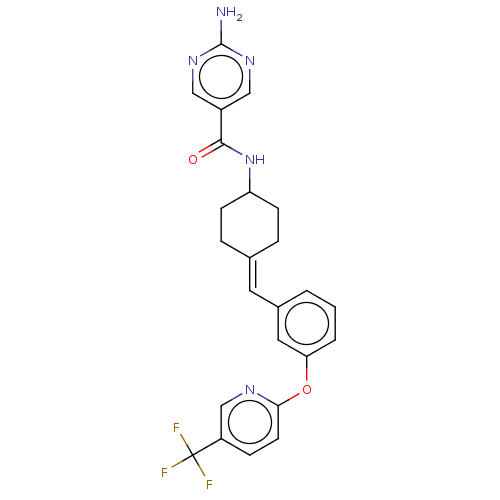
Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Fatty-acid amide hydrolase 1
Ligand
BDBM143373
Substrate
n/a
Meas. Tech.
ChEMBL_1870780 (CHEMBL4371947)
IC50
2.0±n/a nM
Citation
 Bhuniya, D; Kharul, RK; Hajare, A; Shaikh, N; Bhosale, S; Balwe, S; Begum, F; De, S; Athavankar, S; Joshi, D; Madgula, V; Joshi, K; Raje, AA; Meru, AV; Magdum, A; Mookhtiar, KA; Barbhaiya, R Discovery and evaluation of novel FAAH inhibitors in neuropathic pain model. Bioorg Med Chem Lett 29:238-243 (2019) [PubMed] Article
Bhuniya, D; Kharul, RK; Hajare, A; Shaikh, N; Bhosale, S; Balwe, S; Begum, F; De, S; Athavankar, S; Joshi, D; Madgula, V; Joshi, K; Raje, AA; Meru, AV; Magdum, A; Mookhtiar, KA; Barbhaiya, R Discovery and evaluation of novel FAAH inhibitors in neuropathic pain model. Bioorg Med Chem Lett 29:238-243 (2019) [PubMed] Article More Info.:
Target
Name:
Fatty-acid amide hydrolase 1
Synonyms:
Anandamide amidohydrolase | Anandamide amidohydrolase 1 | FAAH | FAAH1 | FAAH1_HUMAN | Fatty Acid Amide Hydrolase (FAAH) | Fatty-acid amide hydrolase (FAAH) | Fatty-acid amide hydrolase 1 | Oleamide hydrolase 1
Type:
Protein
Mol. Mass.:
63071.19
Organism:
Homo sapiens (Human)
Description:
O00519
Residue:
579
Sequence:
MVQYELWAALPGASGVALACCFVAAAVALRWSGRRTARGAVVRARQRQRAGLENMDRAAQRFRLQNPDLDSEALLALPLPQLVQKLHSRELAPEAVLFTYVGKAWEVNKGTNCVTSYLADCETQLSQAPRQGLLYGVPVSLKECFTYKGQDSTLGLSLNEGVPAECDSVVVHVLKLQGAVPFVHTNVPQSMFSYDCSNPLFGQTVNPWKSSKSPGGSSGGEGALIGSGGSPLGLGTDIGGSIRFPSSFCGICGLKPTGNRLSKSGLKGCVYGQEAVRLSVGPMARDVESLALCLRALLCEDMFRLDPTVPPLPFREEVYTSSQPLRVGYYETDNYTMPSPAMRRAVLETKQSLEAAGHTLVPFLPSNIPHALETLSTGGLFSDGGHTFLQNFKGDFVDPCLGDLVSILKLPQWLKGLLAFLVKPLLPRLSAFLSNMKSRSAGKLWELQHEIEVYRKTVIAQWRALDLDVVLTPMLAPALDLNAPGRATGAVSYTMLYNCLDFPAGVVPVTTVTAEDEAQMEHYRGYFGDIWDKMLQKGMKKSVGLPVAVQCVALPWQEELCLRFMREVERLMTPEKQSS
Inhibitor
Name:
BDBM143373
Synonyms:
US9682953, 20.A-10 | US9682953, 20.A-9
Type:
Small organic molecule
Emp. Form.:
C24H22F3N5O2
Mol. Mass.:
469.459
SMILES:
Nc1ncc(cn1)C(=O)NC1CCC(CC1)=Cc1cccc(Oc2ccc(cn2)C(F)(F)F)c1 |(11.34,2.69,;10,1.93,;10,.38,;8.67,-.38,;7.34,.38,;7.34,1.93,;8.67,2.69,;6,-.38,;6,-1.93,;4.67,.38,;3.33,-.38,;3.33,-1.93,;2,-2.69,;.67,-1.93,;.67,-.38,;2,.38,;-.67,-2.69,;-2,-1.93,;-2,-.38,;-3.33,.38,;-4.67,-.38,;-4.67,-1.93,;-6,-2.69,;-7.34,-1.93,;-7.34,-.38,;-8.67,.38,;-10,-.38,;-10,-1.93,;-8.67,-2.69,;-11.34,.38,;-12.67,-.39,;-11.34,1.93,;-11.34,-1.16,;-3.33,-2.69,)|