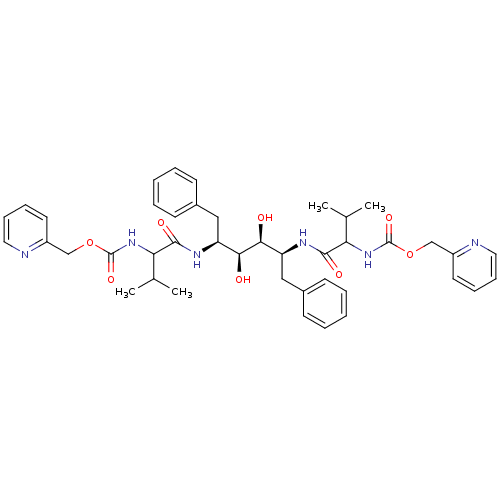
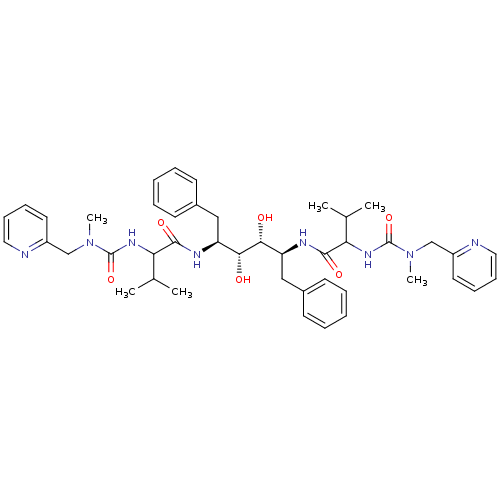
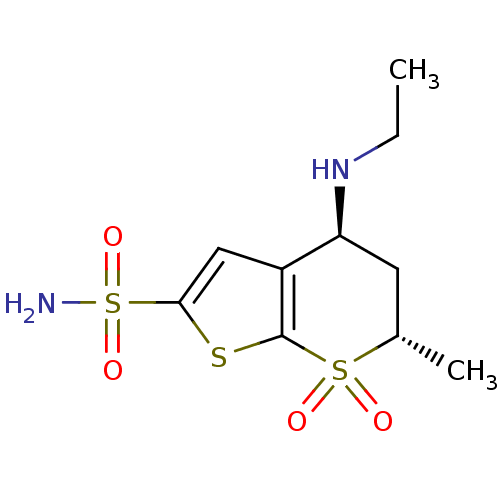
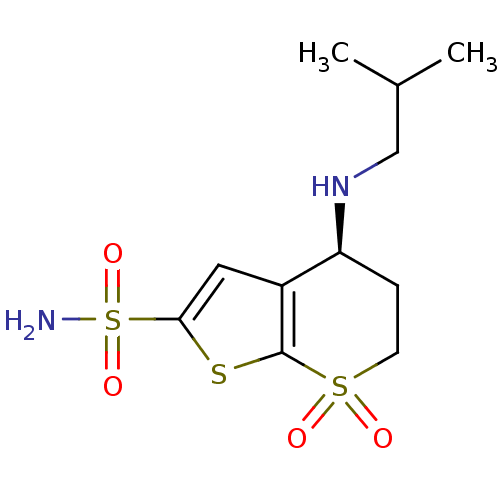
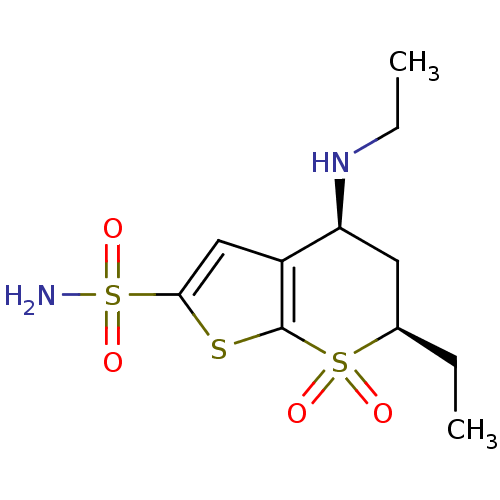
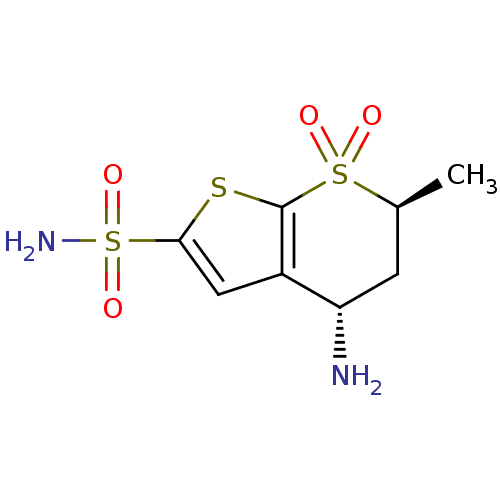
