Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cytochrome P450 3A4
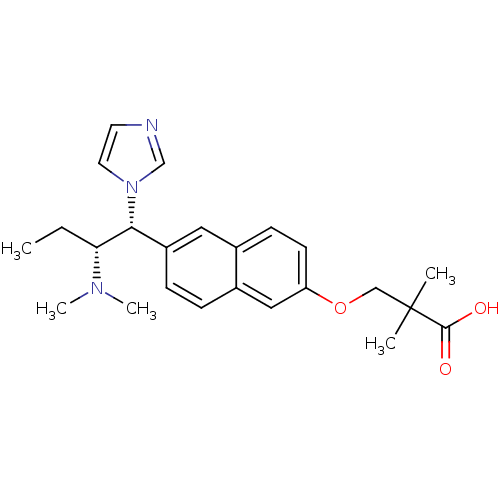
Ligand
BDBM50183224
Substrate
n/a
Meas. Tech.
ChEMBL_355280 (CHEMBL853201)
IC50
3300±n/a nM
Citation
 Mulvihill, MJ; Kan, JL; Cooke, A; Bhagwat, S; Beck, P; Bittner, M; Cesario, C; Keane, D; Lazarescu, V; Nigro, A; Nillson, C; Panicker, B; Smith, V; Srebernak, M; Sun, FL; O'Connor, M; Russo, S; Fischetti, G; Vrkljan, M; Winski, S; Castelhano, AL; Emerson, D; Gibson, NW 3-[6-(2-Dimethylamino-1-imidazol-1-yl-butyl)-naphthalen-2-yloxy]-2,2-dimethyl-propionic acid as a highly potent and selective retinoic acid metabolic blocking agent. Bioorg Med Chem Lett 16:2729-33 (2006) [PubMed] Article
Mulvihill, MJ; Kan, JL; Cooke, A; Bhagwat, S; Beck, P; Bittner, M; Cesario, C; Keane, D; Lazarescu, V; Nigro, A; Nillson, C; Panicker, B; Smith, V; Srebernak, M; Sun, FL; O'Connor, M; Russo, S; Fischetti, G; Vrkljan, M; Winski, S; Castelhano, AL; Emerson, D; Gibson, NW 3-[6-(2-Dimethylamino-1-imidazol-1-yl-butyl)-naphthalen-2-yloxy]-2,2-dimethyl-propionic acid as a highly potent and selective retinoic acid metabolic blocking agent. Bioorg Med Chem Lett 16:2729-33 (2006) [PubMed] Article More Info.:
Target
Name:
Cytochrome P450 3A4
Synonyms:
Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase
Type:
Enzyme
Mol. Mass.:
57349.57
Organism:
Homo sapiens (Human)
Description:
n/a
Residue:
503
Sequence:
MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMFDMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISIAEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYSMDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICVFPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSIIFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVVNETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFSKKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLGGLLQPEKPVVLKVESRDGTVSGA