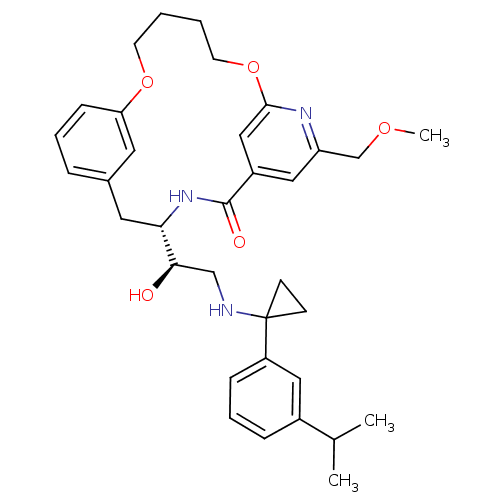
Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cathepsin D
Ligand
BDBM50305536
Substrate
n/a
Meas. Tech.
ChEMBL_604245 (CHEMBL1050390)
IC50
3±n/a nM
Citation
 Lerchner, A; Machauer, R; Betschart, C; Veenstra, S; Rueeger, H; McCarthy, C; Tintelnot-Blomley, M; Jaton, AL; Rabe, S; Desrayaud, S; Enz, A; Staufenbiel, M; Paganetti, P; Rondeau, JM; Neumann, U Macrocyclic BACE-1 inhibitors acutely reduce Abeta in brain after po application. Bioorg Med Chem Lett 20:603-7 (2010) [PubMed] Article
Lerchner, A; Machauer, R; Betschart, C; Veenstra, S; Rueeger, H; McCarthy, C; Tintelnot-Blomley, M; Jaton, AL; Rabe, S; Desrayaud, S; Enz, A; Staufenbiel, M; Paganetti, P; Rondeau, JM; Neumann, U Macrocyclic BACE-1 inhibitors acutely reduce Abeta in brain after po application. Bioorg Med Chem Lett 20:603-7 (2010) [PubMed] Article More Info.:
Target
Name:
Cathepsin D
Synonyms:
CATD_HUMAN | CPSD | CTSD | Cathepsin D [Precursor] | Cathepsin D heavy chain | Cathepsin D light chain | Cathepsin D precursor
Type:
Enzyme
Mol. Mass.:
44551.72
Organism:
Homo sapiens (Human)
Description:
Human proCathepsin D (SwissProt accession number P07339) was expressed in Sf9 cells, purified, and autoactivated.
Residue:
412
Sequence:
MQPSSLLPLALCLLAAPASALVRIPLHKFTSIRRTMSEVGGSVEDLIAKGPVSKYSQAVPAVTEGPIPEVLKNYMDAQYYGEIGIGTPPQCFTVVFDTGSSNLWVPSIHCKLLDIACWIHHKYNSDKSSTYVKNGTSFDIHYGSGSLSGYLSQDTVSVPCQSASSASALGGVKVERQVFGEATKQPGITFIAAKFDGILGMAYPRISVNNVLPVFDNLMQQKLVDQNIFSFYLSRDPDAQPGGELMLGGTDSKYYKGSLSYLNVTRKAYWQVHLDQVEVASGLTLCKEGCEAIVDTGTSLMVGPVDEVRELQKAIGAVPLIQGEYMIPCEKVSTLPAITLKLGGKGYKLSPEDYTLKVSQAGKTLCLSGFMGMDIPPPSGPLWILGDVFIGRYYTVFDRDNNRVGFAEAARL
Inhibitor
Name:
BDBM50305536
Synonyms:
(S)-4-{(R)-1-Hydroxy-2-[1-(3-isopropyl-phenyl)-cyclopropylamino]-ethyl}-19-methoxymethyl-11,16-dioxa-3,18-diaza-tricyclo[15.3.1.1*6,10*]docosa-1(21),6(22),7,9,17,19-hexaen-2-one | CHEMBL595267
Type:
Small organic molecule
Emp. Form.:
C34H43N3O5
Mol. Mass.:
573.7223
SMILES:
COCc1cc2cc(OCCCCOc3cccc(C[C@H](NC2=O)[C@H](O)CNC2(CC2)c2cccc(c2)C(C)C)c3)n1 |r|