analogues with quinone reductase 2. Biochem J 413:81-91 (2008) [PubMed] Article Target
Name:
Ribosyldihydronicotinamide dehydrogenase [quinone]
Synonyms:
Metallothionein-3 | NMOR2 | NQO2 | NQO2_HUMAN | NRH dehydrogenase [quinone] 2 | NRH:quinone oxidoreductase 2 | QR2 | Quinone reductase 2 | Quinone reductase 2 (NQO2) | Ribosyldihydronicotinamide dehydrogenase [quinone]
Type:
Protein
Mol. Mass.:
25917.25
Organism:
Homo sapiens (Human)
Description:
P16083
Residue:
231
Sequence:
MAGKKVLIVYAHQEPKSFNGSLKNVAVDELSRQGCTVTVSDLYAMNLEPRATDKDITGTLSNPEVFNYGVETHEAYKQRSLASDITDEQKKVREADLVIFQFPLYWFSVPAILKGWMDRVLCQGFAFDIPGFYDSGLLQGKLALLSVTTGGTAEMYTKTGVNGDSRYFLWPLQHGTLHFCGFKVLAPQISFAPEIASEEERKGMVAAWSQRLQTIWKEEPIPCTAHWHFGQ
Inhibitor
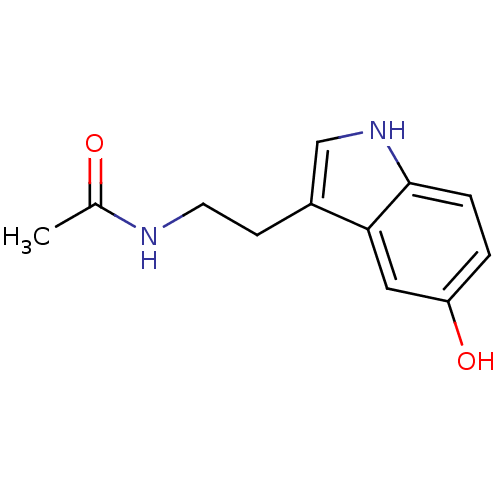
Name:
BDBM29612
Synonyms:
CHEMBL33103 | CVD-0001578 | JOH-MSK-a63bdd1d-4 | N-ACETYL SEROTONIN | N-Acetyl-5-hydroxytryptamine | N-Acetyltryptamine,5-Hydroxy | N-acetylserotonin | Normelatonin | Serotonin,N-acetyl
Type:
Small organic molecule
Emp. Form.:
C12H14N2O2
Mol. Mass.:
218.2518
SMILES:
CC(=O)NCCc1c[nH]c2ccc(O)cc12