Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cytochrome P450 3A4
Ligand
BDBM50338038
Substrate
n/a
Meas. Tech.
ChEMBL_727493 (CHEMBL1685481)
IC50
>100000±n/a nM
Citation
 Isshiki, Y; Kohchi, Y; Iikura, H; Matsubara, Y; Asoh, K; Murata, T; Kohchi, M; Mizuguchi, E; Tsujii, S; Hattori, K; Miura, T; Yoshimura, Y; Aida, S; Miwa, M; Saitoh, R; Murao, N; Okabe, H; Belunis, C; Janson, C; Lukacs, C; Schück, V; Shimma, N Design and synthesis of novel allosteric MEK inhibitor CH4987655 as an orally available anticancer agent. Bioorg Med Chem Lett 21:1795-801 (2011) [PubMed] Article
Isshiki, Y; Kohchi, Y; Iikura, H; Matsubara, Y; Asoh, K; Murata, T; Kohchi, M; Mizuguchi, E; Tsujii, S; Hattori, K; Miura, T; Yoshimura, Y; Aida, S; Miwa, M; Saitoh, R; Murao, N; Okabe, H; Belunis, C; Janson, C; Lukacs, C; Schück, V; Shimma, N Design and synthesis of novel allosteric MEK inhibitor CH4987655 as an orally available anticancer agent. Bioorg Med Chem Lett 21:1795-801 (2011) [PubMed] Article More Info.:
Target
Name:
Cytochrome P450 3A4
Synonyms:
Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase
Type:
Enzyme
Mol. Mass.:
57349.57
Organism:
Homo sapiens (Human)
Description:
n/a
Residue:
503
Sequence:
MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMFDMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISIAEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYSMDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICVFPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSIIFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVVNETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFSKKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLGGLLQPEKPVVLKVESRDGTVSGA
Inhibitor
Name:
BDBM50338038
Synonyms:
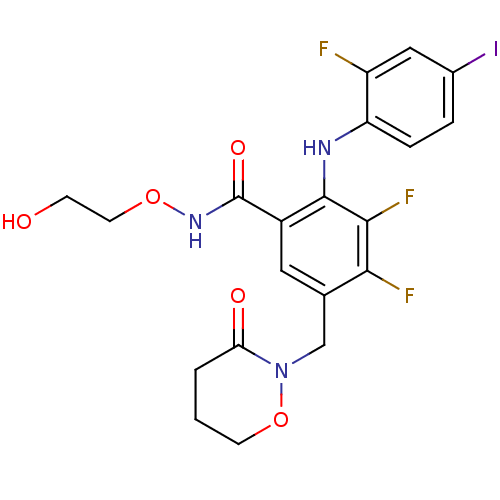
3,4-difluoro-2-(2-fluoro-4-iodophenylamino)-N-(2-hydroxyethoxy)-5-((3-oxomorpholino)methyl)benzamide | 3,4-difluoro-2-[(2-fluoro-4-iodophenyl)amino]-N-(2-hydroxyethoxy)-5-[(3-oxo-1,2-oxazinan-2-yl)methyl]benzamide | CHEMBL1614766 | US8575391, 334
Type:
Small organic molecule
Emp. Form.:
C20H19F3IN3O5
Mol. Mass.:
565.2816
SMILES:
OCCONC(=O)c1cc(CN2OCCCC2=O)c(F)c(F)c1Nc1ccc(I)cc1F