Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Prothrombin
Ligand
BDBM50037974
Substrate
n/a
Meas. Tech.
ChEMBL_208319 (CHEMBL812837)
Ki
10±n/a nM
Citation
 Hilpert, K; Ackermann, J; Banner, DW; Gast, A; Gubernator, K; Hadváry, P; Labler, L; Müller, K; Schmid, G; Tschopp, TB Design and synthesis of potent and highly selective thrombin inhibitors. J Med Chem 37:3889-901 (1994) [PubMed] Article
Hilpert, K; Ackermann, J; Banner, DW; Gast, A; Gubernator, K; Hadváry, P; Labler, L; Müller, K; Schmid, G; Tschopp, TB Design and synthesis of potent and highly selective thrombin inhibitors. J Med Chem 37:3889-901 (1994) [PubMed] Article More Info.:
Target
Name:
Prothrombin
Synonyms:
Activation peptide fragment 1 | Activation peptide fragment 2 | Coagulation factor II | F2 | Prothrombin precursor | THRB_HUMAN | Thrombin heavy chain | Thrombin light chain
Type:
Protein
Mol. Mass.:
70029.57
Organism:
Homo sapiens (Human)
Description:
P00734
Residue:
622
Sequence:
MAHVRGLQLPGCLALAALCSLVHSQHVFLAPQQARSLLQRVRRANTFLEEVRKGNLERECVEETCSYEEAFEALESSTATDVFWAKYTACETARTPRDKLAACLEGNCAEGLGTNYRGHVNITRSGIECQLWRSRYPHKPEINSTTHPGADLQENFCRNPDSSTTGPWCYTTDPTVRRQECSIPVCGQDQVTVAMTPRSEGSSVNLSPPLEQCVPDRGQQYQGRLAVTTHGLPCLAWASAQAKALSKHQDFNSAVQLVENFCRNPDGDEEGVWCYVAGKPGDFGYCDLNYCEEAVEEETGDGLDEDSDRAIEGRTATSEYQTFFNPRTFGSGEADCGLRPLFEKKSLEDKTERELLESYIDGRIVEGSDAEIGMSPWQVMLFRKSPQELLCGASLISDRWVLTAAHCLLYPPWDKNFTENDLLVRIGKHSRTRYERNIEKISMLEKIYIHPRYNWRENLDRDIALMKLKKPVAFSDYIHPVCLPDRETAASLLQAGYKGRVTGWGNLKETWTANVGKGQPSVLQVVNLPIVERPVCKDSTRIRITDNMFCAGYKPDEGKRGDACEGDSGGPFVMKSPFNNRWYQMGIVSWGEGCDRDGKYGFYTHVFRLKKWIQKVIDQFGE
Inhibitor
Name:
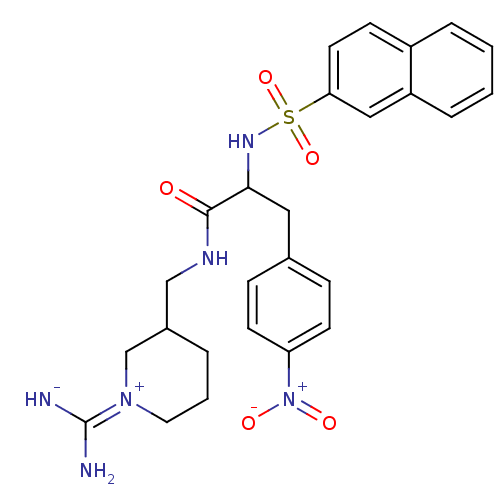
BDBM50037974
Synonyms:
(R)-N-((S)-1-Carbamimidoyl-piperidin-3-ylmethyl)-2-(naphthalene-2-sulfonylamino)-3-(4-nitro-phenyl)-propionamide; hydrochloride | CHEMBL555314
Type:
Small organic molecule
Emp. Form.:
C26H30N6O5S
Mol. Mass.:
538.619
SMILES:
N\C([NH-])=[N+]1/CCCC(CNC(=O)C(Cc2ccc(cc2)[N+]([O-])=O)NS(=O)(=O)c2ccc3ccccc3c2)C1