Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
DNA topoisomerase 2-beta
Ligand
BDBM50286489
Substrate
n/a
Meas. Tech.
ChEBML_51583
EC50
>50000±n/a nM
Citation
 Wentland, MP; Aldous, SC; Gruett, MD; Perni, RB; Powles, RG; Danz, DW; Klingbeil, KM; Peverly, A; Robinson, RG; Corbett, TH; Rake, JB; Coughlin, SA The antitumor activity of novel pyrazoloquinoline derivatives Bioorg Med Chem Lett 5:405-410 (1995) Article
Wentland, MP; Aldous, SC; Gruett, MD; Perni, RB; Powles, RG; Danz, DW; Klingbeil, KM; Peverly, A; Robinson, RG; Corbett, TH; Rake, JB; Coughlin, SA The antitumor activity of novel pyrazoloquinoline derivatives Bioorg Med Chem Lett 5:405-410 (1995) Article More Info.:
Target
Name:
DNA topoisomerase 2-beta
Synonyms:
DNA topoisomerase II | DNA topoisomerase II beta | TOP2B | TOP2B_HUMAN
Type:
PROTEIN
Mol. Mass.:
183284.13
Organism:
Homo sapiens (Human)
Description:
ChEMBL_51583
Residue:
1626
Sequence:
MAKSGGCGAGAGVGGGNGALTWVTLFDQNNAAKKEESETANKNDSSKKLSVERVYQKKTQLEHILLRPDTYIGSVEPLTQFMWVYDEDVGMNCREVTFVPGLYKIFDEILVNAADNKQRDKNMTCIKVSIDPESNIISIWNNGKGIPVVEHKVEKVYVPALIFGQLLTSSNYDDDEKKVTGGRNGYGAKLCNIFSTKFTVETACKEYKHSFKQTWMNNMMKTSEAKIKHFDGEDYTCITFQPDLSKFKMEKLDKDIVALMTRRAYDLAGSCRGVKVMFNGKKLPVNGFRSYVDLYVKDKLDETGVALKVIHELANERWDVCLTLSEKGFQQISFVNSIATTKGGRHVDYVVDQVVGKLIEVVKKKNKAGVSVKPFQVKNHIWVFINCLIENPTFDSQTKENMTLQPKSFGSKCQLSEKFFKAASNCGIVESILNWVKFKAQTQLNKKCSSVKYSKIKGIPKLDDANDAGGKHSLECTLILTEGDSAKSLAVSGLGVIGRDRYGVFPLRGKILNVREASHKQIMENAEINNIIKIVGLQYKKSYDDAESLKTLRYGKIMIMTDQDQDGSHIKGLLINFIHHNWPSLLKHGFLEEFITPIVKASKNKQELSFYSIPEFDEWKKHIENQKAWKIKYYKGLGTSTAKEAKEYFADMERHRILFRYAGPEDDAAITLAFSKKKIDDRKEWLTNFMEDRRQRRLHGLPEQFLYGTATKHLTYNDFINKELILFSNSDNERSIPSLVDGFKPGQRKVLFTCFKRNDKREVKVAQLAGSVAEMSAYHHGEQALMMTIVNLAQNFVGSNNINLLQPIGQFGTRLHGGKDAASPRYIFTMLSTLARLLFPAVDDNLLKFLYDDNQRVEPEWYIPIIPMVLINGAEGIGTGWACKLPNYDAREIVNNVRRMLDGLDPHPMLPNYKNFKGTIQELGQNQYAVSGEIFVVDRNTVEITELPVRTWTQVYKEQVLEPMLNGTDKTPALISDYKEYHTDTTVKFVVKMTEEKLAQAEAAGLHKVFKLQTTLTCNSMVLFDHMGCLKKYETVQDILKEFFDLRLSYYGLRKEWLVGMLGAESTKLNNQARFILEKIQGKITIENRSKKDLIQMLVQRGYESDPVKAWKEAQEKAAEEDETQNQHDDSSSDSGTPSGPDFNYILNMSLWSLTKEKVEELIKQRDAKGREVNDLKRKSPSDLWKEDLAAFVEELDKVESQEREDVLAGMSGKAIKGKVGKPKVKKLQLEETMPSPYGRRIIPEITAMKADASKKLLKKKKGDLDTAAVKVEFDEEFSGAPVEGAGEEALTPSVPINKGPKPKREKKEPGTRVRKTPTSSGKPSAKKVKKRNPWSDDESKSESDLEETEPVVIPRDSLLRRAAAERPKYTFDFSEEEDDDADDDDDDNNDLEELKVKASPITNDGEDEFVPSDGLDKDEYTFSPGKSKATPEKSLHDKKSQDFGNLFSFPSYSQKSEDDSAKFDSNEEDSASVFSPSFGLKQTDKVPSKTVAAKKGKPSSDTVPKPKRAPKQKKVVEAVNSDSDSEFGIPKKTTTPKGKGRGAKKRKASGSENEGDYNPGRKTSKTTSKKPKKTSFDQDSDVDIFPSDFPTEPPSLPRTGRARKEVKYFAESDEEEDDVDFAMFN
Inhibitor
Name:
BDBM50286489
Synonyms:
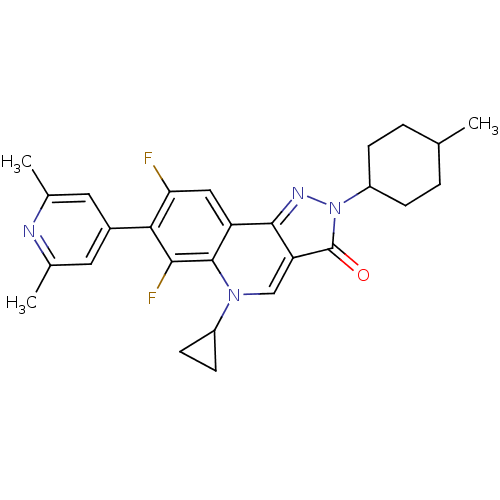
5-Cyclopropyl-7-(2,6-dimethyl-pyridin-4-yl)-6,8-difluoro-2-(4-methyl-cyclohexyl)-2,5-dihydro-pyrazolo[4,3-c]quinolin-3-one | CHEMBL131600
Type:
Small organic molecule
Emp. Form.:
C27H28F2N4O
Mol. Mass.:
462.5342
SMILES:
CC1CCC(CC1)n1nc2c(cn(C3CC3)c3c(F)c(c(F)cc23)-c2cc(C)nc(C)c2)c1=O |(16.12,-2.01,;15.35,-3.34,;13.81,-3.34,;13.03,-4.69,;13.82,-6,;15.35,-6,;16.12,-4.69,;13.05,-7.35,;11.53,-7.5,;11.21,-9.01,;12.54,-9.78,;12.54,-11.32,;11.21,-12.09,;11.21,-13.63,;10.44,-14.98,;11.98,-14.96,;9.88,-11.32,;8.55,-12.09,;8.55,-13.63,;7.21,-11.32,;7.21,-9.78,;5.86,-9.01,;8.55,-9.01,;9.88,-9.78,;5.88,-12.09,;5.88,-13.63,;4.55,-14.4,;4.56,-15.94,;3.2,-13.63,;3.2,-12.09,;1.85,-11.3,;4.53,-11.32,;13.7,-8.73,;15.2,-9.05,)|