Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Protein mono-ADP-ribosyltransferase PARP4
Ligand
BDBM50250875
Substrate
n/a
Meas. Tech.
ChEMBL_1985253 (CHEMBL4618659)
IC50
>10000±n/a nM
Citation
 Waaler, J; Leenders, RGG; Sowa, ST; Alam Brinch, S; Lycke, M; Nieczypor, P; Aertssen, S; Murthy, S; Galera-Prat, A; Damen, E; Wegert, A; Nazar�, M; Lehti�, L; Krauss, S Preclinical Lead Optimization of a 1,2,4-Triazole Based Tankyrase Inhibitor. J Med Chem 63:6834-6846 (2020) [PubMed] Article
Waaler, J; Leenders, RGG; Sowa, ST; Alam Brinch, S; Lycke, M; Nieczypor, P; Aertssen, S; Murthy, S; Galera-Prat, A; Damen, E; Wegert, A; Nazar�, M; Lehti�, L; Krauss, S Preclinical Lead Optimization of a 1,2,4-Triazole Based Tankyrase Inhibitor. J Med Chem 63:6834-6846 (2020) [PubMed] Article More Info.:
Target
Name:
Protein mono-ADP-ribosyltransferase PARP4
Synonyms:
(ARTD4 or PARP4) | 193 kDa vault protein | 2.4.2.- | 2.4.2.30 | ADP-ribosyltransferase diphtheria toxin-like 4 | ADPRTL1 | ADPRTL1 | ARTD4 | KIAA0177 | KIAA0177 GN | PARP-4 | PARP-related/IalphaI-related H5/proline-rich | PARP4 | PARP4_HUMAN | PARPL | PH5P | Poly [ADP-ribose] polymerase 4 | Synonyms=ADPRTL1 | VPARP | Vault poly(ADP-ribose) polymerase
Type:
n/a
Mol. Mass.:
192563.23
Organism:
Homo sapiens (Human)
Description:
Q9UKK3
Residue:
1724
Sequence:
MVMGIFANCIFCLKVKYLPQQQKKKLQTDIKENGGKFSFSLNPQCTHIILDNADVLSQYQLNSIQKNHVHIANPDFIWKSIREKRLLDVKNYDPYKPLDITPPPDQKASSSEVKTEGLCPDSATEEEDTVELTEFGMQNVEIPHLPQDFEVAKYNTLEKVGMEGGQEAVVVELQCSRDSRDCPFLISSHFLLDDGMETRRQFAIKKTSEDASEYFENYIEELKKQGFLLREHFTPEATQLASEQLQALLLEEVMNSSTLSQEVSDLVEMIWAEALGHLEHMLLKPVNRISLNDVSKAEGILLLVKAALKNGETAEQLQKMMTEFYRLIPHKGTMPKEVNLGLLAKKADLCQLIRDMVNVCETNLSKPNPPSLAKYRALRCKIEHVEQNTEEFLRVRKEVLQNHHSKSPVDVLQIFRVGRVNETTEFLSKLGNVRPLLHGSPVQNIVGILCRGLLLPKVVEDRGVQRTDVGNLGSGIYFSDSLSTSIKYSHPGETDGTRLLLICDVALGKCMDLHEKDFSLTEAPPGYDSVHGVSQTASVTTDFEDDEFVVYKTNQVKMKYIIKFSMPGDQIKDFHPSDHTELEEYRPEFSNFSKVEDYQLPDAKTSSSTKAGLQDASGNLVPLEDVHIKGRIIDTVAQVIVFQTYTNKSHVPIEAKYIFPLDDKAAVCGFEAFINGKHIVGEIKEKEEAQQEYLEAVTQGHGAYLMSQDAPDVFTVSVGNLPPKAKVLIKITYITELSILGTVGVFFMPATVAPWQQDKALNENLQDTVEKICIKEIGTKQSFSLTMSIEMPYVIEFIFSDTHELKQKRTDCKAVISTMEGSSLDSSGFSLHIGLSAAYLPRMWVEKHPEKESEACMLVFQPDLDVDLPDLASESEVIICLDCSSSMEGVTFLQAKQIALHALSLVGEKQKVNIIQFGTGYKELFSYPKHITSNTMAAEFIMSATPTMGNTDFWKTLRYLSLLYPARGSRNILLVSDGHLQDESLTLQLVKRSRPHTRLFACGIGSTANRHVLRILSQCGAGVFEYFNAKSKHSWRKQIEDQMTRLCSPSCHSVSVKWQQLNPDVPEALQAPAQVPSLFLNDRLLVYGFIPHCTQATLCALIQEKEFRTMVSTTELQKTTGTMIHKLAARALIRDYEDGILHENETSHEMKKQTLKSLIIKLSKENSLITQFTSFVAVEKRDENESPFPDIPKVSELIAKEDVDFLPYMSWQGEPQEAVRNQSLLASSEWPELRLSKRKHRKIPFSKRKMELSQPEVSEDFEEDGLGVLPAFTSNLERGGVEKLLDLSWTESCKPTATEPLFKKVSPWETSTSSFFPILAPAVGSYLPPTARAHSPASLSFASYRQVASFGSAAPPRQFDASQFSQGPVPGTCADWIPQSASCPTGPPQNPPSSPYCGIVFSGSSLSSAQSAPLQHPGGFTTRPSAGTFPELDSPQLHFSLPTDPDPIRGFGSYHPSASSPFHFQPSAASLTANLRLPMASALPEALCSQSRTTPVDLCLLEESVGSLEGSRCPVFAFQSSDTESDELSEVLQDSCFLQIKCDTKDDSILCFLEVKEEDEIVCIQHWQDAVPWTELLSLQTEDGFWKLTPELGLILNLNTNGLHSFLKQKGIQSLGVKGRECLLDLIATMLVLQFIRTRLEKEGIVFKSLMKMDDASISRNIPWAFEAIKQASEWVRRTEGQYPSICPRLELGNDWDSATKQLLGLQPISTVSPLHRVLHYSQG
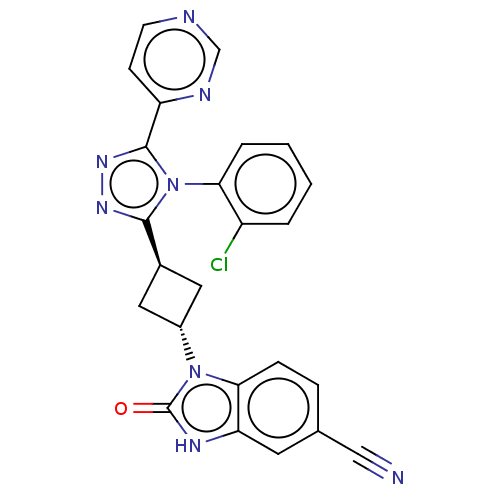
Inhibitor
Name:
BDBM50250875
Synonyms:
CHEMBL4095003
Type:
Small organic molecule
Emp. Form.:
C24H17ClN8O
Mol. Mass.:
468.898
SMILES:
Clc1ccccc1-n1c(nnc1-c1ccncn1)[C@H]1C[C@@H](C1)n1c2ccc(cc2[nH]c1=O)C#N |r,wU:20.25,wD:18.20,(25.39,-29.03,;24.05,-29.8,;24.05,-31.34,;22.72,-32.11,;21.39,-31.34,;21.39,-29.8,;22.72,-29.03,;22.72,-27.49,;23.97,-26.59,;23.49,-25.13,;21.95,-25.13,;21.48,-26.59,;20.01,-27.07,;19.69,-28.57,;18.22,-29.05,;17.08,-28.02,;17.4,-26.52,;18.87,-26.04,;25.43,-27.07,;26.13,-28.44,;27.5,-27.74,;26.8,-26.37,;28.97,-28.22,;29.44,-29.68,;28.67,-31.01,;29.44,-32.35,;30.98,-32.35,;31.75,-31.01,;30.98,-29.68,;31.46,-28.22,;30.21,-27.31,;30.21,-25.77,;31.75,-33.68,;32.52,-35.01,)|