Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Cytochrome P450 3A4
Ligand
BDBM213822
Substrate
Testosterone
Meas. Tech.
CYP Activity Assay
Temperature
310.15±n/a K
IC50
>2.0e+4±n/a nM
Comments
extracted
Citation
 Hargrove, TY; Wawrzak, Z; Alexander, PW; Chaplin, JH; Keenan, M; Charman, SA; Perez, CJ; Waterman, MR; Chatelain, E; Lepesheva, GI Complexes of Trypanosoma cruzi sterol 14a-demethylase (CYP51) with two pyridine-based drug candidates for Chagas disease: structural basis for pathogen selectivity. J Biol Chem 288:31602-15 (2013) [PubMed] Article
Hargrove, TY; Wawrzak, Z; Alexander, PW; Chaplin, JH; Keenan, M; Charman, SA; Perez, CJ; Waterman, MR; Chatelain, E; Lepesheva, GI Complexes of Trypanosoma cruzi sterol 14a-demethylase (CYP51) with two pyridine-based drug candidates for Chagas disease: structural basis for pathogen selectivity. J Biol Chem 288:31602-15 (2013) [PubMed] Article More Info.:
Target
Name:
Cytochrome P450 3A4
Synonyms:
Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase
Type:
Enzyme
Mol. Mass.:
57349.57
Organism:
Homo sapiens (Human)
Description:
n/a
Residue:
503
Sequence:
MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMFDMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISIAEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYSMDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICVFPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSIIFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVVNETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFSKKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLGGLLQPEKPVVLKVESRDGTVSGA
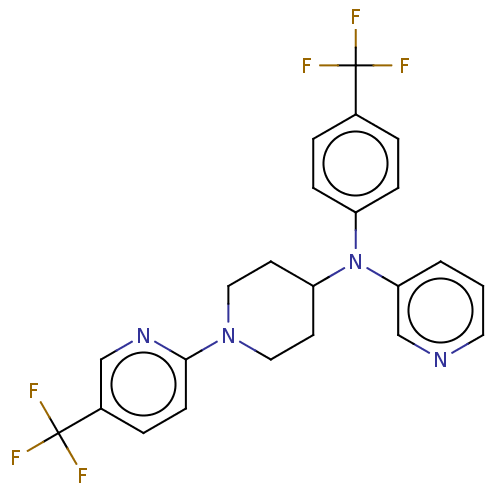
Inhibitor
Name:
BDBM213822
Synonyms:
EPL-BS0967 (UDD)
Type:
Small organic molecule
Emp. Form.:
C23H20F6N4
Mol. Mass.:
466.4221
SMILES:
FC(F)(F)c1ccc(cc1)N(C1CCN(CC1)c1ccc(cn1)C(F)(F)F)c1cccnc1
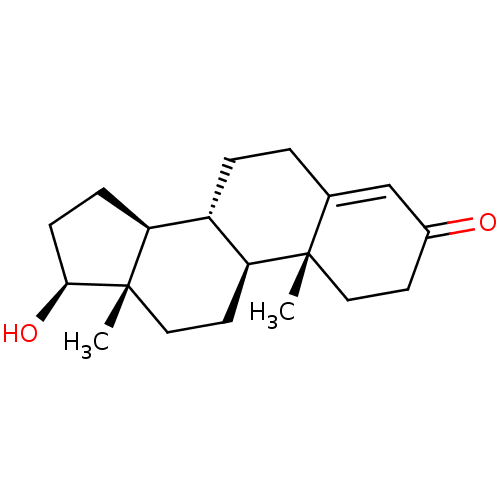
Substrate
Name:
BDBM8885
Synonyms:
(1S,2R,10R,11S,14S,15S)-14-hydroxy-2,15-dimethyltetracyclo[8.7.0.0^{2,7}.0^{11,15}]heptadec-6-en-5-one | 17beta-Hydroxyandrost-4-en-3-one | Testosterone | Testosterone, 1 | US9682960, Testosterone
Type:
Steroid
Emp. Form.:
C19H28O2
Mol. Mass.:
288.4244
SMILES:
[H][C@@]12CC[C@H](O)[C@@]1(C)CC[C@@]1([H])[C@@]2([H])CCC2=CC(=O)CC[C@]12C |r,t:18|