Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cytochrome P450 3A4
Ligand
BDBM50296980
Substrate
n/a
Meas. Tech.
ChEMBL_699230 (CHEMBL1646952)
IC50
34000±n/a nM
Citation
 Simard, D; Leblanc, Y; Berthelette, C; Zaghdane, MH; Molinaro, C; Wang, Z; Gallant, M; Lau, S; Thao, T; Hamel, M; Stocco, R; Sawyer, N; Sillaots, S; Gervais, F; Houle, R; Lévesque, JF Azaindoles as potent CRTH2 receptor antagonists. Bioorg Med Chem Lett 21:841-5 (2011) [PubMed] Article
Simard, D; Leblanc, Y; Berthelette, C; Zaghdane, MH; Molinaro, C; Wang, Z; Gallant, M; Lau, S; Thao, T; Hamel, M; Stocco, R; Sawyer, N; Sillaots, S; Gervais, F; Houle, R; Lévesque, JF Azaindoles as potent CRTH2 receptor antagonists. Bioorg Med Chem Lett 21:841-5 (2011) [PubMed] Article More Info.:
Target
Name:
Cytochrome P450 3A4
Synonyms:
Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase
Type:
Enzyme
Mol. Mass.:
57349.57
Organism:
Homo sapiens (Human)
Description:
n/a
Residue:
503
Sequence:
MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMFDMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISIAEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYSMDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICVFPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSIIFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVVNETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFSKKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLGGLLQPEKPVVLKVESRDGTVSGA
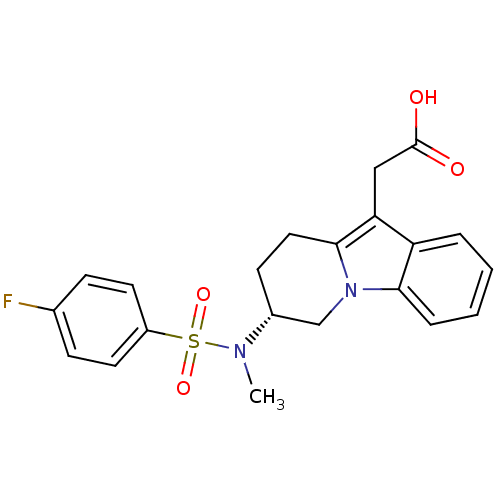
Inhibitor
Name:
BDBM50296980
Synonyms:
(R)-2-(7-(4-fluoro-N-methylphenylsulfonamido)-6,7,8,9-tetrahydropyrido[1,2-a]indol-10-yl)acetic acid | CHEMBL561132
Type:
Small organic molecule
Emp. Form.:
C21H21FN2O4S
Mol. Mass.:
416.466
SMILES:
CN([C@@H]1CCc2c(CC(O)=O)c3ccccc3n2C1)S(=O)(=O)c1ccc(F)cc1 |r|