Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
5-hydroxytryptamine receptor 2A
Ligand
BDBM21398
Substrate
n/a
Meas. Tech.
ChEMBL_2161776 (CHEMBL5046637)
Ki
23±n/a nM
Citation
 Marcinkowska, M; Bucki, A; Sniecikowska, J; Zag�rska, A; Fajkis-Zaj?czkowska, N; Siwek, A; Gluch-Lutwin, M; ?mudzki, P; Jastrzebska-Wiesek, M; Partyka, A; Weso?owska, A; Abram, M; Przejczowska-Pomierny, K; Cios, A; Wyska, E; Mika, K; Kota?ska, M; Mierzejewski, P; Kolaczkowski, M Multifunctional Arylsulfone and Arylsulfonamide-Based Ligands with Prominent Mood-Modulating Activity and Benign Safety Profile, Targeting Neuropsychiatric Symptoms of Dementia. J Med Chem 64:12603-12629 (2021) [PubMed] Article
Marcinkowska, M; Bucki, A; Sniecikowska, J; Zag�rska, A; Fajkis-Zaj?czkowska, N; Siwek, A; Gluch-Lutwin, M; ?mudzki, P; Jastrzebska-Wiesek, M; Partyka, A; Weso?owska, A; Abram, M; Przejczowska-Pomierny, K; Cios, A; Wyska, E; Mika, K; Kota?ska, M; Mierzejewski, P; Kolaczkowski, M Multifunctional Arylsulfone and Arylsulfonamide-Based Ligands with Prominent Mood-Modulating Activity and Benign Safety Profile, Targeting Neuropsychiatric Symptoms of Dementia. J Med Chem 64:12603-12629 (2021) [PubMed] Article More Info.:
Target
Name:
5-hydroxytryptamine receptor 2A
Synonyms:
5-HT-2 | 5-HT-2A | 5-HT2A | 5-hydroxytryptamine receptor 2A (5-HT-2A) | 5-hydroxytryptamine receptor 2A (5HT-2A) | 5-hydroxytryptamine receptor 2A (5HT2A) | 5HT2A_HUMAN | HTR2 | HTR2A | Serotonin receptor 2A
Type:
undefined
Mol. Mass.:
52607.65
Organism:
Homo sapiens (Human)
Description:
P28223
Residue:
471
Sequence:
MDILCEENTSLSSTTNSLMQLNDDTRLYSNDFNSGEANTSDAFNWTVDSENRTNLSCEGCLSPSCLSLLHLQEKNWSALLTAVVIILTIAGNILVIMAVSLEKKLQNATNYFLMSLAIADMLLGFLVMPVSMLTILYGYRWPLPSKLCAVWIYLDVLFSTASIMHLCAISLDRYVAIQNPIHHSRFNSRTKAFLKIIAVWTISVGISMPIPVFGLQDDSKVFKEGSCLLADDNFVLIGSFVSFFIPLTIMVITYFLTIKSLQKEATLCVSDLGTRAKLASFSFLPQSSLSSEKLFQRSIHREPGSYTGRRTMQSISNEQKACKVLGIVFFLFVVMWCPFFITNIMAVICKESCNEDVIGALLNVFVWIGYLSSAVNPLVYTLFNKTYRSAFSRYIQCQYKENKKPLQLILVNTIPALAYKSSQLQMGQKKNSKQDAKTTDNDCSMVALGKQHSEEASKDNSDGVNEKVSCV
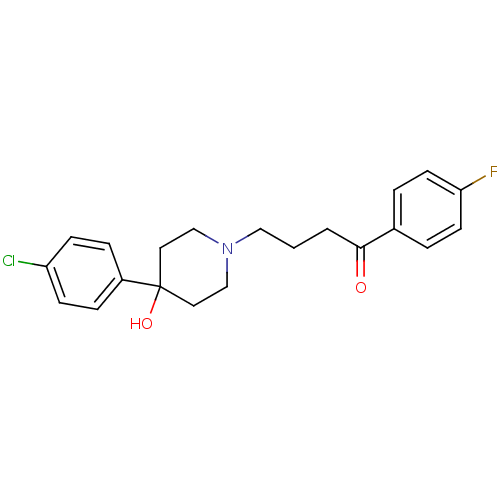
Inhibitor
Name:
BDBM21398
Synonyms:
4-[4-(4-Chloro-phenyl)-4-hydroxy-piperidin-1-yl]-1-(4-fluoro-phenyl)-butan-1-one;propionate(HCl) | 4-[4-(4-chlorophenyl)-4-hydroxypiperidin-1-yl]-1-(4-fluorophenyl)butan-1-one | CHEMBL54 | CHEMBL545608 | Haloperidol | Haloperidol, 1
Type:
Small organic molecule
Emp. Form.:
C21H23ClFNO2
Mol. Mass.:
375.864
SMILES:
OC1(CCN(CCCC(=O)c2ccc(F)cc2)CC1)c1ccc(Cl)cc1