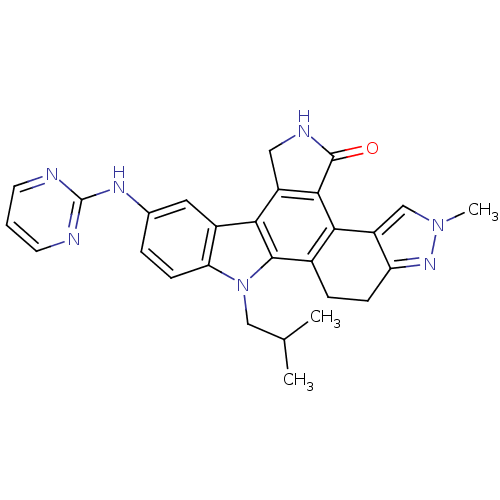
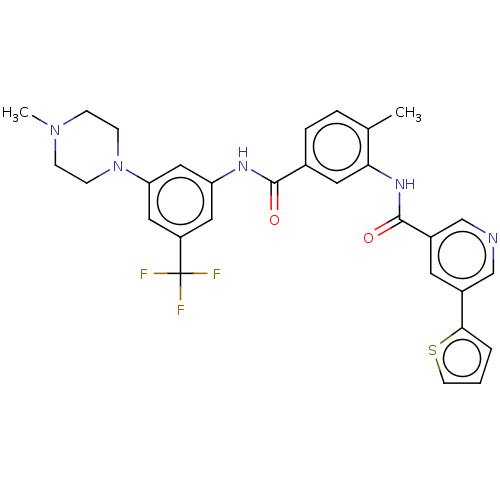
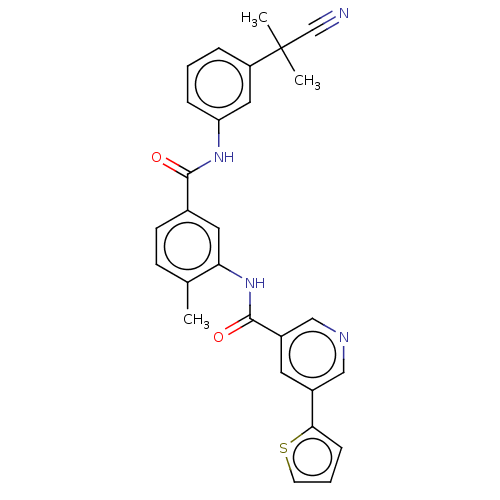
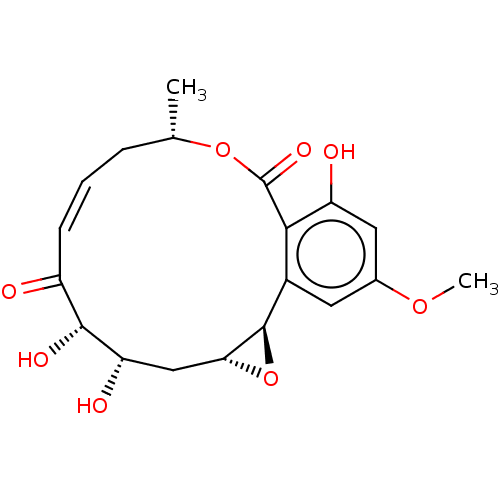
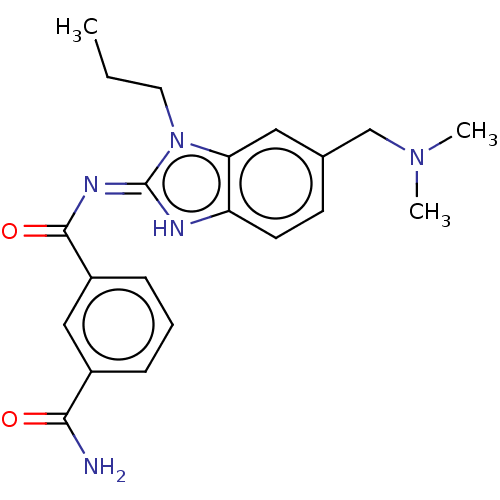
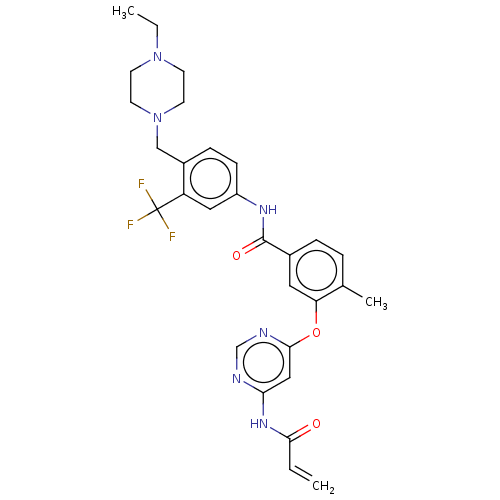
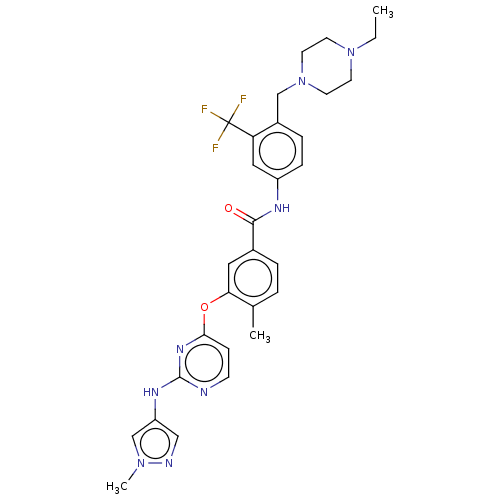
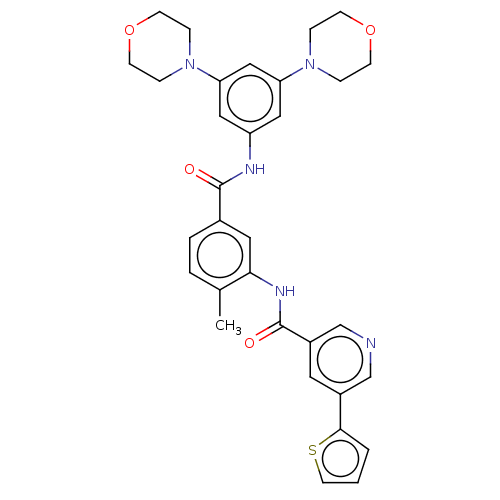
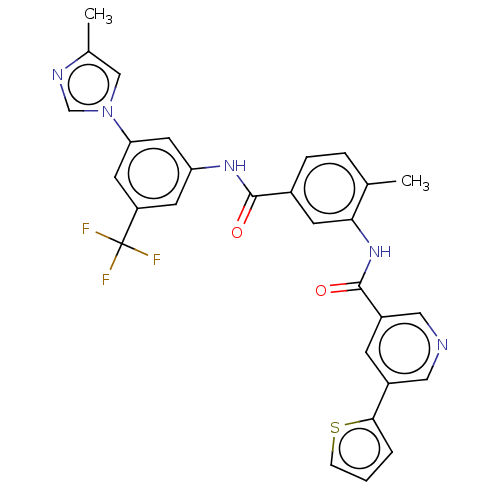
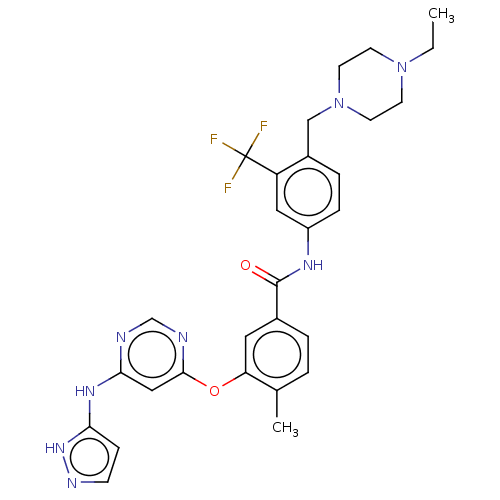
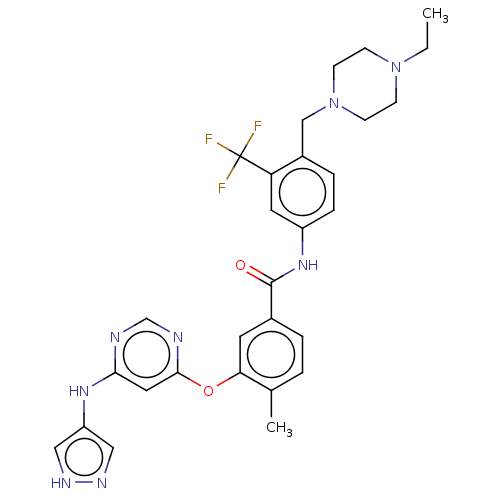
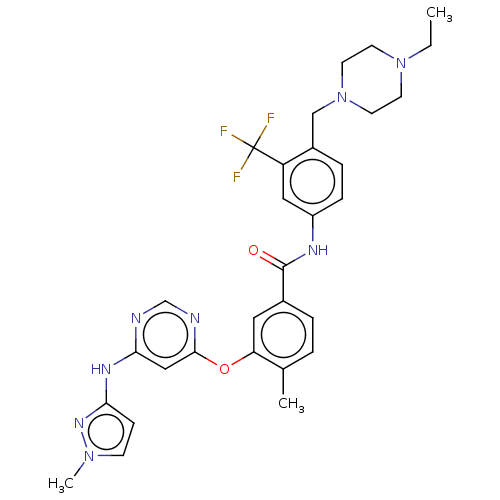
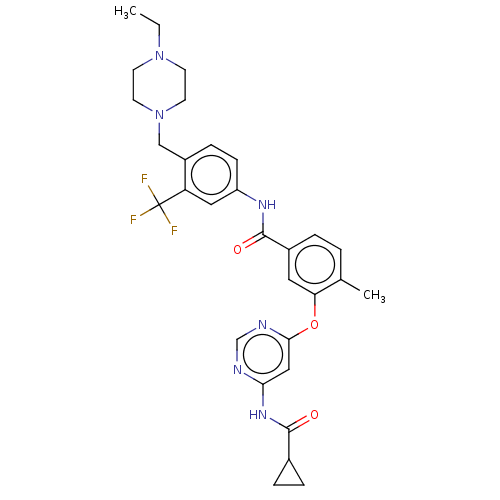
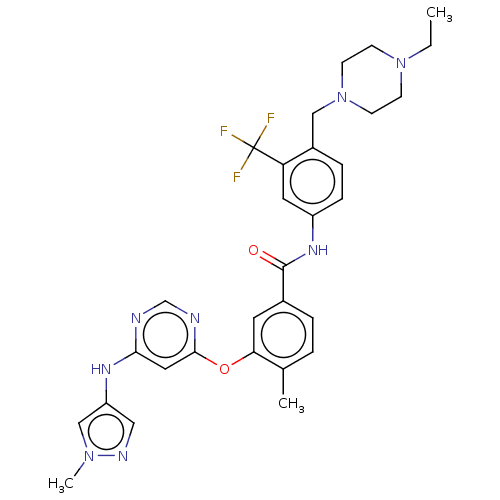
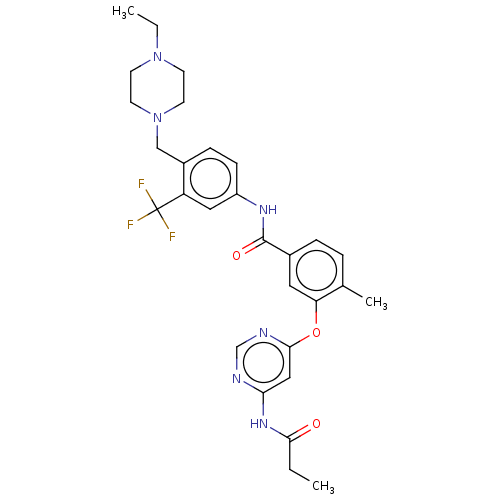
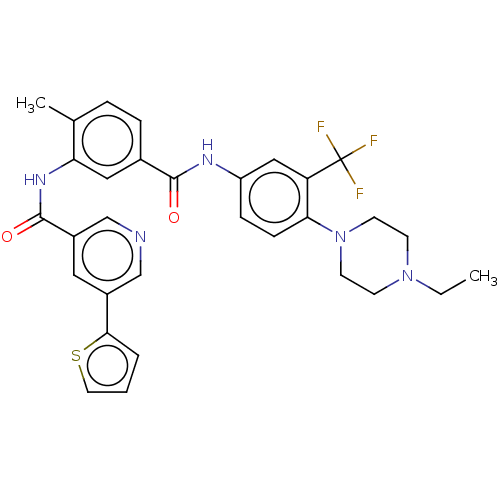
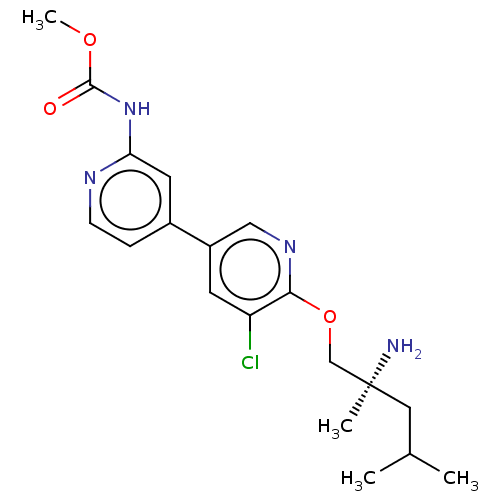
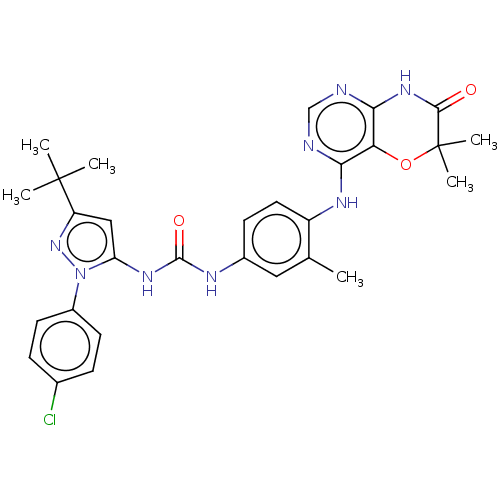
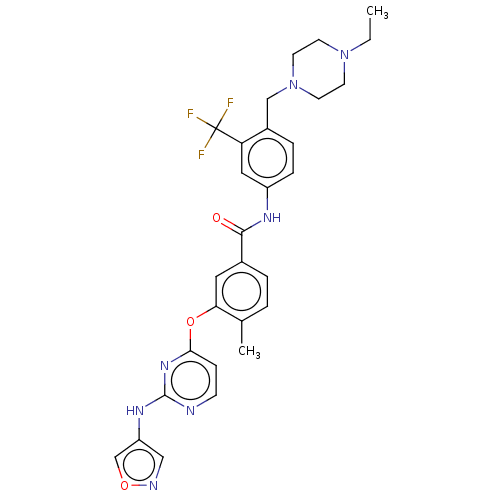
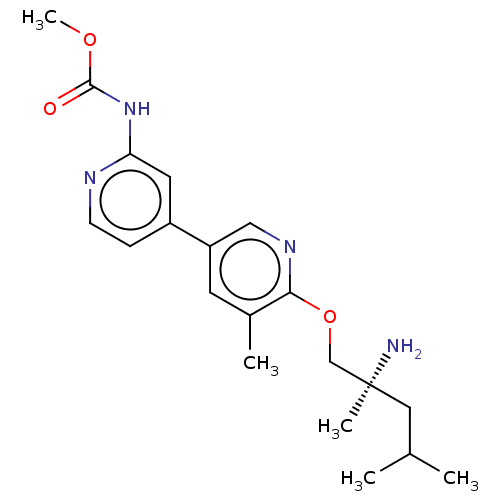
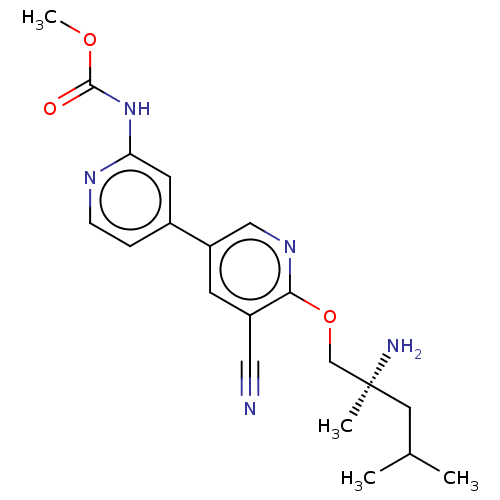
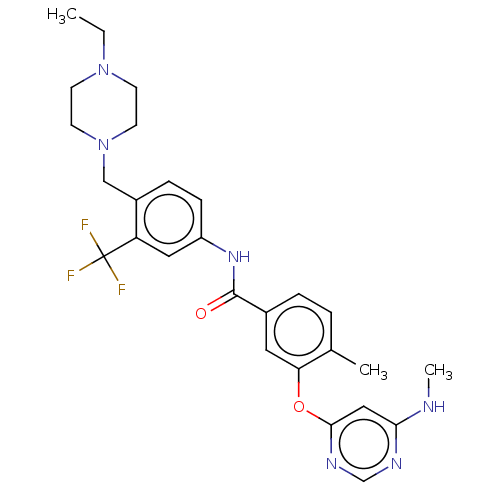
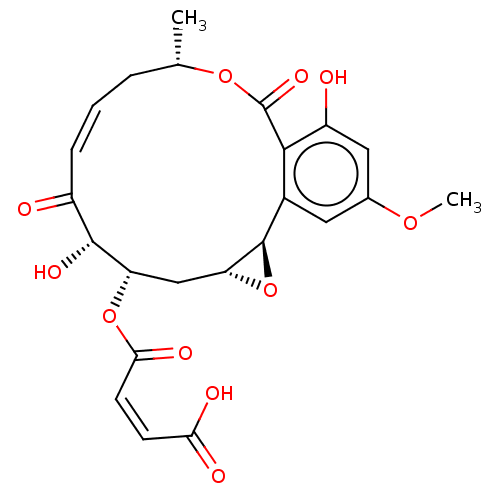
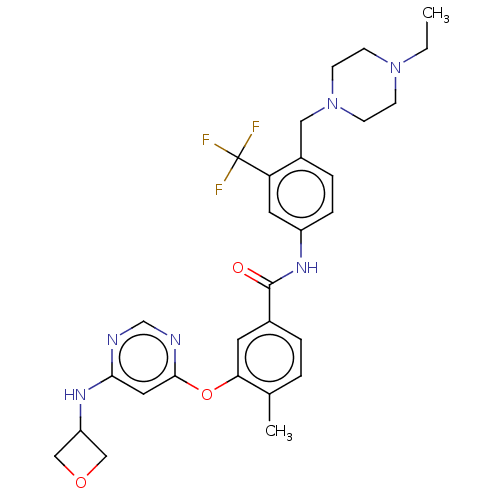
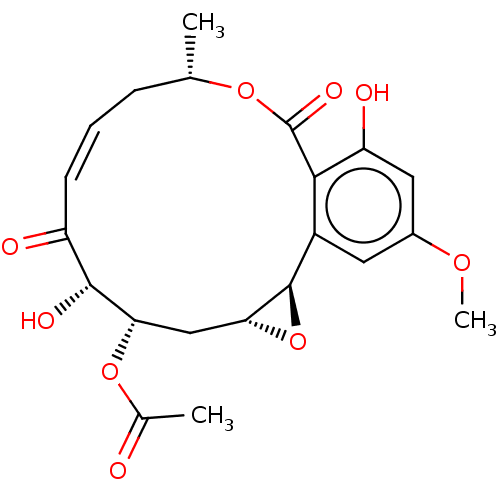
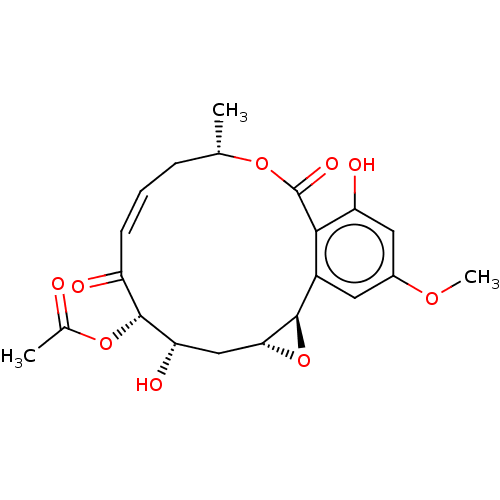
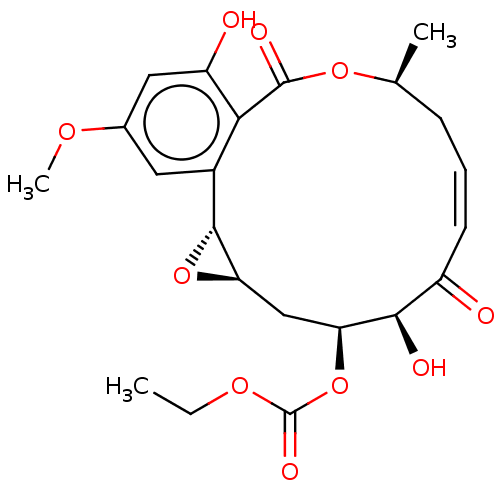
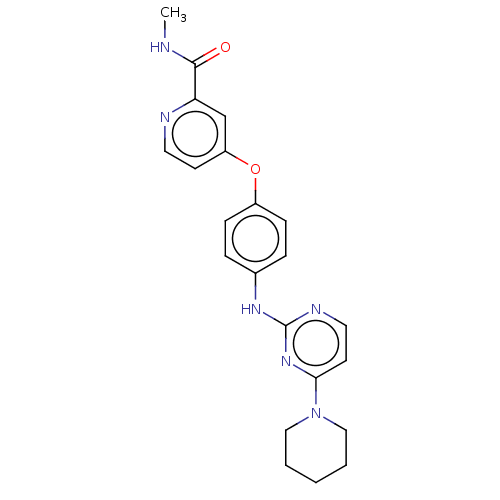
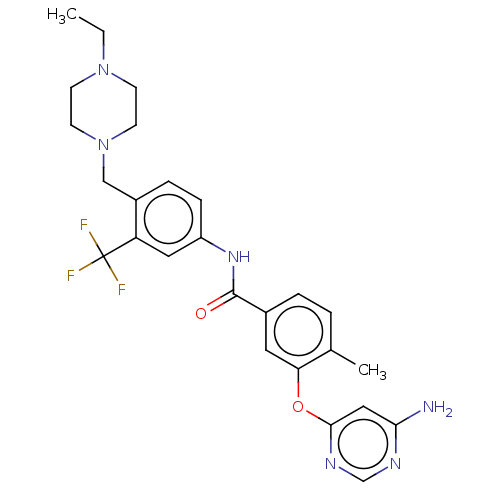
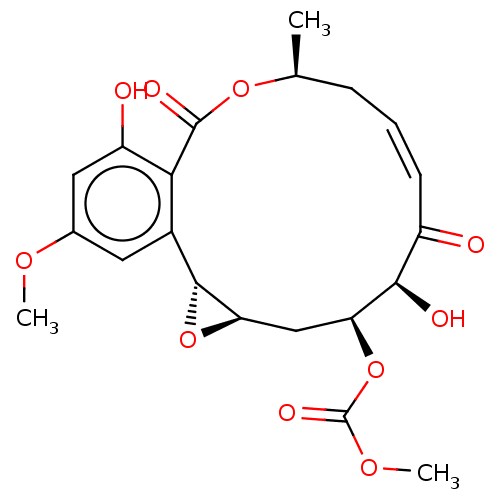
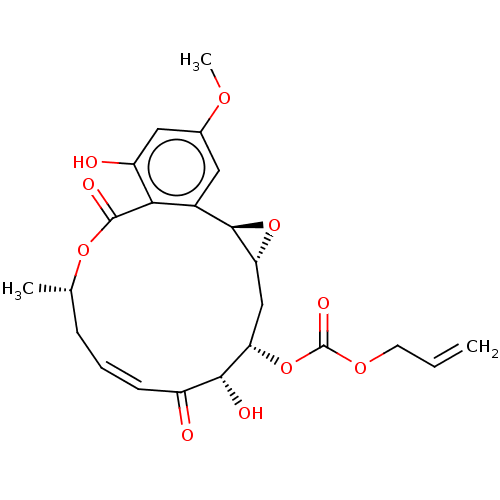
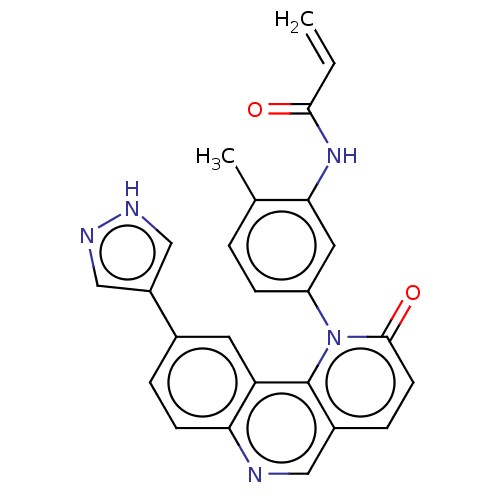
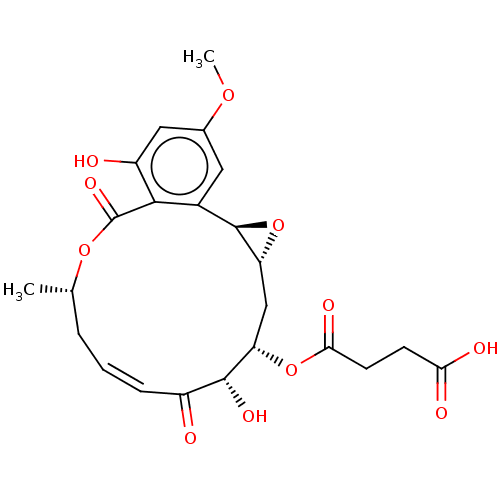
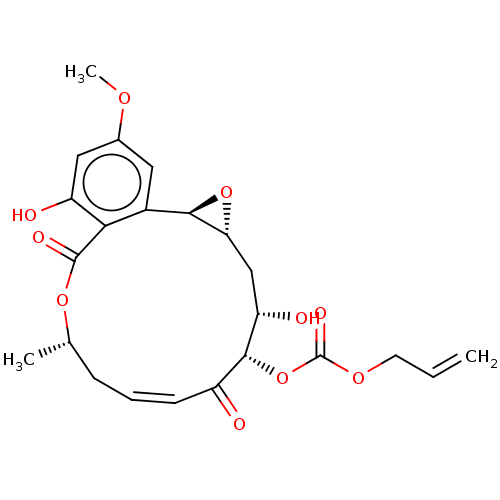
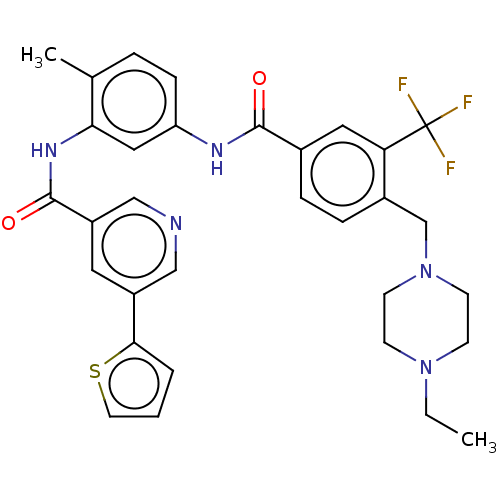
Affinity DataIC50: 1.60nMAssay Description:Accordingly, the present disclosure is directed to the use of these compounds in the preparation and execution of screening assays for compounds whic...More data for this Ligand-Target Pair
