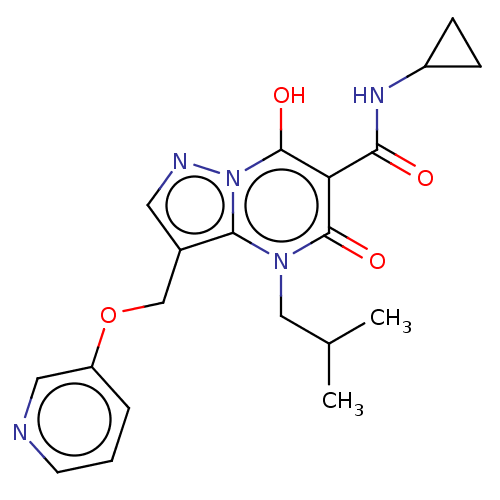
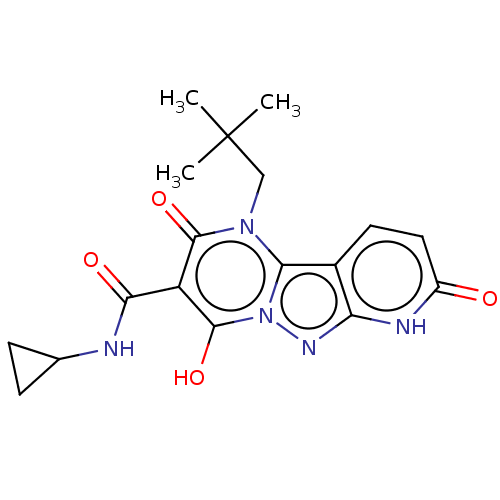
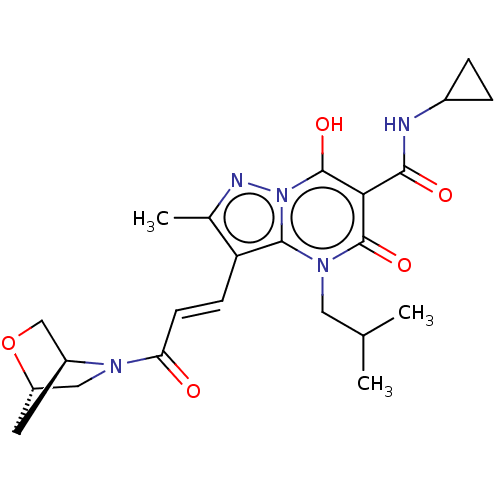
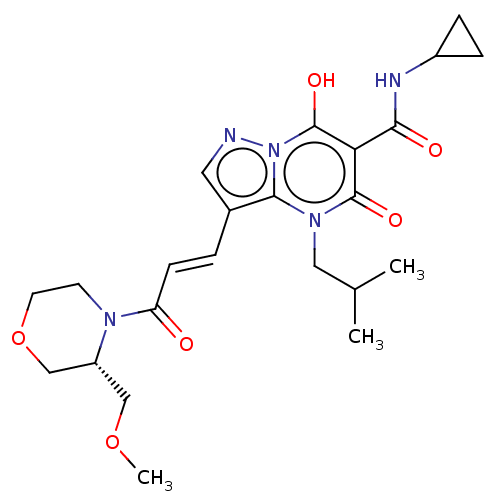
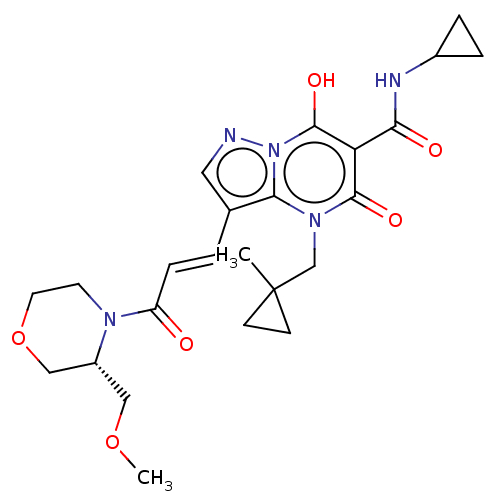
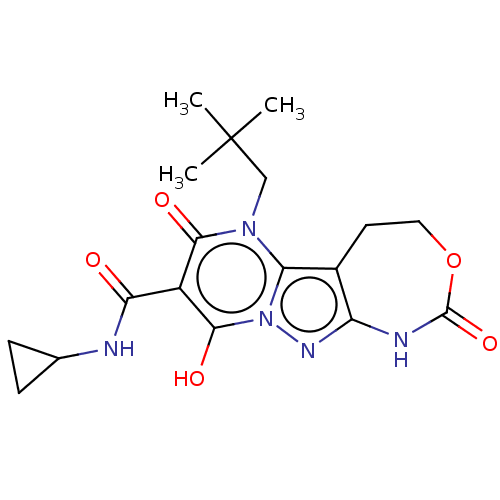
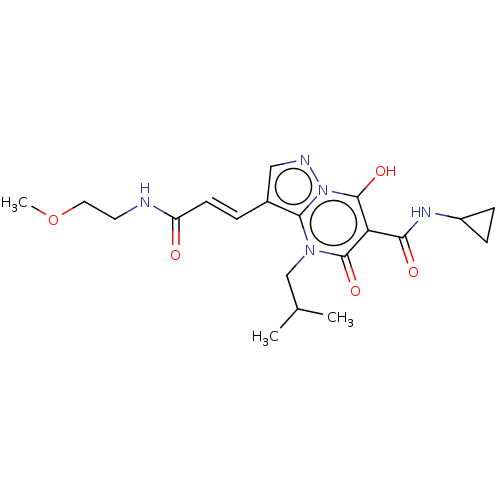
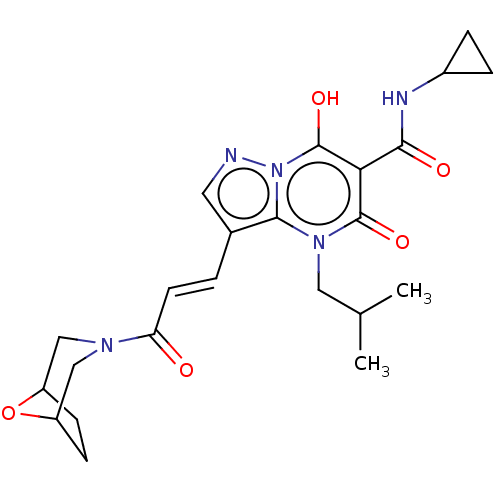
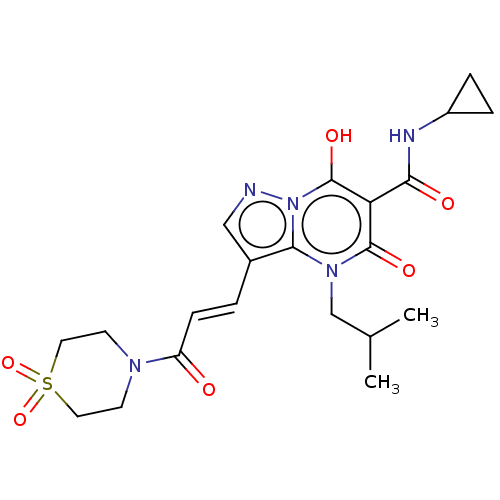
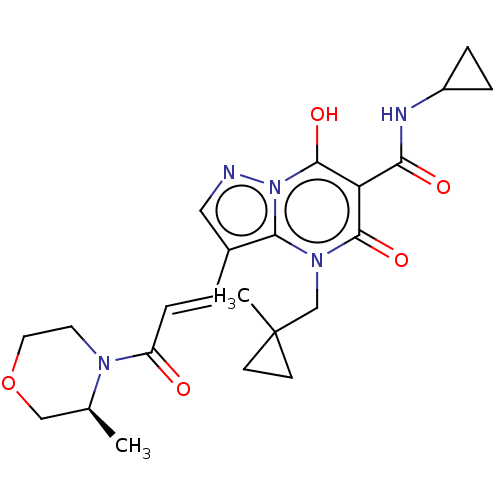
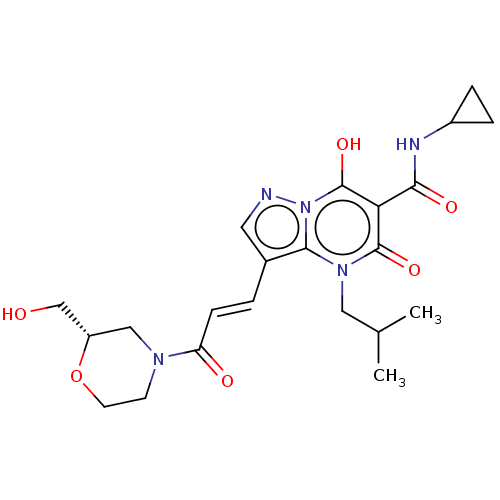
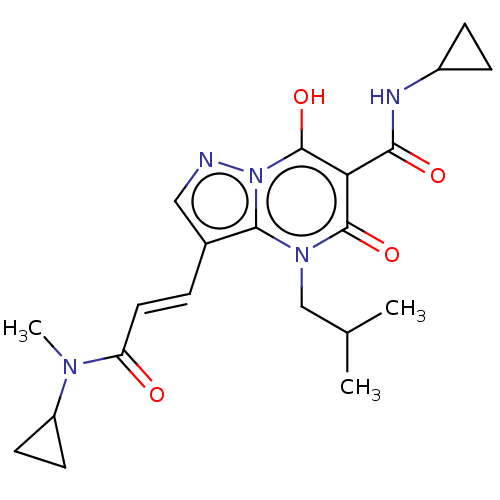
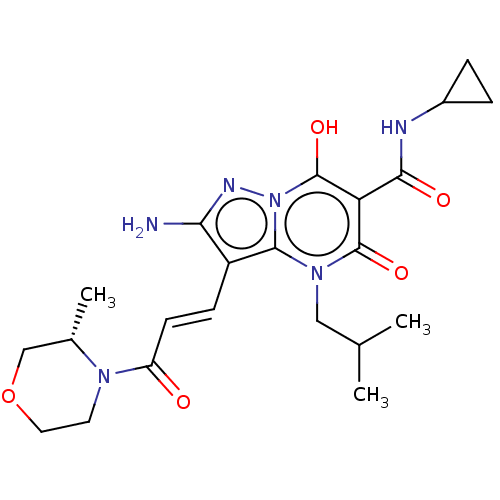
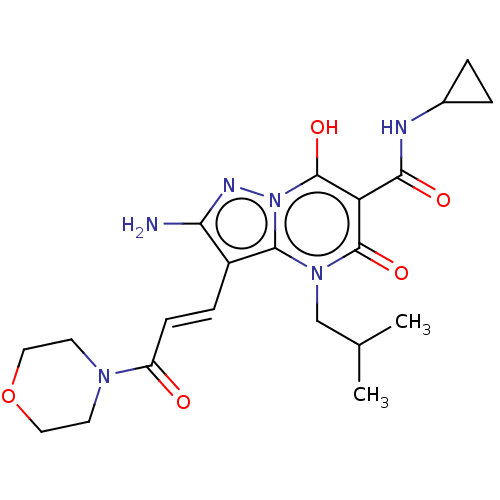
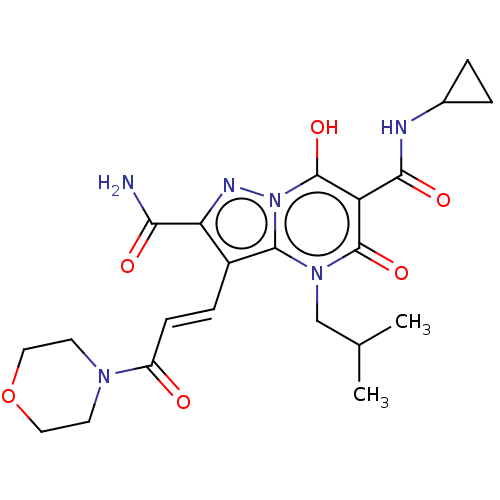
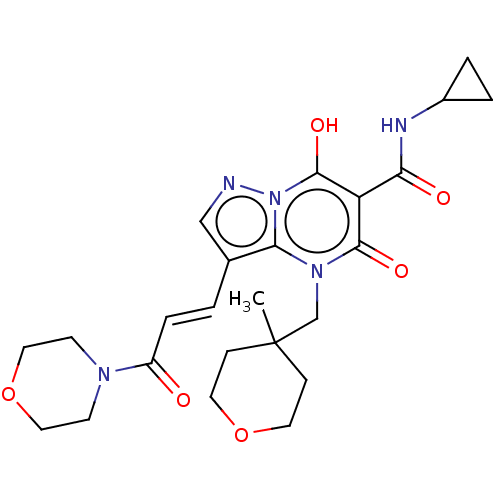