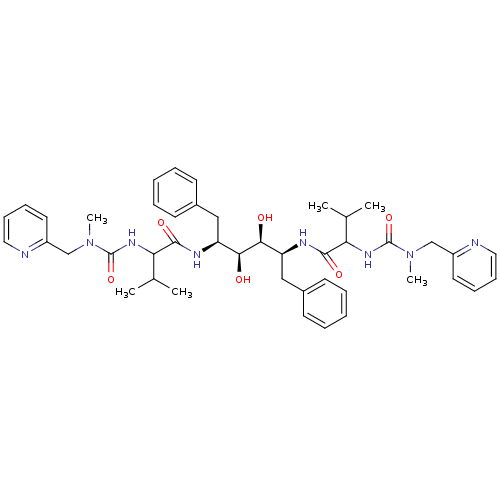
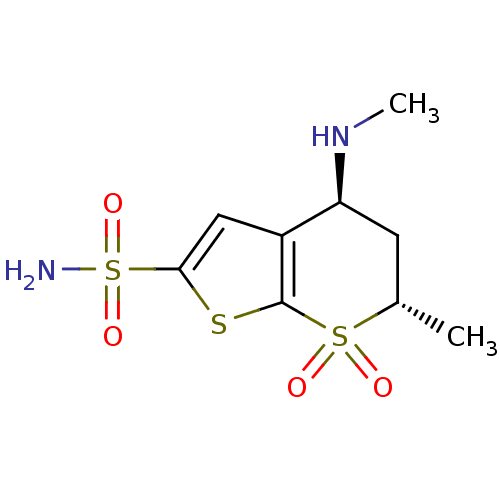
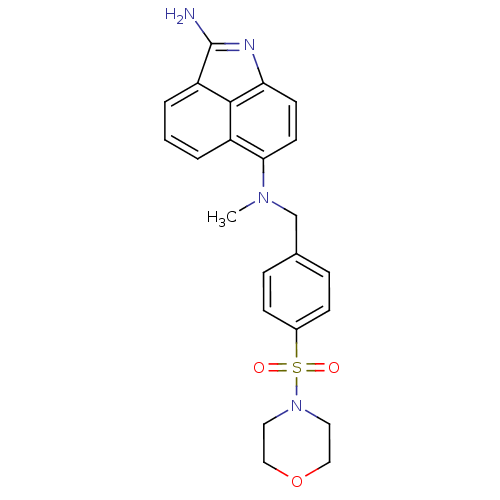
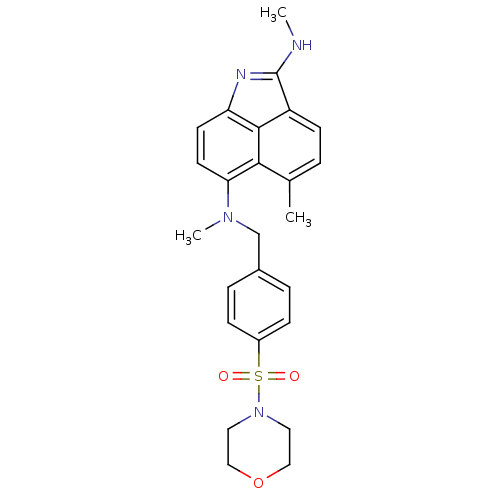
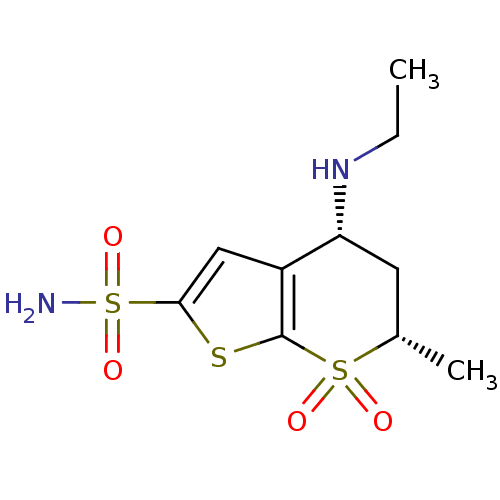