Affinity DataIC50: 1.70nMAssay Description:Accordingly, the present disclosure is directed to the use of these compounds in the preparation and execution of screening assays for compounds whic...More data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Inhibition of Tak1 (unknown origin) by competition binding assayMore data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Inhibition of Tak1 (unknown origin) in presence of ATPMore data for this Ligand-Target Pair
Affinity DataIC50: 2.40nMAssay Description:Accordingly, the present disclosure is directed to the use of these compounds in the preparation and execution of screening assays for compounds whic...More data for this Ligand-Target Pair
Affinity DataIC50: 2.43nMAssay Description:The in vitro activity of the compounds described herein in inhibiting TAK1, HCK, and other kinases were obtained using an Invitrogen Select Screening...More data for this Ligand-Target Pair
Affinity DataIC50: 2.5nMAssay Description:Accordingly, the present disclosure is directed to the use of these compounds in the preparation and execution of screening assays for compounds whic...More data for this Ligand-Target Pair
Affinity DataIC50: 2.60nMAssay Description:Accordingly, the present disclosure is directed to the use of these compounds in the preparation and execution of screening assays for compounds whic...More data for this Ligand-Target Pair
Affinity DataIC50: 2.80nMAssay Description:Accordingly, the present disclosure is directed to the use of these compounds in the preparation and execution of screening assays for compounds whic...More data for this Ligand-Target Pair
Affinity DataIC50: 3.10nMAssay Description:Accordingly, the present disclosure is directed to the use of these compounds in the preparation and execution of screening assays for compounds whic...More data for this Ligand-Target Pair
Affinity DataIC50: 5.33nMAssay Description:The in vitro activity of the compounds described herein in inhibiting TAK1, HCK, and other kinases were obtained using an Invitrogen Select Screening...More data for this Ligand-Target Pair
