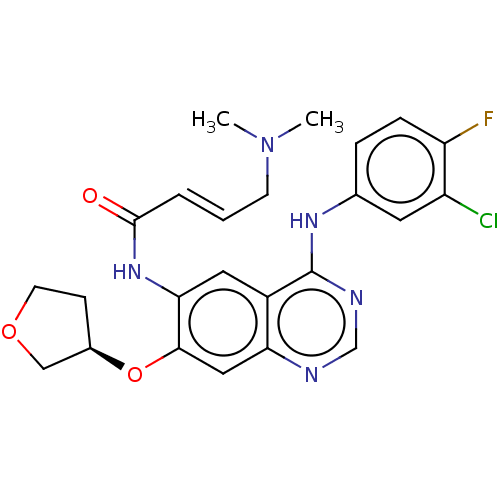
BDBM50406868 BIBW2992::CHEMBL2347958
SMILES CN(C)C\C=C\C(=O)Nc1cc2c(Nc3ccc(F)c(Cl)c3)ncnc2cc1O[C@@H]1CCOC1
InChI Key InChIKey=ULXXDDBFHOBEHA-QDLOVBKTSA-N
Data 9 IC50
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 9 hits for monomerid = 50406868
Found 9 hits for monomerid = 50406868
TargetEpidermal growth factor receptor(Homo sapiens (Human))
Jiangxi Science & Technology Normal University
Curated by ChEMBL
Jiangxi Science & Technology Normal University
Curated by ChEMBL
Affinity DataIC50: 5nMAssay Description:Inhibition of wild type EGFR (unknown origin) using FAM-labeled peptide substrate after 10 mins by mobility shift assayMore data for this Ligand-Target Pair
TargetEpidermal growth factor receptor(Homo sapiens (Human))
Jiangxi Science & Technology Normal University
Curated by ChEMBL
Jiangxi Science & Technology Normal University
Curated by ChEMBL
Affinity DataIC50: 0.5nMAssay Description:Inhibition of EGFR (unknown origin)More data for this Ligand-Target Pair
TargetEpidermal growth factor receptor(Homo sapiens (Human))
Jiangxi Science & Technology Normal University
Curated by ChEMBL
Jiangxi Science & Technology Normal University
Curated by ChEMBL
Affinity DataIC50: 7nMAssay Description:Inhibition of wild type EGFR T790M mutant (unknown origin) using FAM-labeled peptide substrate after 10 mins by mobility shift assayMore data for this Ligand-Target Pair
TargetReceptor tyrosine-protein kinase erbB-2(Homo sapiens (Human))
Ain Shams University
Curated by ChEMBL
Ain Shams University
Curated by ChEMBL
Affinity DataIC50: 14nMAssay Description:Inhibition of GST-tagged human HER2 catalytic domain expressed in insect cellsMore data for this Ligand-Target Pair
TargetEpidermal growth factor receptor(Homo sapiens (Human))
Jiangxi Science & Technology Normal University
Curated by ChEMBL
Jiangxi Science & Technology Normal University
Curated by ChEMBL
Affinity DataIC50: 3.70E+3nMAssay Description:Inhibition of wild type EGFR (unknown origin) expressed in baculovirus expression system by ELISAMore data for this Ligand-Target Pair
TargetEpidermal growth factor receptor(Homo sapiens (Human))
Jiangxi Science & Technology Normal University
Curated by ChEMBL
Jiangxi Science & Technology Normal University
Curated by ChEMBL
Affinity DataIC50: 0.5nMAssay Description:Inhibition of GST-tagged human EGFR catalytic domain expressed in insect cellsMore data for this Ligand-Target Pair
TargetEpidermal growth factor receptor(Homo sapiens (Human))
Jiangxi Science & Technology Normal University
Curated by ChEMBL
Jiangxi Science & Technology Normal University
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Inhibition of human EGFR T790M/L858R mutant expressed in mouse Ba/F3 cellsMore data for this Ligand-Target Pair
TargetEpidermal growth factor receptor(Homo sapiens (Human))
Jiangxi Science & Technology Normal University
Curated by ChEMBL
Jiangxi Science & Technology Normal University
Curated by ChEMBL
Affinity DataIC50: 1.00E+3nMAssay Description:Inhibition of EGFR L858R/T790M mutant (unknown origin) expressed in baculovirus expression system by ELISAMore data for this Ligand-Target Pair
TargetEpidermal growth factor receptor(Homo sapiens (Human))
Jiangxi Science & Technology Normal University
Curated by ChEMBL
Jiangxi Science & Technology Normal University
Curated by ChEMBL
Affinity DataIC50: 0.400nMAssay Description:Inhibition of human EGFR L858R mutant expressed in mouse Ba/F3 cellsMore data for this Ligand-Target Pair