Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cytochrome P450 3A4
Ligand
BDBM8610
Substrate
n/a
Meas. Tech.
ChEMBL_1824514 (CHEMBL4324278)
IC50
17±n/a nM
Citation
 Ramdas, V; Talwar, R; Banerjee, M; Joshi, AA; Das, AK; Walke, DS; Borhade, P; Dhayagude, U; Loriya, R; Gote, G; Bommakanti, A; Sivaram, A; Agarwal, G; Goswami, A; Nigade, P; Mehta, M; Patil, V; Modi, D; Kumar, H; Mallurwar, S; Dash, A; Modi, F; Kuldharan, S; Srivastava, P; Singh, M; Narasimham, L; Gundu, J; Sharma, S; Kamboj, RK; Palle, VP Discovery and Characterization of Potent Pan-Genotypic HCV NS5A Inhibitors Containing Novel Tricyclic Central Core Leading to Clinical Candidate. J Med Chem 62:10563-10582 (2019) [PubMed] Article
Ramdas, V; Talwar, R; Banerjee, M; Joshi, AA; Das, AK; Walke, DS; Borhade, P; Dhayagude, U; Loriya, R; Gote, G; Bommakanti, A; Sivaram, A; Agarwal, G; Goswami, A; Nigade, P; Mehta, M; Patil, V; Modi, D; Kumar, H; Mallurwar, S; Dash, A; Modi, F; Kuldharan, S; Srivastava, P; Singh, M; Narasimham, L; Gundu, J; Sharma, S; Kamboj, RK; Palle, VP Discovery and Characterization of Potent Pan-Genotypic HCV NS5A Inhibitors Containing Novel Tricyclic Central Core Leading to Clinical Candidate. J Med Chem 62:10563-10582 (2019) [PubMed] Article More Info.:
Target
Name:
Cytochrome P450 3A4
Synonyms:
Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase
Type:
Enzyme
Mol. Mass.:
57349.57
Organism:
Homo sapiens (Human)
Description:
n/a
Residue:
503
Sequence:
MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMFDMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISIAEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYSMDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICVFPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSIIFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVVNETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFSKKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLGGLLQPEKPVVLKVESRDGTVSGA
Inhibitor
Name:
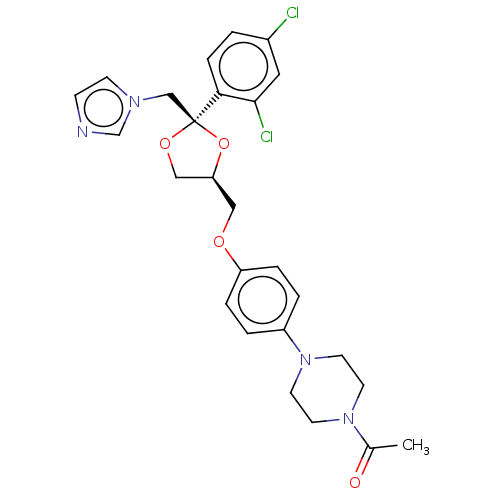
BDBM8610
Synonyms:
1-[4-(4-{[(2R,4S)-2-(2,4-dichlorophenyl)-2-(1H-imidazol-1-ylmethyl)-1,3-dioxolan-4-yl]methoxy}phenyl)piperazin-1-yl]ethan-1-one | 24F2-1,25(OH)D3 | CHEMBL75 | KTZ | Ketoconazole | Ketoconazole (k) | US9138393, Ketoconozole | US9144538, Ketoconozole
Type:
Small organic molecule
Emp. Form.:
C26H28Cl2N4O4
Mol. Mass.:
531.431
SMILES:
[H][C@]1(COc2ccc(cc2)N2CCN(CC2)C(C)=O)CO[C@@](Cn2ccnc2)(O1)c1ccc(Cl)cc1Cl |r|