Affinity DataIC50: 7.40nMAssay Description:The in vitro activity of the compounds described herein in inhibiting TAK1, HCK, and other kinases were obtained using an Invitrogen Select Screening...More data for this Ligand-Target Pair
TargetNuclear receptor subfamily 2 group C member 2/TGF-beta-activated kinase 1 and MAP3K7-binding protein 1(Human)
China Pharmaceutical University
Curated by ChEMBL
China Pharmaceutical University
Curated by ChEMBL
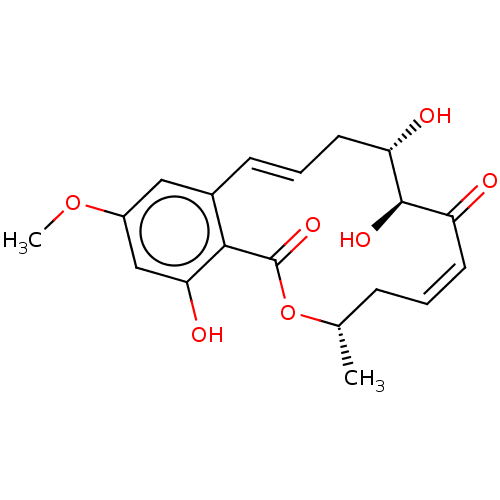
Affinity DataIC50: 9nMAssay Description:Inhibition of Tak1/Tab1 (unknown origin) assessed as inhibition of Tak1 kinase activity preincubated for 30 mins followed by addition of MBP protein ...More data for this Ligand-Target Pair
Affinity DataIC50: 9nMAssay Description:Accordingly, the present disclosure is directed to the use of these compounds in the preparation and execution of screening assays for compounds whic...More data for this Ligand-Target Pair
Affinity DataIC50: 14nMAssay Description:Inhibition of human TAK1 using ATP as substrateMore data for this Ligand-Target Pair
Affinity DataIC50: 22.8nMAssay Description:The in vitro activity of the compounds described herein in inhibiting TAK1, HCK, and other kinases were obtained using an Invitrogen Select Screening...More data for this Ligand-Target Pair
Affinity DataIC50: 29.9nMAssay Description:The in vitro activity of the compounds described herein in inhibiting TAK1, HCK, and other kinases were obtained using an Invitrogen Select Screening...More data for this Ligand-Target Pair
TargetNuclear receptor subfamily 2 group C member 2/TGF-beta-activated kinase 1 and MAP3K7-binding protein 1(Human)
China Pharmaceutical University
Curated by ChEMBL
China Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 30nMAssay Description:Inhibition of recombinant human N-terminal GST-tagged TAK1 (1 to 303 residues)/TAB1 (437 to end residues) expressed in baculovirus infected Sf9 insec...More data for this Ligand-Target Pair