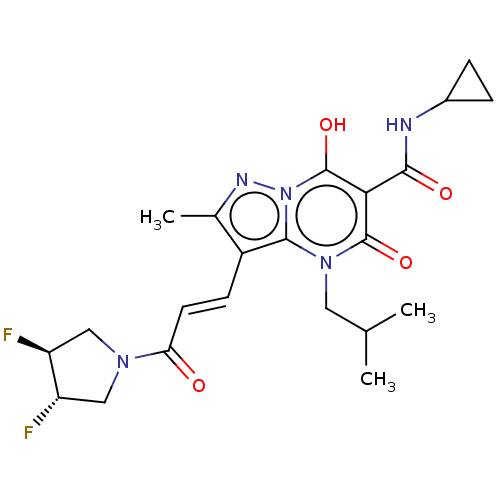
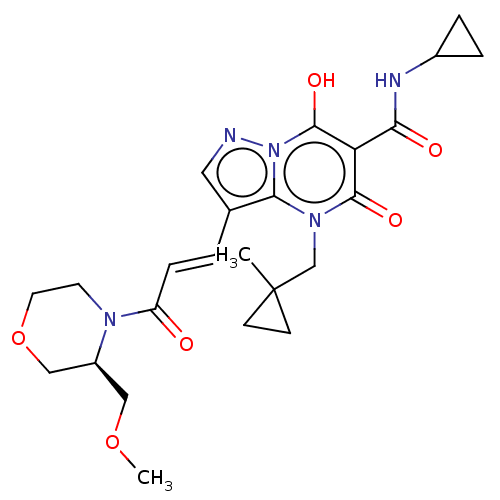
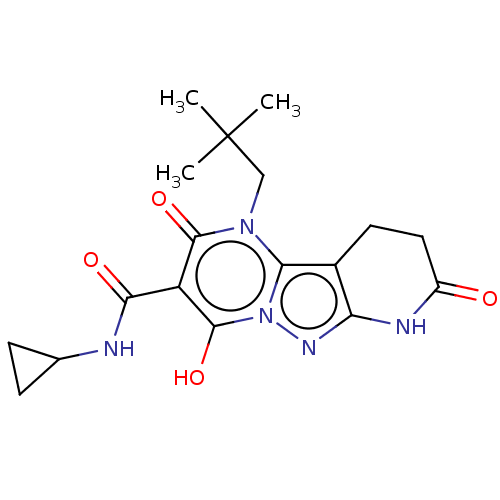
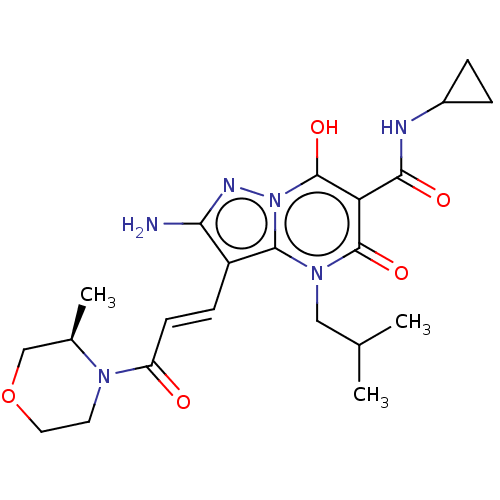
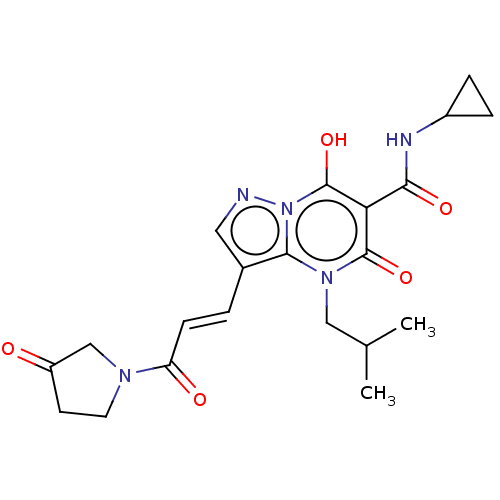
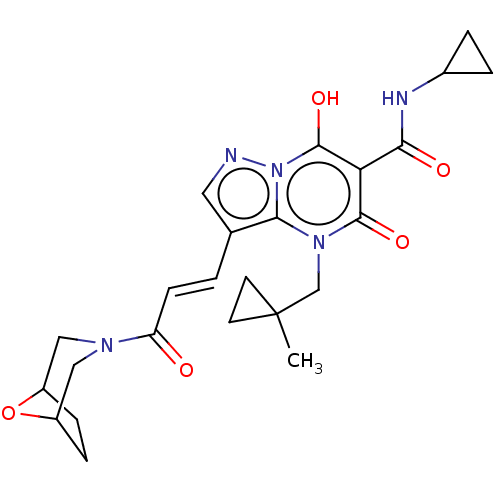
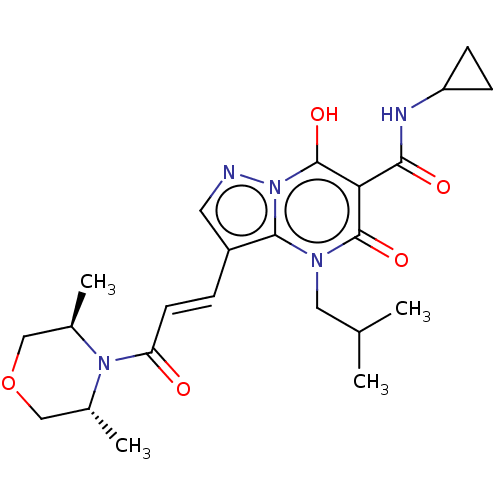
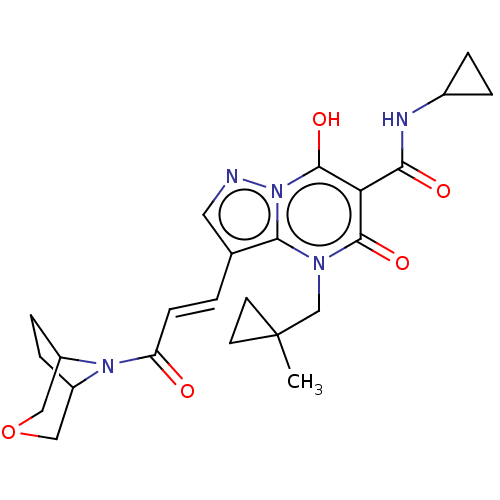
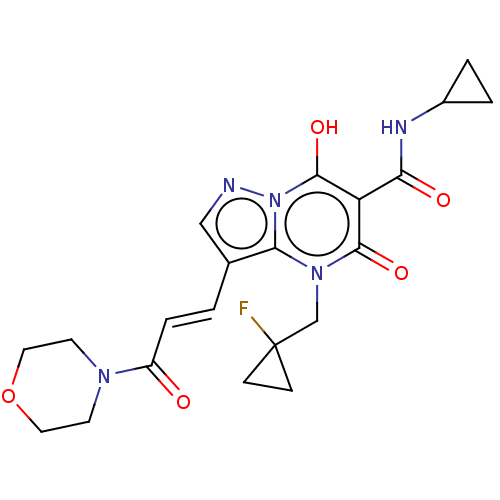
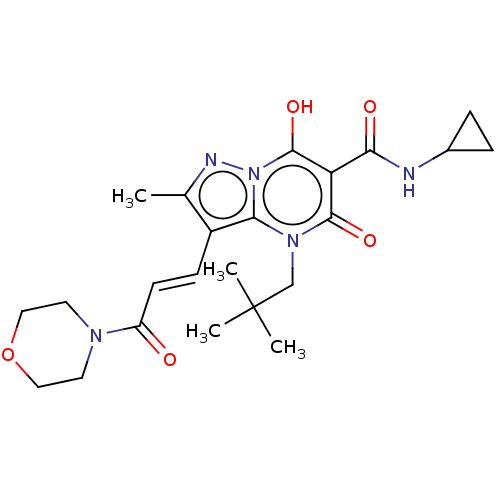
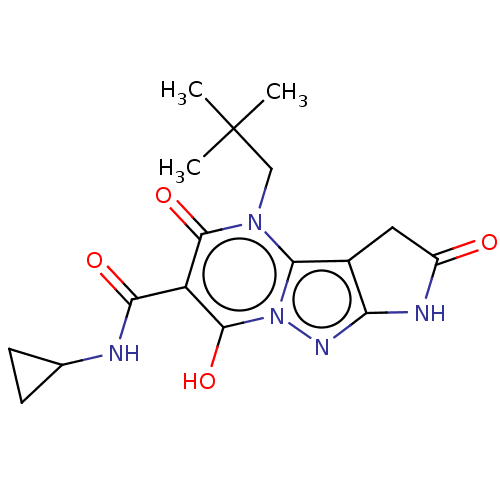
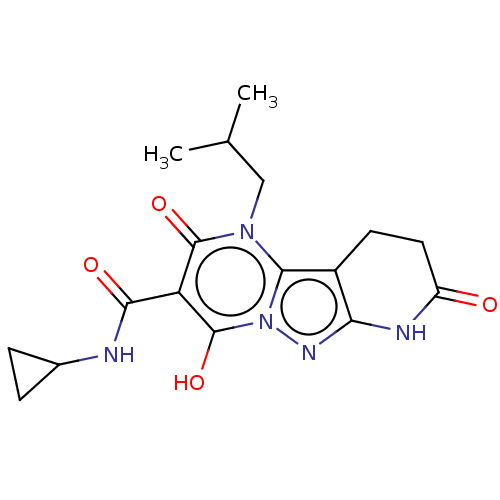
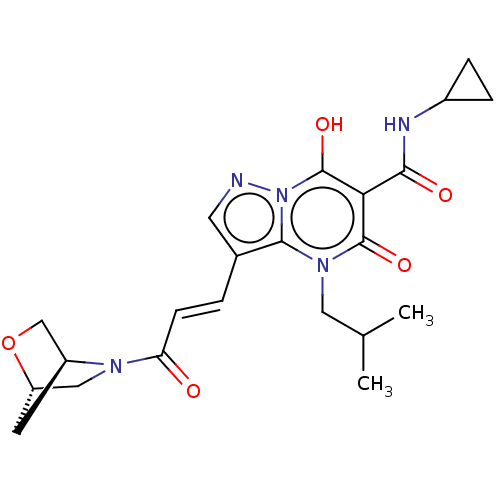
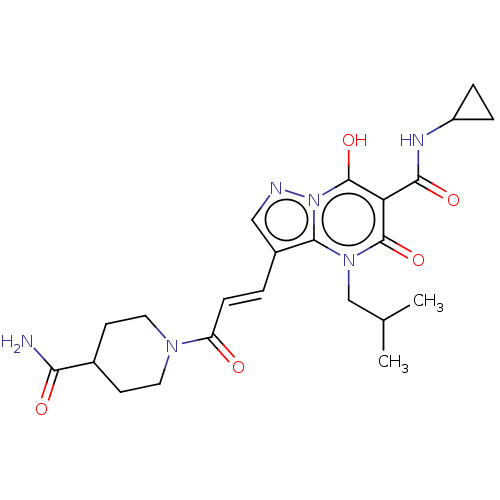
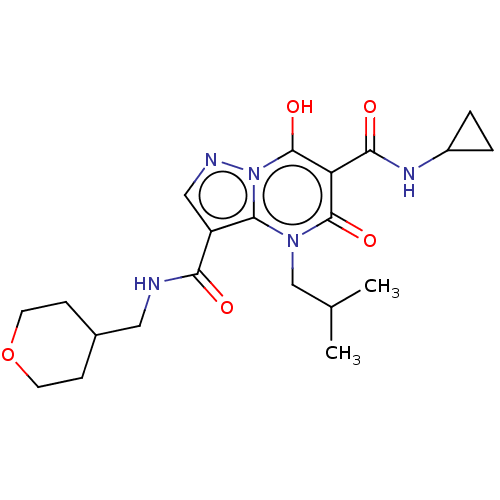
