Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Prostaglandin G/H synthase 1
Ligand
BDBM50044068
Substrate
n/a
Meas. Tech.
ChEMBL_159275 (CHEMBL764259)
IC50
27±n/a nM
Citation
 Song, Y; Connor, DT; Doubleday, R; Sorenson, RJ; Sercel, AD; Unangst, PC; Roth, BD; Gilbertsen, RB; Chan, K; Schrier, DJ; Guglietta, A; Bornemeier, DA; Dyer, RD Synthesis, structure-activity relationships, and in vivo evaluations of substituted di-tert-butylphenols as a novel class of potent, selective, and orally active cyclooxygenase-2 inhibitors. 1. Thiazolone and oxazolone series. J Med Chem 42:1151-60 (1999) [PubMed] Article
Song, Y; Connor, DT; Doubleday, R; Sorenson, RJ; Sercel, AD; Unangst, PC; Roth, BD; Gilbertsen, RB; Chan, K; Schrier, DJ; Guglietta, A; Bornemeier, DA; Dyer, RD Synthesis, structure-activity relationships, and in vivo evaluations of substituted di-tert-butylphenols as a novel class of potent, selective, and orally active cyclooxygenase-2 inhibitors. 1. Thiazolone and oxazolone series. J Med Chem 42:1151-60 (1999) [PubMed] Article More Info.:
Target
Name:
Prostaglandin G/H synthase 1
Synonyms:
COX-1 | COX1 | Cyclooxygenase | Cyclooxygenase-1 | Cyclooxygenase-1 (COX-1) | PGH1_HUMAN | PTGS1 | Prostaglandin G/H synthase (cyclooxygenase)
Type:
Enzyme
Mol. Mass.:
68692.62
Organism:
Homo sapiens (Human)
Description:
P23219
Residue:
599
Sequence:
MSRSLLLWFLLFLLLLPPLPVLLADPGAPTPVNPCCYYPCQHQGICVRFGLDRYQCDCTRTGYSGPNCTIPGLWTWLRNSLRPSPSFTHFLLTHGRWFWEFVNATFIREMLMRLVLTVRSNLIPSPPTYNSAHDYISWESFSNVSYYTRILPSVPKDCPTPMGTKGKKQLPDAQLLARRFLLRRKFIPDPQGTNLMFAFFAQHFTHQFFKTSGKMGPGFTKALGHGVDLGHIYGDNLERQYQLRLFKDGKLKYQVLDGEMYPPSVEEAPVLMHYPRGIPPQSQMAVGQEVFGLLPGLMLYATLWLREHNRVCDLLKAEHPTWGDEQLFQTTRLILIGETIKIVIEEYVQQLSGYFLQLKFDPELLFGVQFQYRNRIAMEFNHLYHWHPLMPDSFKVGSQEYSYEQFLFNTSMLVDYGVEALVDAFSRQIAGRIGGGRNMDHHILHVAVDVIRESREMRLQPFNEYRKRFGMKPYTSFQELVGEKEMAAELEELYGDIDALEFYPGLLLEKCHPNSIFGESMIEIGAPFSLKGLLGNPICSPEYWKPSTFGGEVGFNIVKTATLKKLVCLNTKTCPYVSFRVPDASQDDGPAVERPSTEL
Inhibitor
Name:
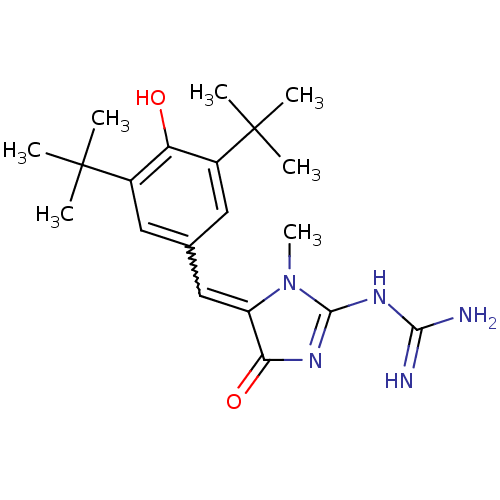
BDBM50044068
Synonyms:
CHEMBL15904 | N-[5-(3,5-Di-tert-butyl-4-hydroxy-benzylidene)-1-methyl-4-oxo-4,5-dihydro-1H-imidazol-2-yl]-guanidine | N-{5-[1-(3,5-Di-tert-butyl-4-hydroxy-phenyl)-meth-(Z)-ylidene]-1-methyl-4-oxo-4,5-dihydro-1H-imidazol-2-yl}-guanidine
Type:
Small organic molecule
Emp. Form.:
C20H29N5O2
Mol. Mass.:
371.4766
SMILES:
CN1C(NC(N)=N)=NC(=O)C1=Cc1cc(c(O)c(c1)C(C)(C)C)C(C)(C)C |w:11.12,c:6|