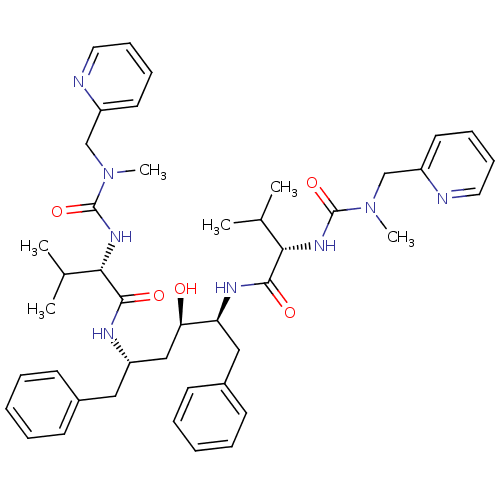
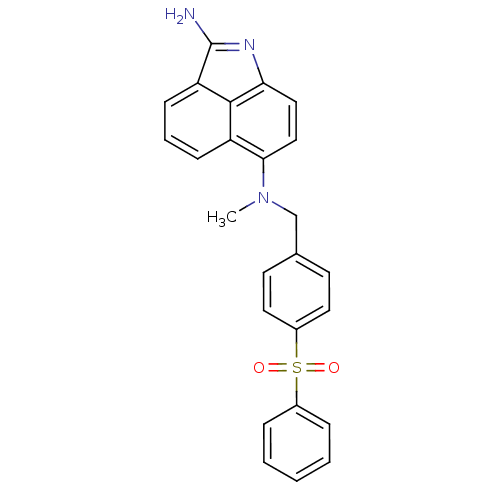
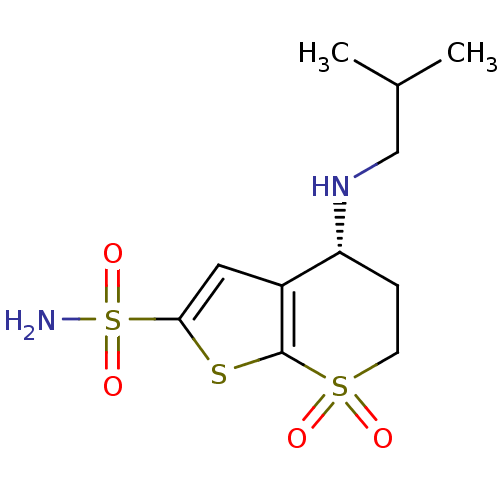
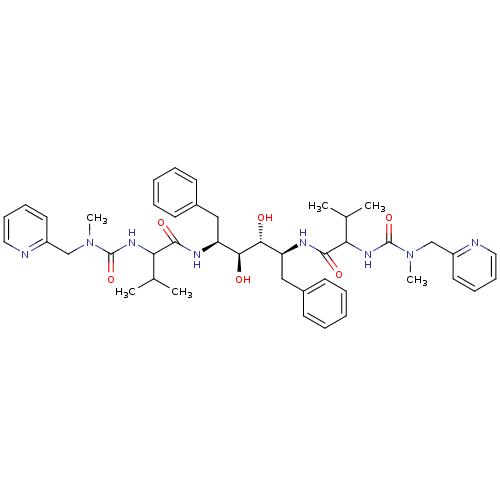
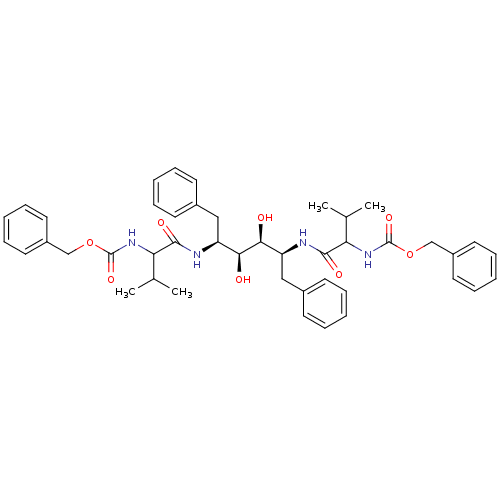
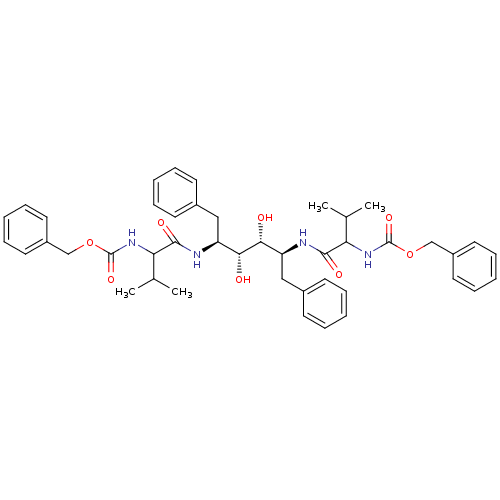
