Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Endoplasmin
Ligand
BDBM21220
Substrate
n/a
Meas. Tech.
ChEMBL_1478279 (CHEMBL3428519)
Kd
200±n/a nM
Citation
 Patel, HJ; Patel, PD; Ochiana, SO; Yan, P; Sun, W; Patel, MR; Shah, SK; Tramentozzi, E; Brooks, J; Bolaender, A; Shrestha, L; Stephani, R; Finotti, P; Leifer, C; Li, Z; Gewirth, DT; Taldone, T; Chiosis, G Structure-activity relationship in a purine-scaffold compound series with selectivity for the endoplasmic reticulum Hsp90 paralog Grp94. J Med Chem 58:3922-43 (2015) [PubMed] Article
Patel, HJ; Patel, PD; Ochiana, SO; Yan, P; Sun, W; Patel, MR; Shah, SK; Tramentozzi, E; Brooks, J; Bolaender, A; Shrestha, L; Stephani, R; Finotti, P; Leifer, C; Li, Z; Gewirth, DT; Taldone, T; Chiosis, G Structure-activity relationship in a purine-scaffold compound series with selectivity for the endoplasmic reticulum Hsp90 paralog Grp94. J Med Chem 58:3922-43 (2015) [PubMed] Article More Info.:
Target
Name:
Endoplasmin
Synonyms:
94 kDa glucose-regulated protein | 98 kDa protein kinase | ENPL_PIG | Endoplasmin | GRP-94 | GRP94 | GRP94 | HSP90B1 | Heat shock protein 90 kDa beta member 1 | PPK 98 | TRA1 | gp96 homolog | ppk98
Type:
PROTEIN
Mol. Mass.:
92418.20
Organism:
Sus scrofa
Description:
ChEMBL_109618
Residue:
804
Sequence:
MRALWVLGLCCVLLTFGSVRAEDEVDVDGTVEEDLGKSREGSRTDDEVVQREEEAIQLDGLNASQIRELREKSEKFAFQAEVNRMMKLIINSLYKNKEIFLRELISNASDALDKIRLISLTDENALAGNEELTVKIKCDKEKNLLHVTDTGVGMTREELVKNLGTIAKSGTSEFLNKMAEAQEDGQSTSELIGQFGVGFYSAFLVADKVIVTSKHNNDTQHIWESDSNEFSVIADPRGNTLGRGTTITLVLKEEASDYLELDTIKNLVKKYSQFINFPIYVWSSKTETVEEPMEEEEAAKEEKEESDDEAAVEEEEEEKKPKTKKVEKTVWDWELMNDIKPIWQRPSKEVEDDEYKAFYKSFSKESDDPMAYIHFTAEGEVTFKSILFVPTSAPRGLFDEYGSKKSDYIKLYVRRVFITDDFHDMMPKYLNFVKGVVDSDDLPLNVSRETLQQHKLLKVIRKKLVRKTLDMIKKIADEKYNDTFWKEFGTNIKLGVIEDHSNRTRLAKLLRFQSSHHPSDITSLDQYVERMKEKQDKIYFMAGSSRKEAESSPFVERLLKKGYEVIYLTEPVDEYCIQALPEFDGKRFQNVAKEGVKFDESEKSKENREAVEKEFEPLLNWMKDKALKDKIEKAVVSQRLTESPCALVASQYGWSGNMERIMKAQAYQTGKDISTNYYASQKKTFEINPRHPLIRDMLRRVKEDEDDKTVSDLAVVLFETATLRSGYLLPDTKAYGDRIERMLRLSLNIDPDAKVEEEPEEEPEETTEDTTEDTEQDDDEEMDAGADEEEQETSETSTAEKDEL
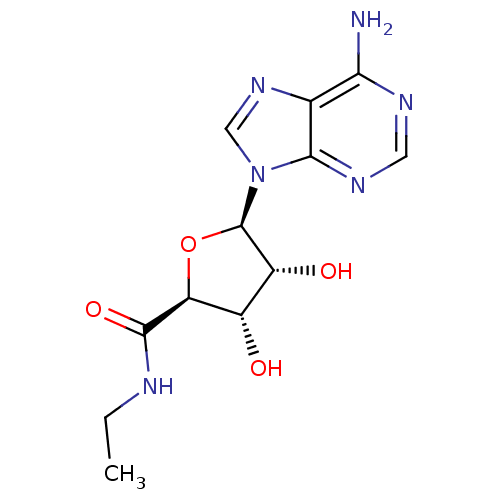
Inhibitor
Name:
BDBM21220
Synonyms:
(2S,3S,4R,5R)-5-(6-amino-9H-purin-9-yl)-N-ethyl-3,4-dihydroxyoxolane-2-carboxamide | Adenosine analog, 3 | Adenosine, N6-Ethyl-carboxamido | Adenosine-5-(N-ethylcarboxamide) | CHEMBL464859 | N-Ethylcarboxamidoadenosine | NECA
Type:
Antineoplastic Agent
Emp. Form.:
C12H16N6O4
Mol. Mass.:
308.2932
SMILES:
CCNC(=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(N)ncnc12