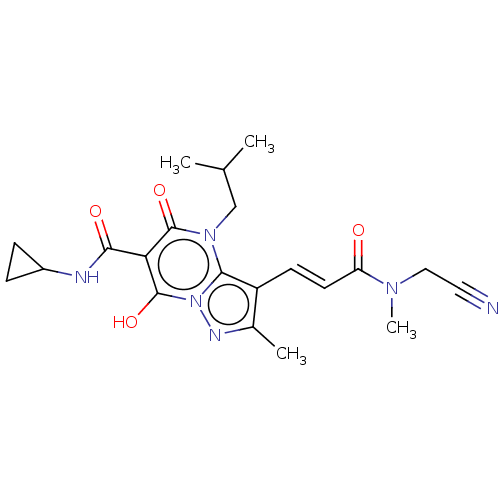
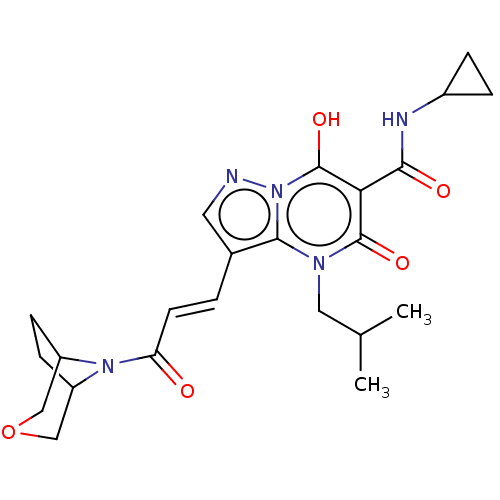
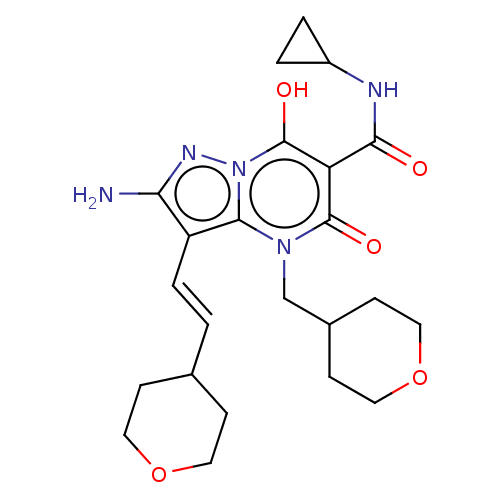
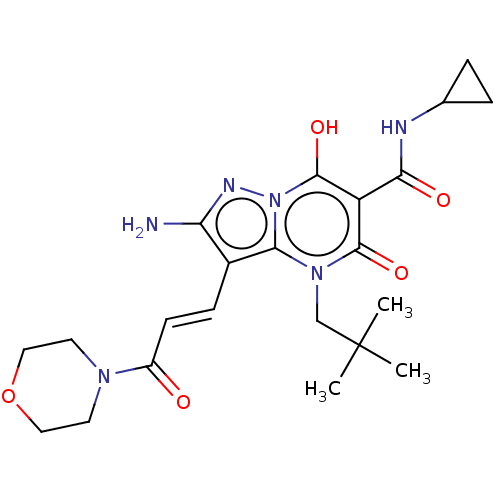
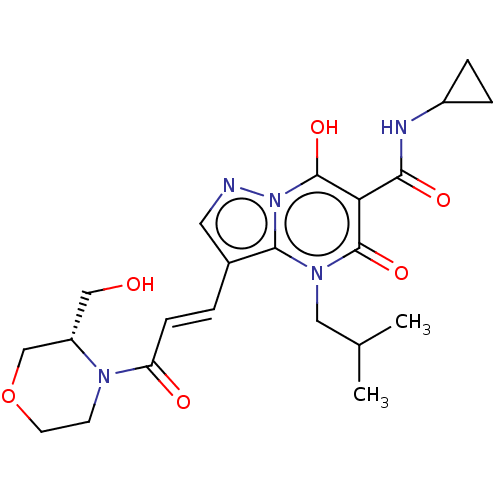
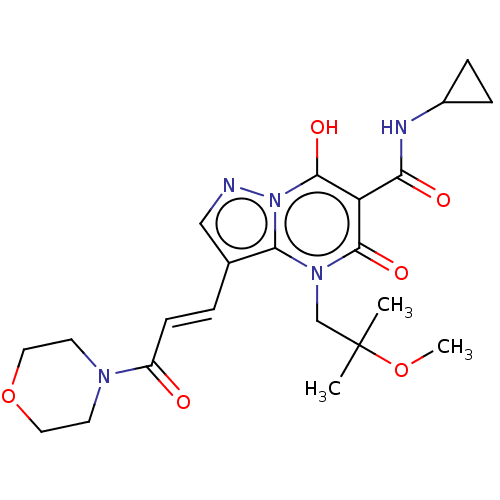
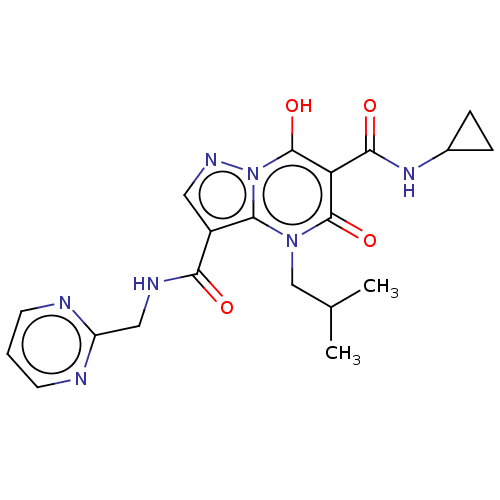
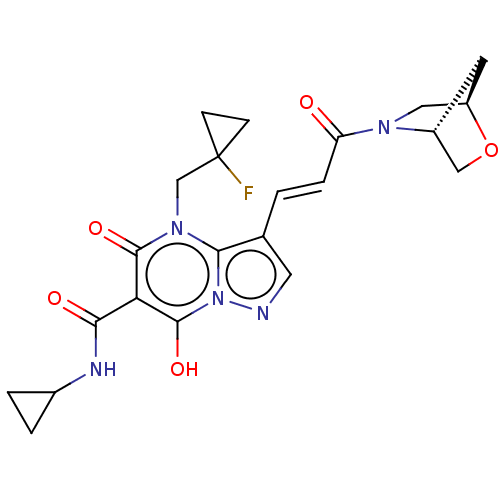
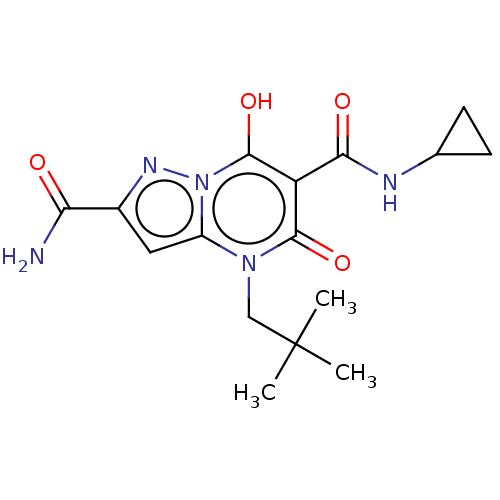
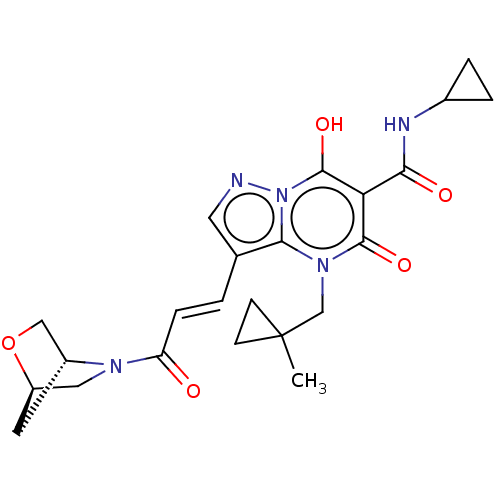
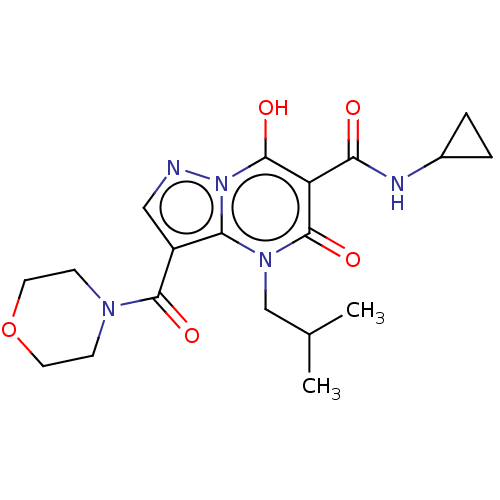
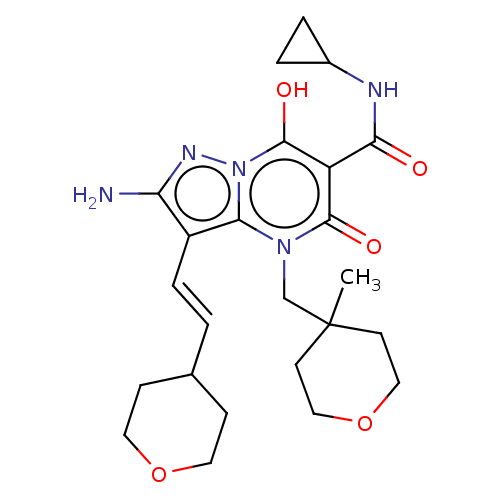
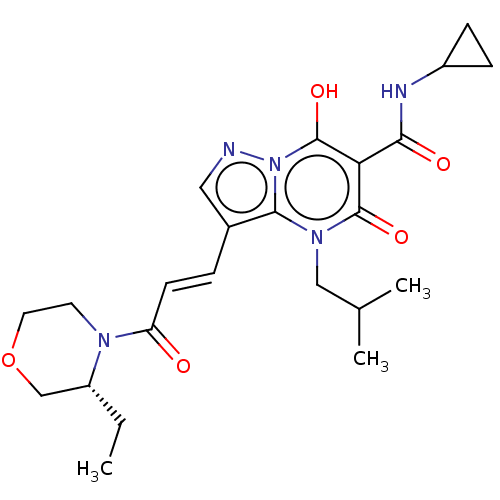
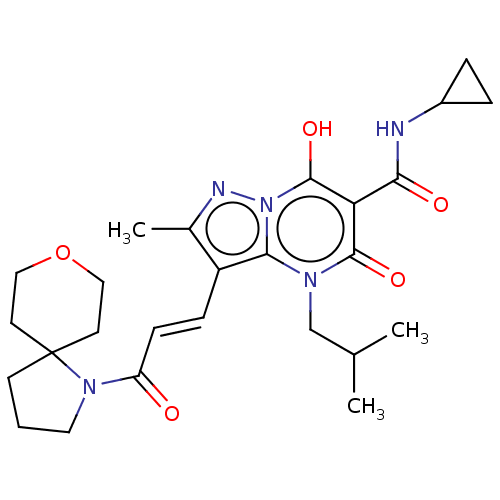
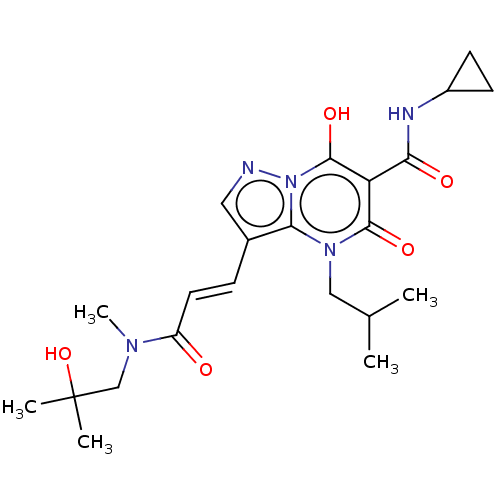
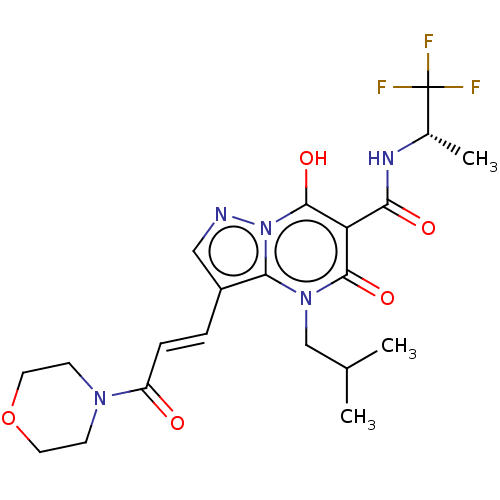