Affinity DataIC50: 32nMAssay Description:Accordingly, the present disclosure is directed to the use of these compounds in the preparation and execution of screening assays for compounds whic...More data for this Ligand-Target Pair
Affinity DataIC50: 45nMAssay Description:The in vitro activity of the compounds described herein in inhibiting TAK1, HCK and other kinases were obtained using an Invitrogen Select Screening ...More data for this Ligand-Target Pair
Affinity DataIC50: 53.8nMAssay Description:The in vitro activity of the compounds described herein in inhibiting TAK1, HCK and other kinases were obtained using an Invitrogen Select Screening ...More data for this Ligand-Target Pair
Affinity DataIC50: 56.5nMAssay Description:The in vitro activity of the compounds described herein in inhibiting TAK1, HCK, and other kinases were obtained using an Invitrogen Select Screening...More data for this Ligand-Target Pair
Affinity DataIC50: 61nMAssay Description:Inhibition of human TAK1More data for this Ligand-Target Pair
Affinity DataIC50: 62.2nMAssay Description:The in vitro activity of the compounds described herein in inhibiting TAK1, HCK, and other kinases were obtained using an Invitrogen Select Screening...More data for this Ligand-Target Pair
Affinity DataIC50: 63.5nMAssay Description:The in vitro activity of the compounds described herein in inhibiting TAK1, HCK and other kinases were obtained using an Invitrogen Select Screening ...More data for this Ligand-Target Pair
Affinity DataIC50: 71.7nMAssay Description:The in vitro activity of the compounds described herein in inhibiting TAK1, HCK and other kinases were obtained using an Invitrogen Select Screening ...More data for this Ligand-Target Pair
