Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Mycothiol S-conjugate amidase
Ligand
BDBM50227092
Substrate
n/a
Meas. Tech.
ChEMBL_452330 (CHEMBL902567)
IC50
33000±n/a nM
Citation
 Metaferia, BB; Fetterolf, BJ; Shazad-Ul-Hussan, S; Moravec, M; Smith, JA; Ray, S; Gutierrez-Lugo, MT; Bewley, CA Synthesis of natural product-inspired inhibitors of Mycobacterium tuberculosis mycothiol-associated enzymes: the first inhibitors of GlcNAc-Ins deacetylase. J Med Chem 50:6326-36 (2007) [PubMed] Article
Metaferia, BB; Fetterolf, BJ; Shazad-Ul-Hussan, S; Moravec, M; Smith, JA; Ray, S; Gutierrez-Lugo, MT; Bewley, CA Synthesis of natural product-inspired inhibitors of Mycobacterium tuberculosis mycothiol-associated enzymes: the first inhibitors of GlcNAc-Ins deacetylase. J Med Chem 50:6326-36 (2007) [PubMed] Article More Info.:
Target
Name:
Mycothiol S-conjugate amidase
Synonyms:
MCA_MYCTU | mca
Type:
PROTEIN
Mol. Mass.:
32719.79
Organism:
Mycobacterium tuberculosis
Description:
ChEMBL_626858
Residue:
288
Sequence:
MSELRLMAVHAHPDDESSKGAATLARYADEGHRVLVVTLTGGERGEILNPAMDLPDVHGRIAEIRRDEMTKAAEILGVEHTWLGFVDSGLPKGDLPPPLPDDCFARVPLEVSTEALVRVVREFRPHVMTTYDENGGYPHPDHIRCHQVSVAAYEAAGDFCRFPDAGEPWTVSKLYYVHGFLRERMQMLQDEFARHGQRGPFEQWLAYWDPDHDFLTSRVTTRVECSKYFSQRDDALRAHATQIDPNAEFFAAPLAWQERLWPTEEFELARSRIPARPPETELFAGIEP
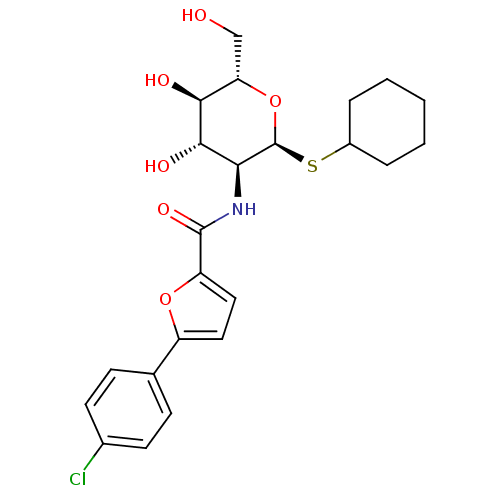
Inhibitor
Name:
BDBM50227092
Synonyms:
5-(4-chlorophenyl)-N-((2R,3R,4R,5S,6R)-2-(cyclohexylthio)-tetrahydro-4,5-dihydroxy-6-(hydroxymethyl)-2H-pyran-3-yl)furan-2-carboxamide | CHEMBL238766
Type:
Small organic molecule
Emp. Form.:
C23H28ClNO6S
Mol. Mass.:
481.99
SMILES:
OC[C@@H]1O[C@@H](SC2CCCCC2)[C@@H](NC(=O)c2ccc(o2)-c2ccc(Cl)cc2)[C@H](O)[C@H]1O