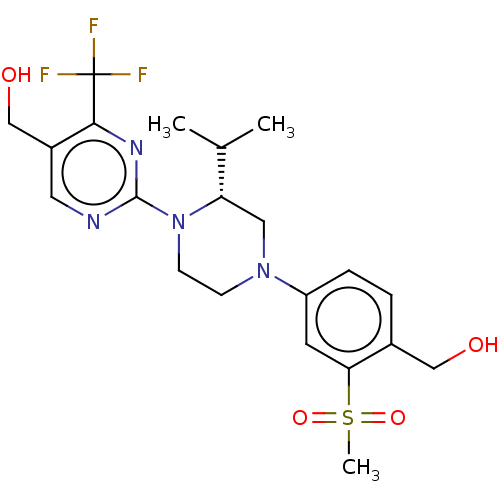
BDBM50177015 CHEMBL3814206::US10144715, Compound 19-1
SMILES CC(C)[C@@H]1CN(CCN1c2ncc(c(n2)C(F)(F)F)CO)c3ccc(c(c3)S(=O)(=O)C)CO
InChI Key InChIKey=FPVIRRAMNBCEDK-UHFFFAOYSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 25 hits for monomerid = 50177015
Found 25 hits for monomerid = 50177015
Affinity DataKi: 1.30nMAssay Description:Binding affinity to recombinant human LXRbeta-LBD expressed in Escherichia coli BL21 (DE3) assessed as inhibitory constant incubated for 30 mins by f...More data for this Ligand-Target Pair
Affinity DataKi: 3nMAssay Description:Compounds of the invention were assessed in a competition binding assay where different concentrations of compounds were incubated with the LXR ligan...More data for this Ligand-Target Pair
Affinity DataKi: 3nMAssay Description:Displacement of [3H]TO901317 from LXRbeta ligand binding domain (unknown origin) after 30 mins by liquid scintillation countingMore data for this Ligand-Target Pair
Affinity DataKi: 3nMAssay Description:Displacement of radiolabeled T0901317 from LXRbeta LBD (unknown origin)More data for this Ligand-Target Pair
Affinity DataEC50: 4nMAssay Description:Agonist activity at LXR in human THP1 cells assessed as upregulation of ABCA1 gene expression after 24 hrs by RT-PCR analysisMore data for this Ligand-Target Pair
Affinity DataEC50: 6nMAssay Description:Agonist activity at LXR in human HepG2 cells assessed as upregulation of SREBP1C gene expression after 24 hrs by RT-PCR analysisMore data for this Ligand-Target Pair
Affinity DataEC50: 21nMAssay Description:Agonist activity at LXRbeta LBD (unknown origin) fused with Gal4-DNA binding domain expressed in HEK293 cells assessed as modulation of receptor tran...More data for this Ligand-Target Pair
Affinity DataEC50: 21nMAssay Description:Agonist activity at human LXRbeta ligand binding domain(155 to 460 residues) transfected in HEK293 cells after 16 hrs by Gal4-luciferase reporter gen...More data for this Ligand-Target Pair
Affinity DataKi: 81nMAssay Description:Compounds of the invention were assessed in a competition binding assay where different concentrations of compounds were incubated with the LXR ligan...More data for this Ligand-Target Pair
Affinity DataKi: 81nMAssay Description:Displacement of [3H]TO901317 from LXRalpha ligand binding domain (unknown origin) after 30 mins by liquid scintillation countingMore data for this Ligand-Target Pair
Affinity DataKi: 81nMAssay Description:Displacement of radiolabeled T0901317 from LXRalpha LBD (unknown origin)More data for this Ligand-Target Pair
Affinity DataEC50: 244nMAssay Description:Agonist activity at human LXRalpha ligand binding domain(167 to 447 residues) transfected in HEK293 cells after 16 hrs by Gal4-luciferase reporter ge...More data for this Ligand-Target Pair
Affinity DataEC50: 244nMAssay Description:Agonist activity at LXRalpha LBD (unknown origin) fused with Gal4-DNA binding domain expressed in HEK293 cells assessed as modulation of receptor tra...More data for this Ligand-Target Pair
Affinity DataIC50: 608nMAssay Description:Inhibition of CYP2C9 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 610nMAssay Description:Inhibition of recombinant CYP2C9 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 2.20E+3nMAssay Description:Inhibition of CYP2C9 in human liver microsomesMore data for this Ligand-Target Pair
Affinity DataIC50: 6.60E+3nMAssay Description:Inhibition of CYP3A4 in human liver microsomesMore data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of RORalpha receptor (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of PXR (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of Mineralocorticoid receptor (unknown origin)More data for this Ligand-Target Pair
TargetRetinoic acid receptor RXR-alpha/RXR-beta/RXR-gamma(Human)
Vitae Pharmaceuticals
Curated by ChEMBL
Vitae Pharmaceuticals
Curated by ChEMBL
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of RXR (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 2.94E+4nMAssay Description:Inhibition of recombinant CYP3A4 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 2.95E+4nMAssay Description:Inhibition of CYP3A4 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+4nMAssay Description:Inhibition of recombinant CYP2D6 (unknown origin)More data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+4nMAssay Description:Inhibition of CYP2D6 (unknown origin)More data for this Ligand-Target Pair
 3D Structure (crystal)
3D Structure (crystal)