Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Discoidin domain-containing receptor 2
Ligand
BDBM4779
Substrate
n/a
Meas. Tech.
ChEMBL_774257 (CHEMBL1908474)
Kd
>10000±n/a nM
Citation
 Davis, MI; Hunt, JP; Herrgard, S; Ciceri, P; Wodicka, LM; Pallares, G; Hocker, M; Treiber, DK; Zarrinkar, PP Comprehensive analysis of kinase inhibitor selectivity. Nat Biotechnol 29:1046-51 (2011) [PubMed] Article
Davis, MI; Hunt, JP; Herrgard, S; Ciceri, P; Wodicka, LM; Pallares, G; Hocker, M; Treiber, DK; Zarrinkar, PP Comprehensive analysis of kinase inhibitor selectivity. Nat Biotechnol 29:1046-51 (2011) [PubMed] Article More Info.:
Target
Name:
Discoidin domain-containing receptor 2
Synonyms:
DDR2 | DDR2_HUMAN | Discoidin domain receptor 2 (DDR2) | Discoidin domain-containing receptor 2 | Discoidin domain-containing receptor 2 (DDR2) | Epithelial discoidin domain-containing receptor 2 (DDR2) | NTRKR3 | TKT | TYRO10
Type:
Protein
Mol. Mass.:
96717.07
Organism:
Homo sapiens (Human)
Description:
Q16832
Residue:
855
Sequence:
MILIPRMLLVLFLLLPILSSAKAQVNPAICRYPLGMSGGQIPDEDITASSQWSESTAAKYGRLDSEEGDGAWCPEIPVEPDDLKEFLQIDLHTLHFITLVGTQGRHAGGHGIEFAPMYKINYSRDGTRWISWRNRHGKQVLDGNSNPYDIFLKDLEPPIVARFVRFIPVTDHSMNVCMRVELYGCVWLDGLVSYNAPAGQQFVLPGGSIIYLNDSVYDGAVGYSMTEGLGQLTDGVSGLDDFTQTHEYHVWPGYDYVGWRNESATNGYIEIMFEFDRIRNFTTMKVHCNNMFAKGVKIFKEVQCYFRSEASEWEPNAISFPLVLDDVNPSARFVTVPLHHRMASAIKCQYHFADTWMMFSEITFQSDAAMYNNSEALPTSPMAPTTYDPMLKVDDSNTRILIGCLVAIIFILLAIIVIILWRQFWQKMLEKASRRMLDDEMTVSLSLPSDSSMFNNNRSSSPSEQGSNSTYDRIFPLRPDYQEPSRLIRKLPEFAPGEEESGCSGVVKPVQPSGPEGVPHYAEADIVNLQGVTGGNTYSVPAVTMDLLSGKDVAVEEFPRKLLTFKEKLGEGQFGEVHLCEVEGMEKFKDKDFALDVSANQPVLVAVKMLRADANKNARNDFLKEIKIMSRLKDPNIIHLLAVCITDDPLCMITEYMENGDLNQFLSRHEPPNSSSSDVRTVSYTNLKFMATQIASGMKYLSSLNFVHRDLATRNCLVGKNYTIKIADFGMSRNLYSGDYYRIQGRAVLPIRWMSWESILLGKFTTASDVWAFGVTLWETFTFCQEQPYSQLSDEQVIENTGEFFRDQGRQTYLPQPAICPDSVYKLMLSCWRRDTKNRPSFQEIHLLLLQQGDE
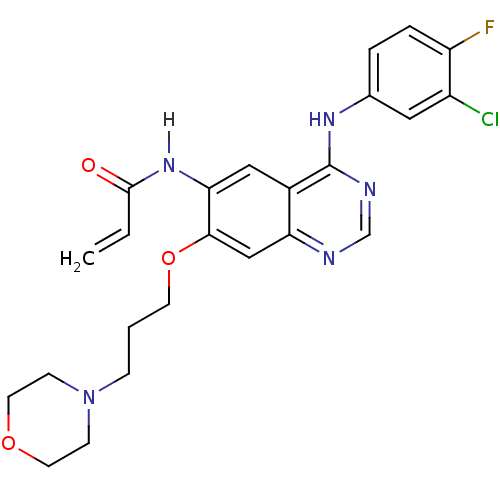
Inhibitor
Name:
BDBM4779
Synonyms:
CHEMBL31965 | CHEMBL545315 | CI-1033 | Canertinib | N-{4-[(3-chloro-4-fluorophenyl)amino]-7-[3-(morpholin-4-yl)propoxy]quinazolin-6-yl}prop-2-enamide | PD0183805 | US10507209, Compound CI-1033 | US9730934, CI-1033 | cid_156414
Type:
Small organic molecule
Emp. Form.:
C24H25ClFN5O3
Mol. Mass.:
485.938
SMILES:
Fc1ccc(Nc2ncnc3cc(OCCCN4CCOCC4)c(NC(=O)C=C)cc23)cc1Cl