Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Integrase
Ligand
BDBM50155343
Substrate
n/a
Meas. Tech.
ChEMBL_305350 (CHEMBL832730)
IC50
16000±n/a nM
Citation
 Krajewski, K; Marchand, C; Long, YQ; Pommier, Y; Roller, PP Synthesis and HIV-1 integrase inhibitory activity of dimeric and tetrameric analogs of indolicidin. Bioorg Med Chem Lett 14:5595-8 (2004) [PubMed] Article
Krajewski, K; Marchand, C; Long, YQ; Pommier, Y; Roller, PP Synthesis and HIV-1 integrase inhibitory activity of dimeric and tetrameric analogs of indolicidin. Bioorg Med Chem Lett 14:5595-8 (2004) [PubMed] Article More Info.:
Target
Name:
Integrase
Synonyms:
Human immunodeficiency virus type 1 integrase
Type:
PROTEIN
Mol. Mass.:
32231.48
Organism:
Human immunodeficiency virus 1
Description:
ChEMBL_90865
Residue:
288
Sequence:
FLDGIDKAQDEHEKYHSNWRAMASDFNLPPVVAKEIVASCDKCQLKGEAMHGQVDCSPGIWQLDCTHLEGKVILVAVHVASGYIEAEVIPAETGQETAYFLLKLAGRWPVKTIHTDNGSNFTSTTVKAACWWAGIKQEFGIPYNPQSQGVVESMNKELKKIIGQVRDQAEHLKTAVQMAVFIHNFKRKGGIGGYSAGERIVDIIATDIQTKELQKQITKIQNFRVYYRDSRDPLWKGPAKLLWKGEGAVVIQDNSDIKVVPRRKVKIIRDYGKQMAGDDCVASRQDED
Inhibitor
Name:
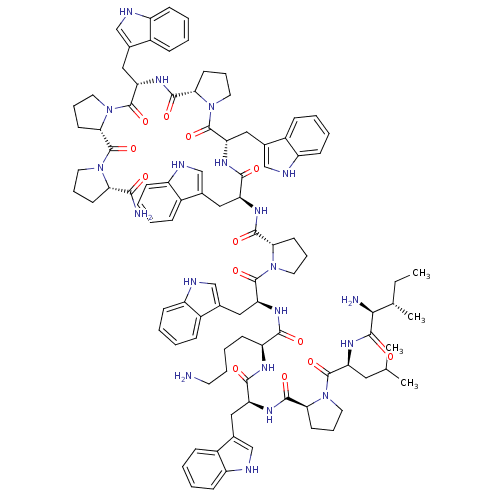
BDBM50155343
Synonyms:
CHEMBL407700 | ILPWKWPWWPWPP-NH2
Type:
Small organic molecule
Emp. Form.:
C98H122N20O13
Mol. Mass.:
1788.1435
SMILES:
CC[C@H](C)[C@H](N)C(=O)N[C@@H](CC(C)C)C(=O)N1CCC[C@H]1C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N1CCC[C@H]1C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N1CCC[C@H]1C(=O)N[C@@H](Cc1c[nH]c2ccccc12)C(=O)N1CCC[C@H]1C(=O)N1CCC[C@H]1C(N)=O