Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
DNA gyrase subunit A
Ligand
BDBM21690
Substrate
n/a
Meas. Tech.
ChEMBL_1897347 (CHEMBL4399382)
IC50
280±n/a nM
Citation
 Lim, CSQ; Ha, KP; Clarke, RS; Gavin, LA; Cook, DT; Hutton, JA; Sutherell, CL; Edwards, AM; Evans, LE; Tate, EW; Lanyon-Hogg, T Identification of a potent small-molecule inhibitor of bacterial DNA repair that potentiates quinolone antibiotic activity in methicillin-resistant Staphylococcus aureus. Bioorg Med Chem 27:0 (2019) [PubMed] Article
Lim, CSQ; Ha, KP; Clarke, RS; Gavin, LA; Cook, DT; Hutton, JA; Sutherell, CL; Edwards, AM; Evans, LE; Tate, EW; Lanyon-Hogg, T Identification of a potent small-molecule inhibitor of bacterial DNA repair that potentiates quinolone antibiotic activity in methicillin-resistant Staphylococcus aureus. Bioorg Med Chem 27:0 (2019) [PubMed] Article More Info.:
Target
Name:
DNA gyrase subunit A
Synonyms:
5.6.2.2 | DNA gyrase subunit A | gyrA
Type:
PROTEIN
Mol. Mass.:
97006.20
Organism:
Escherichia coli
Description:
ChEMBL_117105
Residue:
875
Sequence:
MSDLAREITPVNIEEELKSSYLDYAMSVIVGRALPDVRDGLKPVHRRVLYAMNVLGNDWNKAYKKSARVVGDVIGKYHPHGDSAVYDTIVRMAQPFSLRYMLVDGQGNFGSIDGDSAAAMRYTEIRLAKIAHELMADLEKETVDFVDNYDGTEKIPDVMPTKIPNLLVNGSSGIAVGMATNIPPHNLTEVINGCLAYIDDEDISIEGLMEHIPEPDFPTAAIINGRRGIEEAYRTGRGKVYIRARAEVEVDAKTGRETIIVHEIPYQVNKARLIEKIAELVKEKRVEGISALRDESDKDGMRIVIEVKRDAVGEVVLNNLYSQTQLQVSFGINMVALHHGQPKIMNLKDIIAAFVRHRREVVTRRTIFELRKARDRAHILEALAVALANIDPIIELIRHAPTPAEAKTALVANPWQLGNVAAMLERAGDDAARPEWLEPEFGVRDGLYYLTEQQAQAILDLRLQKLTGLEHEKLLDEYKELLDQIAELLRILGSADRLMEVIREELELVREQFGDKRRTEITANSADINLEDLITQEDVVVTLSHQGYVKYQPLSEYEAQRRGGKGKSAARIKEEDFIDRLLVANTHDHILCFSSRGRVYSMKVYQLPEATRGARGRPIVNLLPLEQDERITAILPVTEFEEGVKVFMATANGTVKKTVLTEFNRLRTAGKVAIKLVDGDELIGVDLTSGEDEVMLFSAEGKVVRFKESSVRAMGCNTTGVRGIRLGEGDKVVSLIVPRGDGAILTATQNGYGKRTAVAEYPTKSRATKGVISIKVTERNGLVVGAVQVDDCDQIMMITDAGTLVRTRVSEISIVGRNTQGVILIRTAEDENVVGLQRVAEPVDEEDLDTIDGSAAEGDDEIAPEVDVDDEPEEE
Inhibitor
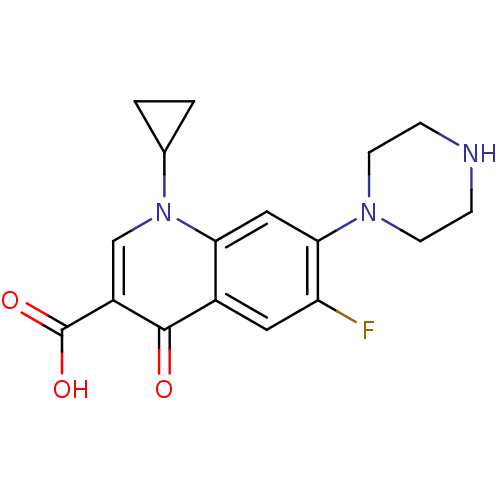
Name:
BDBM21690
Synonyms:
1-cyclopropyl-6-fluoro-4-oxo-7-(piperazin-1-yl)-1,4-dihydroquinoline-3-carboxylic acid | 1-cyclopropyl-6-fluoro-4-oxo-7-piperazin-1-yl-1,4-dihydroquinoline-3-carboxylic acid | Bay 09867 | CHEMBL8 | Ciprinol | Cipro | Ciprofloxacin | US11590142, Compound Ciprofloxacin | US9138393, Ciprofloxacin HCl | US9144538, Ciprofloxacin HCl
Type:
Small organic molecule
Emp. Form.:
C17H18FN3O3
Mol. Mass.:
331.3415
SMILES:
OC(=O)c1cn(C2CC2)c2cc(N3CCNCC3)c(F)cc2c1=O