Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Adenosine deaminase
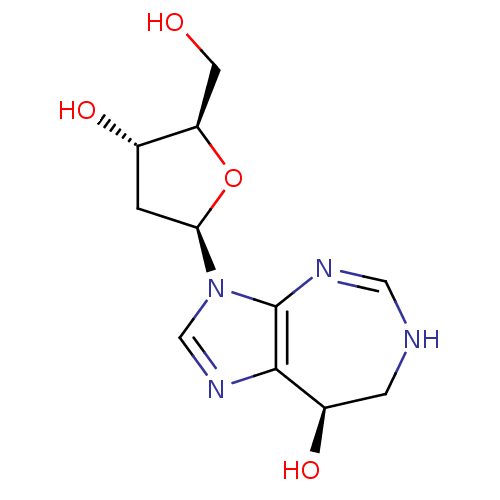
Ligand
BDBM22925
Substrate
n/a
Meas. Tech.
ChEMBL_1875740 (CHEMBL4377134)
Ki
0.038000±n/a nM
Citation
 Cheviet, T; Lefebvre-Tournier, I; Wein, S; Peyrottes, S Plasmodium Purine Metabolism and Its Inhibition by Nucleoside and Nucleotide Analogues. J Med Chem 62:8365-8391 (2019) [PubMed] Article
Cheviet, T; Lefebvre-Tournier, I; Wein, S; Peyrottes, S Plasmodium Purine Metabolism and Its Inhibition by Nucleoside and Nucleotide Analogues. J Med Chem 62:8365-8391 (2019) [PubMed] Article More Info.:
Target
Name:
Adenosine deaminase
Synonyms:
3.5.4.4 | Adenosine deaminase
Type:
PROTEIN
Mol. Mass.:
42457.37
Organism:
Plasmodium falciparum
Description:
ChEMBL_119120
Residue:
367
Sequence:
MNCKNMDTSYEIINYLTKDELDIDLSCMDKKERYKIWKRLPKCELHCHLDVCFSVDFFLNVIRKYNIQPNMSDEEIIDYYLFSKPGKSLDEFVEKALRLTDIYIDYTVVEDLAKHAVFNKYKEGVVLMEFRYSPSFMSFKHNLDKDLIHEAIVKGLNEAVALLEYKIQVGLLCTGDGGLSHERMKEAAEFCIKHKKDFVGYDHAGHEVDLKPFKDIFDNIREEGISLSVHAGEDVSIPNLNSLYTAINLLHVKRIGHGIRVSESQELIDLVKEKDILLEVCPISNVLLNNVKSMDTHPIRMLYDAGVKVSVNSDDPGMFLTNITDNYEELYTHLNFTLADFMKMNLWAVQKSFVDPDIKNKIISKYF
Inhibitor
Name:
BDBM22925
Synonyms:
(8R)-3-[(2R,4S,5R)-4-hydroxy-5-(hydroxymethyl)oxolan-2-yl]-3H,6H,7H,8H-imidazo[4,5-d][1,3]diazepin-8-ol | Covidarabine | Deoxycoformycin | Nipent | Pentostatin
Type:
Nucleoside or nucleotide
Emp. Form.:
C11H16N4O4
Mol. Mass.:
268.2691
SMILES:
OC[C@H]1O[C@H](C[C@@H]1O)n1cnc2[C@H](O)CNC=Nc12 |c:17|