Affinity DataIC50: 76.2nMAssay Description:The in vitro activity of the compounds described herein in inhibiting TAK1, HCK and other kinases were obtained using an Invitrogen Select Screening ...More data for this Ligand-Target Pair
Affinity DataIC50: 92nMAssay Description:The in vitro activity of the compounds described herein in inhibiting TAK1, HCK and other kinases were obtained using an Invitrogen Select Screening ...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:The in vitro activity of the compounds described herein in inhibiting TAK1, HCK and other kinases were obtained using an Invitrogen Select Screening ...More data for this Ligand-Target Pair
Affinity DataIC50: 100nMAssay Description:The in vitro activity of the compounds described herein in inhibiting TAK1, HCK and other kinases were obtained using an Invitrogen Select Screening ...More data for this Ligand-Target Pair
Affinity DataIC50: 117nMAssay Description:The in vitro activity of the compounds described herein in inhibiting TAK1, HCK, and other kinases were obtained using an Invitrogen Select Screening...More data for this Ligand-Target Pair
Affinity DataIC50: 120nMAssay Description:Inhibition of TAK (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 126nMAssay Description:Inhibition of human TAK1 in presence of ATP by radiometric assayMore data for this Ligand-Target Pair
Affinity DataIC50: 136nMAssay Description:The in vitro activity of the compounds described herein in inhibiting TAK1, HCK and other kinases were obtained using an Invitrogen Select Screening ...More data for this Ligand-Target Pair
TargetNuclear receptor subfamily 2 group C member 2/TGF-beta-activated kinase 1 and MAP3K7-binding protein 1(Human)
China Pharmaceutical University
Curated by ChEMBL
China Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 180nMAssay Description:Inhibition of TAK/TAB (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 260nMAssay Description:Inhibition of TAK (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 364nMAssay Description:The in vitro activity of the compounds described herein in inhibiting TAK1, HCK and other kinases were obtained using an Invitrogen Select Screening ...More data for this Ligand-Target Pair
TargetNuclear receptor subfamily 2 group C member 2/TGF-beta-activated kinase 1 and MAP3K7-binding protein 1(Human)
China Pharmaceutical University
Curated by ChEMBL
China Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 380nMAssay Description:Inhibition of recombinant human N-terminal GST-tagged TAK1 (1 to 303 residues)/TAB1 (437 to end residues) expressed in baculovirus infected Sf9 insec...More data for this Ligand-Target Pair
Affinity DataIC50: 400nMAssay Description:The in vitro activity of the compounds described herein in inhibiting TAK1, HCK and other kinases were obtained using an Invitrogen Select Screening ...More data for this Ligand-Target Pair
TargetNuclear receptor subfamily 2 group C member 2/TGF-beta-activated kinase 1 and MAP3K7-binding protein 1(Human)
China Pharmaceutical University
Curated by ChEMBL
China Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 430nMAssay Description:Inhibition of recombinant human N-terminal GST-tagged TAK1 (1 to 303 residues)/TAB1 (437 to end residues) expressed in baculovirus infected Sf9 insec...More data for this Ligand-Target Pair
TargetNuclear receptor subfamily 2 group C member 2/TGF-beta-activated kinase 1 and MAP3K7-binding protein 1(Human)
China Pharmaceutical University
Curated by ChEMBL
China Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 450nMAssay Description:Inhibition of recombinant human N-terminal GST-tagged TAK1 (1 to 303 residues)/TAB1 (437 to end residues) expressed in baculovirus infected Sf9 insec...More data for this Ligand-Target Pair
