Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
D(1B) dopamine receptor
Ligand
BDBM50001888
Substrate
n/a
Ki
133±n/a nM
Comments
PDSP_513
Citation
 Sunahara, RK; Guan, HC; O'Dowd, BF; Seeman, P; Laurier, LG; Ng, G; George, SR; Torchia, J; Van Tol, HH; Niznik, HB Cloning of the gene for a human dopamine D5 receptor with higher affinity for dopamine than D1. Nature 350:614-9 (1991) [PubMed] Article
Sunahara, RK; Guan, HC; O'Dowd, BF; Seeman, P; Laurier, LG; Ng, G; George, SR; Torchia, J; Van Tol, HH; Niznik, HB Cloning of the gene for a human dopamine D5 receptor with higher affinity for dopamine than D1. Nature 350:614-9 (1991) [PubMed] Article More Info.:
Target
Name:
D(1B) dopamine receptor
Synonyms:
D(5) dopamine receptor | D1beta dopamine receptor | DOPAMINE D5 | DRD1B | DRD1L2 | DRD5 | DRD5_HUMAN | dopamine receptor D5
Type:
Protein
Mol. Mass.:
52943.41
Organism:
Homo sapiens (Human)
Description:
P21918
Residue:
477
Sequence:
MLPPGSNGTAYPGQFALYQQLAQGNAVGGSAGAPPLGPSQVVTACLLTLLIIWTLLGNVLVCAAIVRSRHLRANMTNVFIVSLAVSDLFVALLVMPWKAVAEVAGYWPFGAFCDVWVAFDIMCSTASILNLCVISVDRYWAISRPFRYKRKMTQRMALVMVGLAWTLSILISFIPVQLNWHRDQAASWGGLDLPNNLANWTPWEEDFWEPDVNAENCDSSLNRTYAISSSLISFYIPVAIMIVTYTRIYRIAQVQIRRISSLERAAEHAQSCRSSAACAPDTSLRASIKKETKVLKTLSVIMGVFVCCWLPFFILNCMVPFCSGHPEGPPAGFPCVSETTFDVFVWFGWANSSLNPVIYAFNADFQKVFAQLLGCSHFCSRTPVETVNISNELISYNQDIVFHKEIAAAYIHMMPNAVTPGNREVDNDEEEGPFDRMFQIYQTSPDGDPVAESVWELDCEGEISLDKITPFTPNGFH
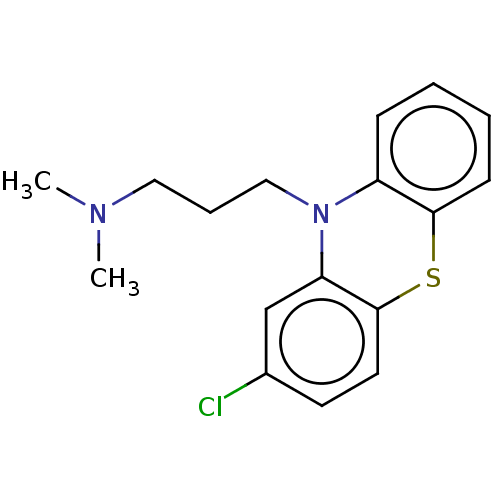
Inhibitor
Name:
BDBM50001888
Synonyms:
(chloropromazine) [3-(2-Chloro-phenothiazin-10-yl)-propyl]-dimethyl-amine | (chlorpromazine)[3-(2-Chloro-phenothiazin-10-yl)-propyl]-dimethyl-amine | 1-(2-Allyl-phenoxy)-3-isopropylamino-propan-2-ol | 1N,1N-dimethyl-3-(2-chloro-10H-10-phenothiazinyl)-1-propanamine | 3-(2-chloro-10H-phenothiazin-10-yl)-N,N-dimethylpropan-1-amine | CHEMBL71 | CHLORPROMAZINE | CHLORPROMAZINE HIBENZATE | CHLORPROMAZINE HYDROCHLORIDE | CHLORPROMAZINE PHENOLPHTHALINATE | CHLORPROMAZINE TANNATE | Chlorpromazine;[3-(2-Chloro-phenothiazin-10-yl)-propyl]-dimethyl-amine | PROMAPAR | SONAZINE | THORAZINE | [3-(2-Chloro-phenothiazin-10-yl)-propyl]-dimethyl-amine (chlor-promazine) | [3-(2-Chloro-phenothiazin-10-yl)-propyl]-dimethyl-amine( Chlorpromazine) | [3-(2-Chloro-phenothiazin-10-yl)-propyl]-dimethyl-amine(clorpromazine) | chloropromazine | med.21724, Compound 15
Type:
Small organic molecule
Emp. Form.:
C17H19ClN2S
Mol. Mass.:
318.864
SMILES:
CN(C)CCCN1c2ccccc2Sc2ccc(Cl)cc12