Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Histamine H1 receptor
Ligand
BDBM35938
Substrate
n/a
Meas. Tech.
ChEMBL_741662 (CHEMBL1769654)
Ki
7±n/a nM
Citation
 Procopiou, PA; Browning, C; Buckley, JM; Clark, KL; Fechner, L; Gore, PM; Hancock, AP; Hodgson, ST; Holmes, DS; Kranz, M; Looker, BE; Morriss, KM; Parton, DL; Russell, LJ; Slack, RJ; Sollis, SL; Vile, S; Watts, CJ The discovery of phthalazinone-based human H1 and H3 single-ligand antagonists suitable for intranasal administration for the treatment of allergic rhinitis. J Med Chem 54:2183-95 (2011) [PubMed] Article
Procopiou, PA; Browning, C; Buckley, JM; Clark, KL; Fechner, L; Gore, PM; Hancock, AP; Hodgson, ST; Holmes, DS; Kranz, M; Looker, BE; Morriss, KM; Parton, DL; Russell, LJ; Slack, RJ; Sollis, SL; Vile, S; Watts, CJ The discovery of phthalazinone-based human H1 and H3 single-ligand antagonists suitable for intranasal administration for the treatment of allergic rhinitis. J Med Chem 54:2183-95 (2011) [PubMed] Article More Info.:
Target
Name:
Histamine H1 receptor
Synonyms:
H1R | HH1R | HISTAMINE H1 | HRH1 | HRH1_HUMAN
Type:
G Protein-Coupled Receptor (GPCR)
Mol. Mass.:
55808.72
Organism:
Homo sapiens (Human)
Description:
Cell pellets from SK-N-MC cells transfected with human H1 receptor were used in binding assay.
Residue:
487
Sequence:
MSLPNSSCLLEDKMCEGNKTTMASPQLMPLVVVLSTICLVTVGLNLLVLYAVRSERKLHTVGNLYIVSLSVADLIVGAVVMPMNILYLLMSKWSLGRPLCLFWLSMDYVASTASIFSVFILCIDRYRSVQQPLRYLKYRTKTRASATILGAWFLSFLWVIPILGWNHFMQQTSVRREDKCETDFYDVTWFKVMTAIINFYLPTLLMLWFYAKIYKAVRQHCQHRELINRSLPSFSEIKLRPENPKGDAKKPGKESPWEVLKRKPKDAGGGSVLKSPSQTPKEMKSPVVFSQEDDREVDKLYCFPLDIVHMQAAAEGSSRDYVAVNRSHGQLKTDEQGLNTHGASEISEDQMLGDSQSFSRTDSDTTTETAPGKGKLRSGSNTGLDYIKFTWKRLRSHSRQYVSGLHMNRERKAAKQLGFIMAAFILCWIPYFIFFMVIAFCKNCCNEHLHMFTIWLGYINSTLNPLIYPLCNENFKKTFKRILHIRS
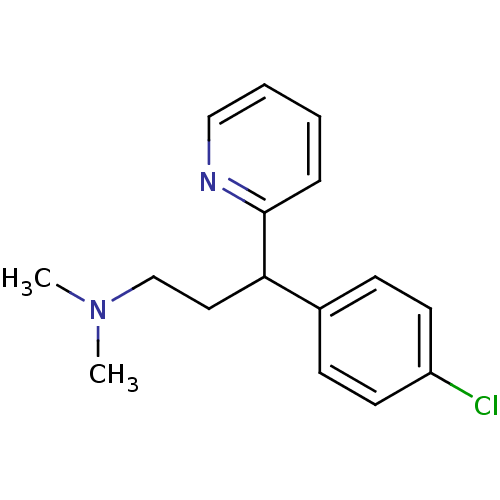
Inhibitor
Name:
BDBM35938
Synonyms:
1-(p-chlorophenyl)-1-(2-pyridyl)-3-N,N-dimethylpropylamine | 1-(p-chlorophenyl)-1-(2-pyridyl)-3-dimethylaminopropane | 3-(p-chlorophenyl)-3-(2-pyridyl)-N,N-dimethylpropylamine | 5-HT,N-ACETYL | CHEMBL505 | CHLORPHENIRAMINE | CHLORPHENIRAMINE MALEATE | Polaramin | Polaramine | Polaronil | [3H]Chlorphenamine | [3H]Chlorpheniramine | chlorophenylpyridamine | chlorphenamine | clorfeniramina | d-Chlorpheniramine | gamma-(4-chlorophenyl)-N,N-dimethyl-2-pyridinepropanamine | gamma-(4-chlorophenyl)-gamma-(2-pyridyl)propyldimethylamine
Type:
radiolabeled ligand
Emp. Form.:
C16H19ClN2
Mol. Mass.:
274.788
SMILES:
CN(C)CCC(c1ccc(Cl)cc1)c1ccccn1