Affinity DataIC50: 487nMAssay Description:The in vitro activity of the compounds described herein in inhibiting TAK1, HCK and other kinases were obtained using an Invitrogen Select Screening ...More data for this Ligand-Target Pair
TargetNuclear receptor subfamily 2 group C member 2/TGF-beta-activated kinase 1 and MAP3K7-binding protein 1(Human)
China Pharmaceutical University
Curated by ChEMBL
China Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 500nMAssay Description:Inhibition of recombinant human N-terminal GST-tagged TAK1 (1 to 303 residues)/TAB1 (437 to end residues) expressed in baculovirus infected Sf9 insec...More data for this Ligand-Target Pair
TargetNuclear receptor subfamily 2 group C member 2/TGF-beta-activated kinase 1 and MAP3K7-binding protein 1(Human)
China Pharmaceutical University
Curated by ChEMBL
China Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 580nMAssay Description:Inhibition of Tak1/Tab1 (unknown origin) assessed as inhibition of Tak1 kinase activity preincubated for 30 mins followed by addition of MBP protein ...More data for this Ligand-Target Pair
Affinity DataIC50: 591nMAssay Description:The in vitro activity of the compounds described herein in inhibiting TAK1, HCK and other kinases were obtained using an Invitrogen Select Screening ...More data for this Ligand-Target Pair
TargetNuclear receptor subfamily 2 group C member 2/TGF-beta-activated kinase 1 and MAP3K7-binding protein 1(Human)
China Pharmaceutical University
Curated by ChEMBL
China Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 630nMAssay Description:Inhibition of recombinant human N-terminal GST-tagged TAK1 (1 to 303 residues)/TAB1 (437 to end residues) expressed in baculovirus infected Sf9 insec...More data for this Ligand-Target Pair
TargetNuclear receptor subfamily 2 group C member 2/TGF-beta-activated kinase 1 and MAP3K7-binding protein 1(Human)
