Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Zinc finger protein 664
Ligand
BDBM21398
Substrate
n/a
Meas. Tech.
ChEBML_1659535
Ki
6.6±n/a nM
Citation
 Soeberdt, M; Molenveld, P; Storcken, RP; Bouzanne des Mazery, R; Sterk, GJ; Autar, R; Bolster, MG; Wagner, C; Aerts, SN; van Holst, FR; Wegert, A; Tangherlini, G; Frehland, B; Schepmann, D; Metze, D; Lotts, T; Knie, U; Lin, KY; Huang, TY; Lai, CC; St�nder, S; W�nsch, B; Abels, C Design and Synthesis of Enantiomerically Pure Decahydroquinoxalines as Potent and Selective�-Opioid Receptor Agonists with Anti-Inflammatory Activity in Vivo. J Med Chem 60:2526-2551 (2017) [PubMed] Article
Soeberdt, M; Molenveld, P; Storcken, RP; Bouzanne des Mazery, R; Sterk, GJ; Autar, R; Bolster, MG; Wagner, C; Aerts, SN; van Holst, FR; Wegert, A; Tangherlini, G; Frehland, B; Schepmann, D; Metze, D; Lotts, T; Knie, U; Lin, KY; Huang, TY; Lai, CC; St�nder, S; W�nsch, B; Abels, C Design and Synthesis of Enantiomerically Pure Decahydroquinoxalines as Potent and Selective�-Opioid Receptor Agonists with Anti-Inflammatory Activity in Vivo. J Med Chem 60:2526-2551 (2017) [PubMed] Article More Info.:
Target
Name:
Zinc finger protein 664
Synonyms:
ZFOC1 | ZN664_CAVPO | ZNF664 | Zinc finger protein 664 | Zinc finger protein from organ of Corti
Type:
PROTEIN
Mol. Mass.:
30308.74
Organism:
Cavia porcellus
Description:
ChEMBL_117362
Residue:
261
Sequence:
MIYKCPMCREFFSERADLFMHQKVHTAEKPHKCDKCDKGFFHISELHIHWRDHTGEKVYKCDDCGKDFSTTTKLNRHKKIHTVEKPYKCYECGKAFNWSPHLQIHMRVHTGEKPYVCSECGRGFSNSSNLCMHQRVHTGEKPFKCEECGKAFRHTSSLCMHQRVHTGEKPYKCYECGKAFSQSSSLCIHQRVHTGEKPYRCCGCGKAFSQSSSLCIHQRVHTGEKPFKCDECGKAFSQSTSLCIHQRVHTKERNHLKISVI
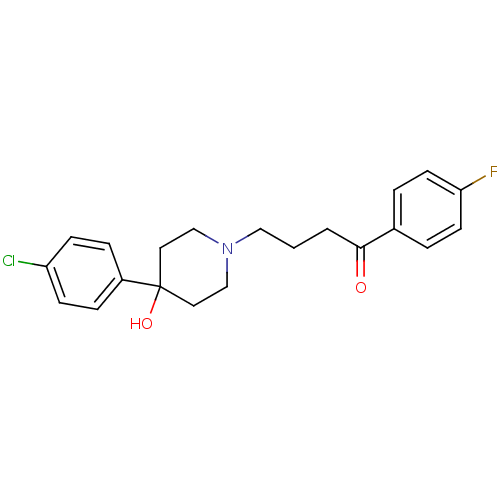
Inhibitor
Name:
BDBM21398
Synonyms:
4-[4-(4-Chloro-phenyl)-4-hydroxy-piperidin-1-yl]-1-(4-fluoro-phenyl)-butan-1-one;propionate(HCl) | 4-[4-(4-chlorophenyl)-4-hydroxypiperidin-1-yl]-1-(4-fluorophenyl)butan-1-one | CHEMBL54 | CHEMBL545608 | Haloperidol | Haloperidol, 1
Type:
Small organic molecule
Emp. Form.:
C21H23ClFNO2
Mol. Mass.:
375.864
SMILES:
OC1(CCN(CCCC(=O)c2ccc(F)cc2)CC1)c1ccc(Cl)cc1