Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Glutamate receptor ionotropic, NMDA 1
Ligand
BDBM50038168
Substrate
n/a
Meas. Tech.
ChEMBL_141012 (CHEMBL747502)
Ki
1.000000±n/a nM
Citation
 Di Fabio, R; Capelli, AM; Conti, N; Cugola, A; Donati, D; Feriani, A; Gastaldi, P; Gaviraghi, G; Hewkin, CT; Micheli, F; Missio, A; Mugnaini, M; Pecunioso, A; Quaglia, AM; Ratti, E; Rossi, L; Tedesco, G; Trist, DG; Reggiani, A Substituted indole-2-carboxylates as in vivo potent antagonists acting as the strychnine-insensitive glycine binding site. J Med Chem 40:841-50 (1997) [PubMed] Article
Di Fabio, R; Capelli, AM; Conti, N; Cugola, A; Donati, D; Feriani, A; Gastaldi, P; Gaviraghi, G; Hewkin, CT; Micheli, F; Missio, A; Mugnaini, M; Pecunioso, A; Quaglia, AM; Ratti, E; Rossi, L; Tedesco, G; Trist, DG; Reggiani, A Substituted indole-2-carboxylates as in vivo potent antagonists acting as the strychnine-insensitive glycine binding site. J Med Chem 40:841-50 (1997) [PubMed] Article More Info.:
Target
Name:
Glutamate receptor ionotropic, NMDA 1
Synonyms:
Glutamate (NMDA) receptor subunit zeta 1 | Glutamate [NMDA] receptor subunit zeta-1 | Glutamate-NMDA-Channel | Glutamate-NMDA-MK801 | Glutamate-NMDA-Polyamine | Grin1 | NMDA | NMDZ1_RAT | Nmdar1 | phencyclidine
Type:
Enzyme Catalytic Domain
Mol. Mass.:
105533.40
Organism:
RAT
Description:
P35439
Residue:
938
Sequence:
MSTMHLLTFALLFSCSFARAACDPKIVNIGAVLSTRKHEQMFREAVNQANKRHGSWKIQLNATSVTHKPNAIQMALSVCEDLISSQVYAILVSHPPTPNDHFTPTPVSYTAGFYRIPVLGLTTRMSIYSDKSIHLSFLRTVPPYSHQSSVWFEMMRVYNWNHIILLVSDDHEGRAAQKRLETLLEERESKAEKVLQFDPGTKNVTALLMEARELEARVIILSASEDDAATVYRAAAMLNMTGSGYVWLVGEREISGNALRYAPDGIIGLQLINGKNESAHISDAVGVVAQAVHELLEKENITDPPRGCVGNTNIWKTGPLFKRVLMSSKYADGVTGRVEFNEDGDRKFANYSIMNLQNRKLVQVGIYNGTHVIPNDRKIIWPGGETEKPRGYQMSTRLKIVTIHQEPFVYVKPTMSDGTCKEEFTVNGDPVKKVICTGPNDTSPGSPRHTVPQCCYGFCIDLLIKLARTMNFTYEVHLVADGKFGTQERVNNSNKKEWNGMMGELLSGQADMIVAPLTINNERAQYIEFSKPFKYQGLTILVKKEIPRSTLDSFMQPFQSTLWLLVGLSVHVVAVMLYLLDRFSPFGRFKVNSEEEEEDALTLSSAMWFSWGVLLNSGIGEGAPRSFSARILGMVWAGFAMIIVASYTANLAAFLVLDRPEERITGINDPRLRNPSDKFIYATVKQSSVDIYFRRQVELSTMYRHMEKHNYESAAEAIQAVRDNKLHAFIWDSAVLEFEASQKCDLVTTGELFFRSGFGIGMRKDSPWKQNVSLSILKSHENGFMEDLDKTWVRYQECDSRSNAPATLTFENMAGVFMLVAGGIVAGIFLIFIEIAYKRHKDARRKQMQLAFAAVNVWRKNLQDRKSGRAEPDPKKKATFRAITSTLASSFKRRRSSKDTSTGGGRGALQNQKDTVLPRRAIEREEGQLQLCSRHRES
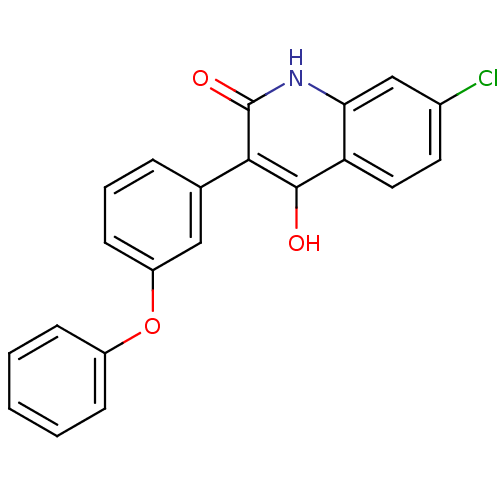
Inhibitor
Name:
BDBM50038168
Synonyms:
7-Chloro-4-hydroxy-3-(3-phenoxy-phenyl)-1H-quinolin-2-one | 7-chloro-4-hydroxy-3-(3-phenoxyphenyl)quinolin-2(1H)-one | CHEMBL31741 | L-701324
Type:
Small organic molecule
Emp. Form.:
C21H14ClNO3
Mol. Mass.:
363.794
SMILES:
Oc1c(-c2cccc(Oc3ccccc3)c2)c(=O)[nH]c2cc(Cl)ccc12