Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Endothelin receptor type B
Ligand
BDBM50068674
Substrate
n/a
Meas. Tech.
ChEMBL_64185 (CHEMBL878144)
Ki
1700±n/a nM
Citation
 Murugesan, N; Gu, Z; Stein, PD; Bisaha, S; Spergel, S; Girotra, R; Lee, VG; Lloyd, J; Misra, RN; Schmidt, J; Mathur, A; Stratton, L; Kelly, YF; Bird, E; Waldron, T; Liu, EC; Zhang, R; Lee, H; Serafino, R; Abboa-Offei, B; Mathers, P; Giancarli, M; Seymour, AA; Webb, ML; Hunt, JT Biphenylsulfonamide endothelin antagonists: structure-activity relationships of a series of mono- and disubstituted analogues and pharmacology of the orally active endothelin antagonist 2'-amino-N- (3,4-dimethyl-5-isoxazolyl)-4'-(2-methylpropyl)[1, 1'-biphenyl]-2-sulfonamide (BMS-187308). J Med Chem 41:5198-218 (1999) [PubMed] Article
Murugesan, N; Gu, Z; Stein, PD; Bisaha, S; Spergel, S; Girotra, R; Lee, VG; Lloyd, J; Misra, RN; Schmidt, J; Mathur, A; Stratton, L; Kelly, YF; Bird, E; Waldron, T; Liu, EC; Zhang, R; Lee, H; Serafino, R; Abboa-Offei, B; Mathers, P; Giancarli, M; Seymour, AA; Webb, ML; Hunt, JT Biphenylsulfonamide endothelin antagonists: structure-activity relationships of a series of mono- and disubstituted analogues and pharmacology of the orally active endothelin antagonist 2'-amino-N- (3,4-dimethyl-5-isoxazolyl)-4'-(2-methylpropyl)[1, 1'-biphenyl]-2-sulfonamide (BMS-187308). J Med Chem 41:5198-218 (1999) [PubMed] Article More Info.:
Target
Name:
Endothelin receptor type B
Synonyms:
EDNRB_RAT | ENDOTHELIN B | ET-B | Ednrb | Endothelin receptor | Endothelin receptor non-selective type
Type:
Enzyme Catalytic Domain
Mol. Mass.:
49483.43
Organism:
RAT
Description:
ENDOTHELIN B EDNRB RAT::P21451
Residue:
442
Sequence:
MQSSASRCGRALVALLLACGLLGVWGEKRGFPPAQATPSLLGTKEVMTPPTKTSWTRGSNSSLMRSSAPAEVTKGGRVAGVPPRSFPPPCQRKIEINKTFKYINTIVSCLVFVLGIIGNSTLLRIIYKNKCMRNGPNILIASLALGDLLHIIIDIPINAYKLLAGDWPFGAEMCKLVPFIQKASVGITVLSLCALSIDRYRAVASWSRIKGIGVPKWTAVEIVLIWVVSVVLAVPEAIGFDVITSDYKGKPLRVCMLNPFQKTAFMQFYKTAKDWWLFSFYFCLPLAITAIFYTLMTCEMLRKKSGMQIALNDHLKQRREVAKTVFCLVLVFALCWLPLHLSRILKLTLYDQSNPQRCELLSFLLVLDYIGINMASLNSCINPIALYLVSKRFKNCFKSCLCCWCQTFEEKQSLEEKQSCLKFKANDHGYDNFRSSNKYSSS
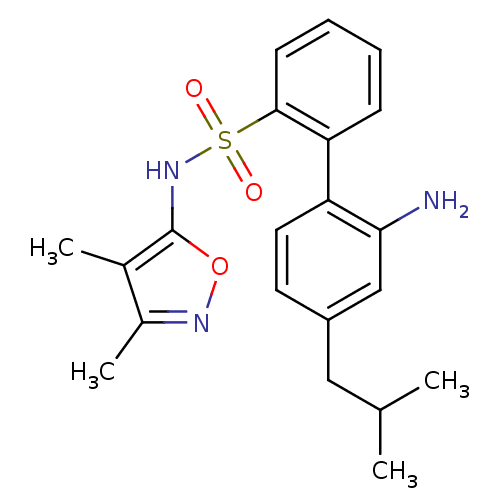
Inhibitor
Name:
BDBM50068674
Synonyms:
2'-Amino-4'-isobutyl-biphenyl-2-sulfonic acid (3,4-dimethyl-isoxazol-5-yl)-amide | 2'-Amino-4'-isobutyl-biphenyl-2-sulfonic acid (3,4-dimethyl-isoxazol-5-yl)-amide (BMS 187308) | BMS-187308 | CHEMBL106684
Type:
Small organic molecule
Emp. Form.:
C21H25N3O3S
Mol. Mass.:
399.507
SMILES:
CC(C)Cc1ccc(c(N)c1)-c1ccccc1S(=O)(=O)Nc1onc(C)c1C