China Pharmaceutical University
Curated by ChEMBL
China Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 700nMAssay Description:Inhibition of recombinant human N-terminal GST-tagged TAK1 (1 to 303 residues)/TAB1 (437 to end residues) expressed in baculovirus infected Sf9 insec...More data for this Ligand-Target Pair
Affinity DataIC50: 717nMAssay Description:DiscoverX binding assays were performed according to published methods (Fabian et al., Nat. Biotechnol. 23, 329-36 (2005); Davis et al., Nat. Biotech...More data for this Ligand-Target Pair
TargetNuclear receptor subfamily 2 group C member 2/TGF-beta-activated kinase 1 and MAP3K7-binding protein 1(Human)
China Pharmaceutical University
Curated by ChEMBL
China Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 720nMAssay Description:Inhibition of recombinant human N-terminal GST-tagged TAK1 (1 to 303 residues)/TAB1 (437 to end residues) expressed in baculovirus infected Sf9 insec...More data for this Ligand-Target Pair
TargetNuclear receptor subfamily 2 group C member 2/TGF-beta-activated kinase 1 and MAP3K7-binding protein 1(Human)
China Pharmaceutical University
Curated by ChEMBL
China Pharmaceutical University
Curated by ChEMBL
Affinity DataIC50: 820nMAssay Description:Inhibition of recombinant human N-terminal GST-tagged TAK1 (1 to 303 residues)/TAB1 (437 to end residues) expressed in baculovirus infected Sf9 insec...More data for this Ligand-Target Pair
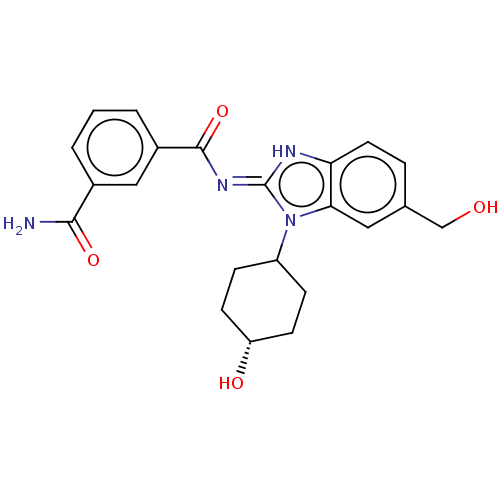
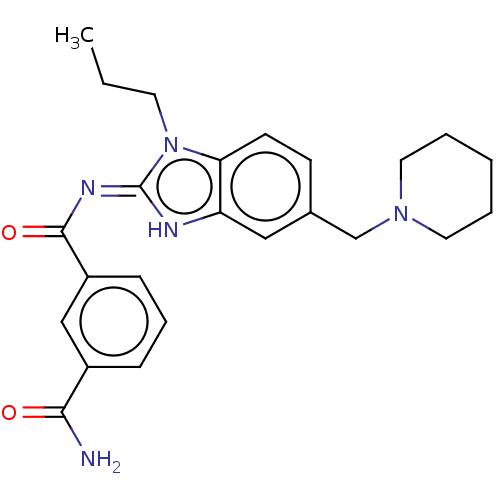
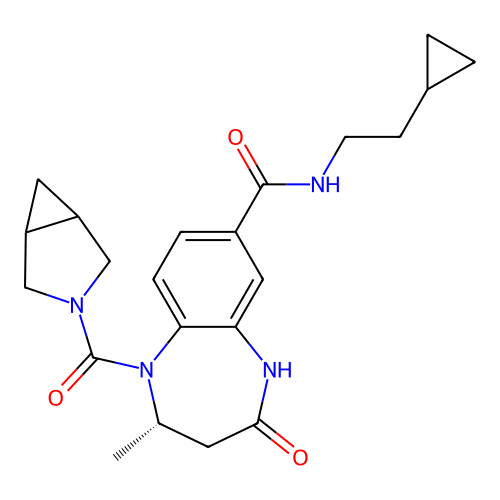
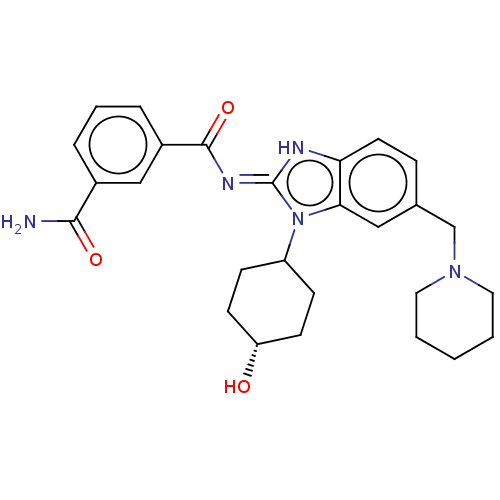
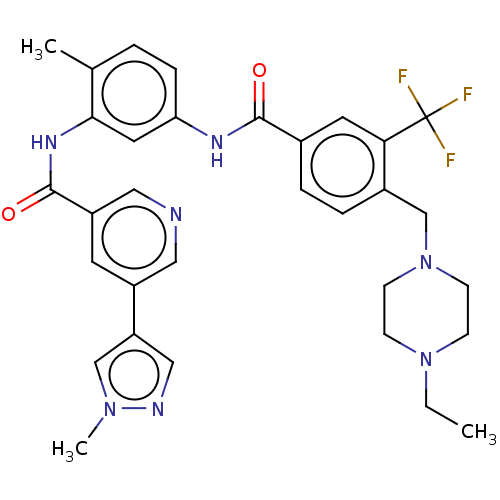
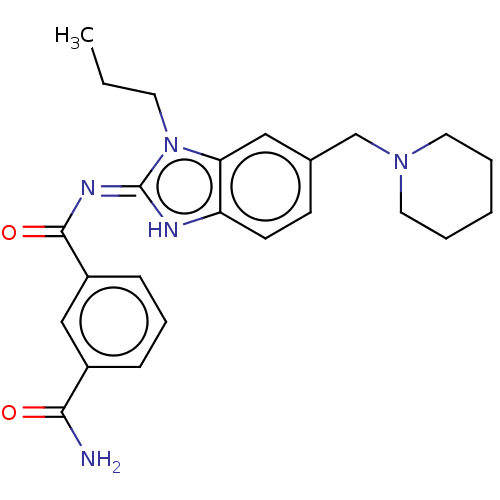
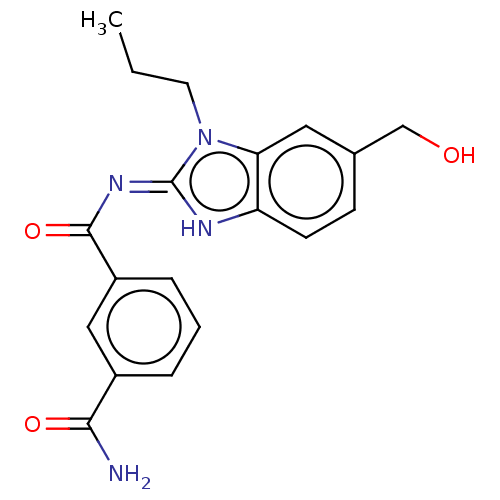
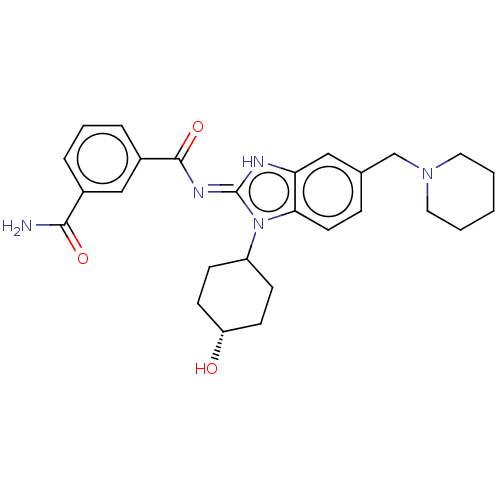
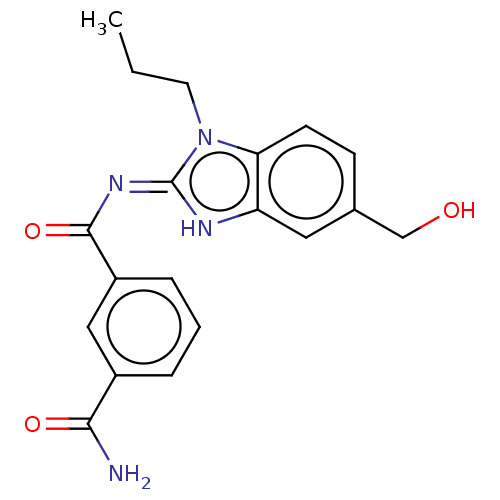
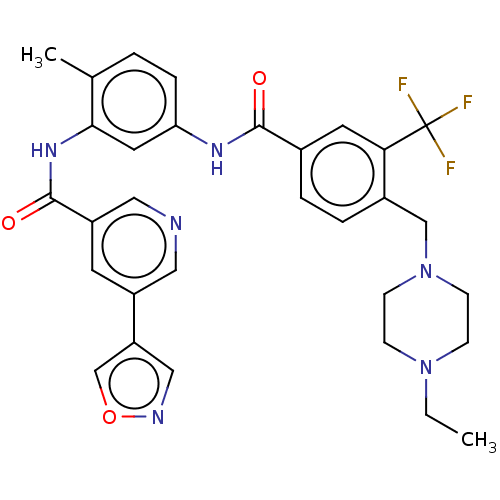
 3D Structure (crystal)
3D Structure (crystal)