Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cytochrome P450 2C9
Ligand
BDBM50201124
Substrate
n/a
Meas. Tech.
ChEMBL_321361 (CHEMBL881505)
IC50
<18±n/a nM
Citation
 Wittman, M; Carboni, J; Attar, R; Balasubramanian, B; Balimane, P; Brassil, P; Beaulieu, F; Chang, C; Clarke, W; Dell, J; Eummer, J; Frennesson, D; Gottardis, M; Greer, A; Hansel, S; Hurlburt, W; Jacobson, B; Krishnananthan, S; Lee, FY; Li, A; Lin, TA; Liu, P; Ouellet, C; Sang, X; Saulnier, MG; Stoffan, K; Sun, Y; Velaparthi, U; Wong, H; Yang, Z; Zimmermann, K; Zoeckler, M; Vyas, D Discovery of a (1H-benzoimidazol-2-yl)-1H-pyridin-2-one (BMS-536924) inhibitor of insulin-like growth factor I receptor kinase with in vivo antitumor activity. J Med Chem 48:5639-43 (2005) [PubMed] Article
Wittman, M; Carboni, J; Attar, R; Balasubramanian, B; Balimane, P; Brassil, P; Beaulieu, F; Chang, C; Clarke, W; Dell, J; Eummer, J; Frennesson, D; Gottardis, M; Greer, A; Hansel, S; Hurlburt, W; Jacobson, B; Krishnananthan, S; Lee, FY; Li, A; Lin, TA; Liu, P; Ouellet, C; Sang, X; Saulnier, MG; Stoffan, K; Sun, Y; Velaparthi, U; Wong, H; Yang, Z; Zimmermann, K; Zoeckler, M; Vyas, D Discovery of a (1H-benzoimidazol-2-yl)-1H-pyridin-2-one (BMS-536924) inhibitor of insulin-like growth factor I receptor kinase with in vivo antitumor activity. J Med Chem 48:5639-43 (2005) [PubMed] Article More Info.:
Target
Name:
Cytochrome P450 2C9
Synonyms:
(R)-limonene 6-monooxygenase | (S)-limonene 6-monooxygenase | CP2C9_HUMAN | CYP2C10 | CYP2C9 | CYPIIC9 | Cytochrome P450 2C9 (CYP2C9 ) | Cytochrome P450 2C9 (CYP2C9) | P-450MP | P450 MP-4/MP-8 | P450 PB-1 | S-mephenytoin 4-hydroxylase
Type:
Enzyme
Mol. Mass.:
55636.33
Organism:
Homo sapiens (Human)
Description:
P11712
Residue:
490
Sequence:
MDSLVVLVLCLSCLLLLSLWRQSSGRGKLPPGPTPLPVIGNILQIGIKDISKSLTNLSKVYGPVFTLYFGLKPIVVLHGYEAVKEALIDLGEEFSGRGIFPLAERANRGFGIVFSNGKKWKEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICSIIFHKRFDYKDQQFLNLMEKLNENIKILSSPWIQICNNFSPIIDYFPGTHNKLLKNVAFMKSYILEKVKEHQESMDMNNPQDFIDCFLMKMEKEKHNQPSEFTIESLENTAVDLFGAGTETTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRSHMPYTDAVVHEVQRYIDLLPTSLPHAVTCDIKFRNYLIPKGTTILISLTSVLHDNKEFPNPEMFDPHHFLDEGGNFKKSKYFMPFSAGKRICVGEALAGMELFLFLTSILQNFNLKSLVDPKNLDTTPVVNGFASVPPFYQLCFIPV
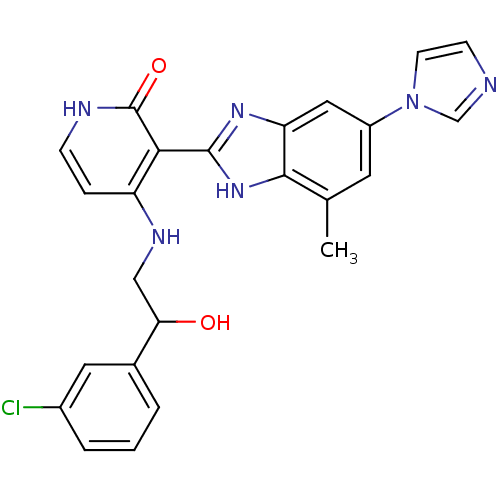
Inhibitor
Name:
BDBM50201124
Synonyms:
3-(6-(1H-imidazol-1-yl)-4-methyl-1H-benzo[d]imidazol-2-yl)-4-(2-(3-chlorophenyl)-2-hydroxyethylamino)pyridin-2(1H)-one | 4-[2-(3-Chloro-phenyl)-2-hydroxy-ethylamino]-3-(6-imidazol-1-yl-4-methyl-1H-benzoimidazol-2-yl)-1H-pyridin-2-one | CHEMBL231306
Type:
Small organic molecule
Emp. Form.:
C24H21ClN6O2
Mol. Mass.:
460.916
SMILES:
Cc1cc(cc2nc([nH]c12)-c1c(NCC(O)c2cccc(Cl)c2)cc[nH]c1=O)-n1ccnc1