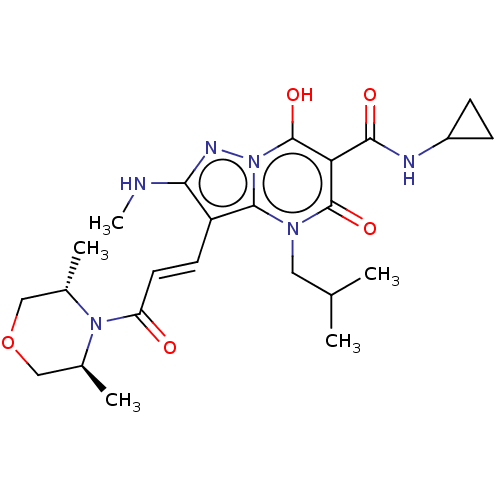
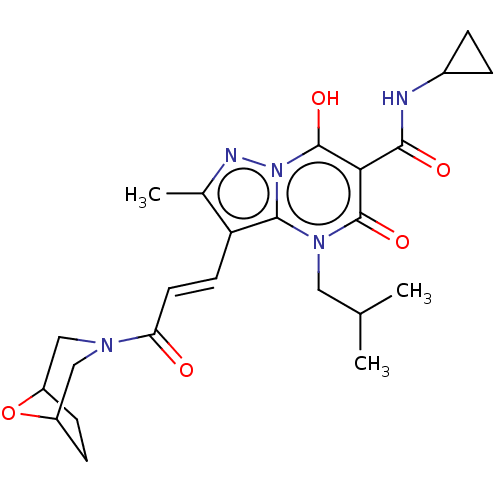
Affinity DataIC50: 0.180nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.390nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.480nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.540nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.550nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.550nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.560nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.570nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.580nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.580nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.580nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.590nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.590nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.590nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.590nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.600nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.600nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.610nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.610nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.610nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.610nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.610nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.610nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.620nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.620nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.630nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.630nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.630nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.630nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.640nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.640nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.640nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.640nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.640nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.650nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.650nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.650nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.650nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.650nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.650nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.650nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.650nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.650nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.660nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.660nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.660nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.660nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.660nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.660nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair
Affinity DataIC50: 0.670nMAssay Description:Human SPR inhibitory activity was measured by using a 384-well low adsorption clear plate (Greiner) with buffer D containing 100 mM Tris-HCl (pH 7.5)...More data for this Ligand-Target Pair