Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cytochrome P450 3A4
Ligand
BDBM50072064
Substrate
n/a
Meas. Tech.
ChEBML_44699
IC50
>50000±n/a nM
Citation
 Chauret, N; Yergey, JA; Brideau, C; Friesen, RW; Mancini, J; Riendeau, D; Silva, J; Styhler, A; Trimble, LA; Nicoll-Griffith, DA In vitro metabolism considerations, including activity testing of metabolites, in the discovery and selection of the COX-2 inhibitor etoricoxib (MK-0663). Bioorg Med Chem Lett 11:1059-62 (2001) [PubMed] Article
Chauret, N; Yergey, JA; Brideau, C; Friesen, RW; Mancini, J; Riendeau, D; Silva, J; Styhler, A; Trimble, LA; Nicoll-Griffith, DA In vitro metabolism considerations, including activity testing of metabolites, in the discovery and selection of the COX-2 inhibitor etoricoxib (MK-0663). Bioorg Med Chem Lett 11:1059-62 (2001) [PubMed] Article More Info.:
Target
Name:
Cytochrome P450 3A4
Synonyms:
Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase
Type:
Enzyme
Mol. Mass.:
57349.57
Organism:
Homo sapiens (Human)
Description:
n/a
Residue:
503
Sequence:
MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMFDMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISIAEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYSMDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICVFPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSIIFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVVNETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFSKKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLGGLLQPEKPVVLKVESRDGTVSGA
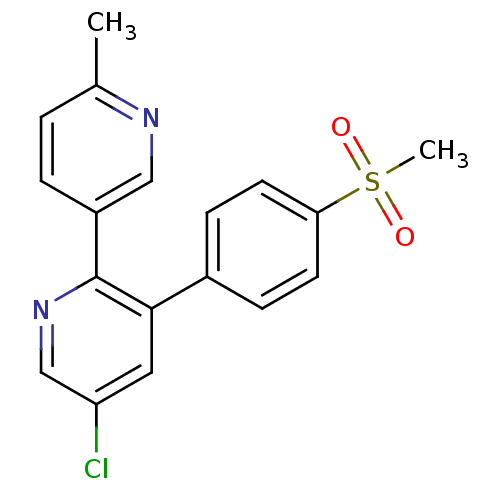
Inhibitor
Name:
BDBM50072064
Synonyms:
5-Chloro-3-(4-methanesulfonyl-phenyl)-6''-methyl-[2,3'']bipyridinyl | 5-Chloro-3-(4-methanesulfonyl-phenyl)-6'-methyl-[2,3']bipyridinyl | 5-chloro-2-(6-methylpyridin-3-yl)-3-(4-(methylsulfonyl)phenyl)pyridine | 5-chloro-6'-methyl-3-[4-(methylsulfonyl)phenyl]-2,3'-bipyridine | CHEMBL416146 | ETORICOXIB
Type:
Small organic molecule
Emp. Form.:
C18H15ClN2O2S
Mol. Mass.:
358.842
SMILES:
Cc1ccc(cn1)-c1ncc(Cl)cc1-c1ccc(cc1)S(C)(=O)=O