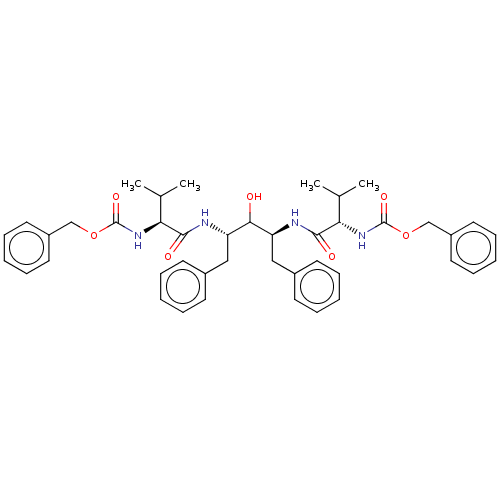
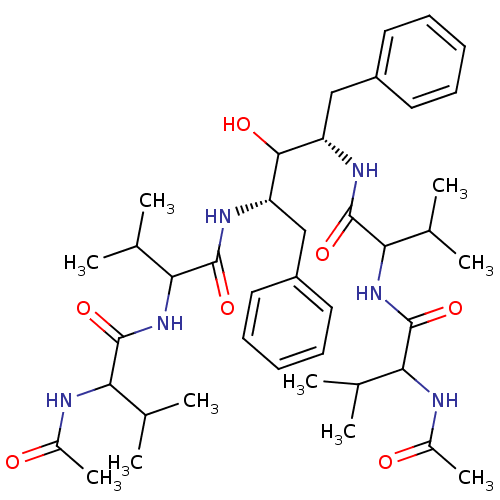
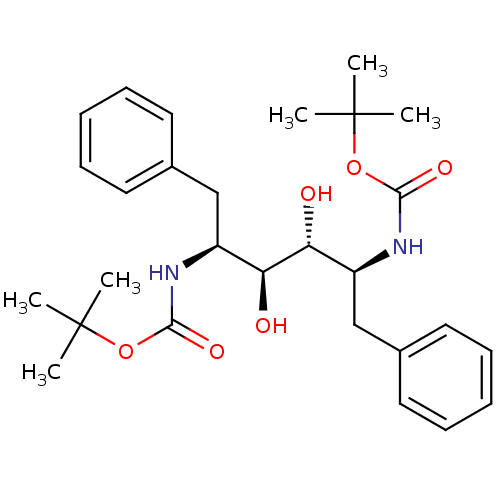
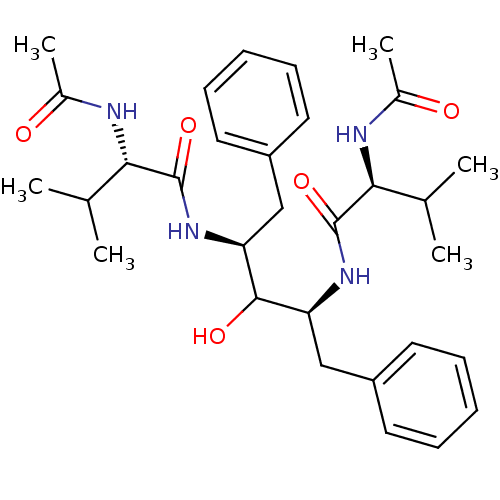
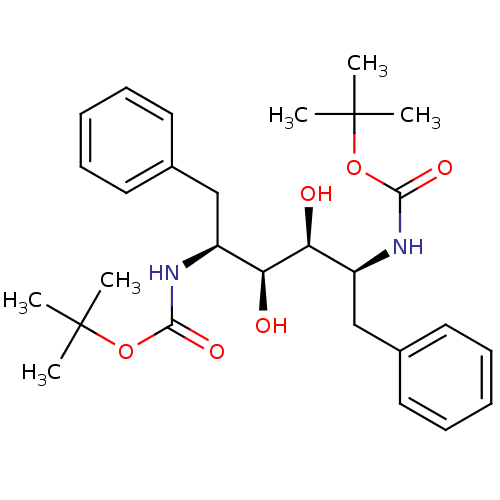
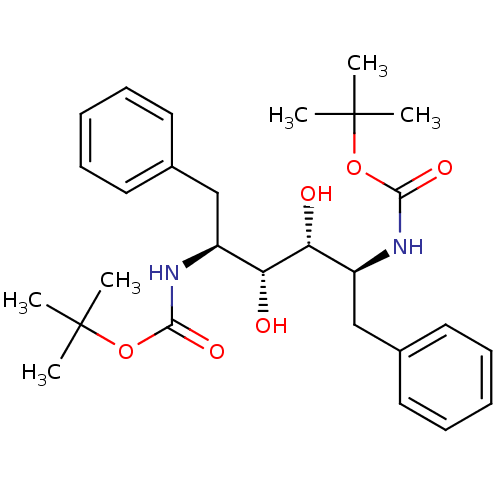
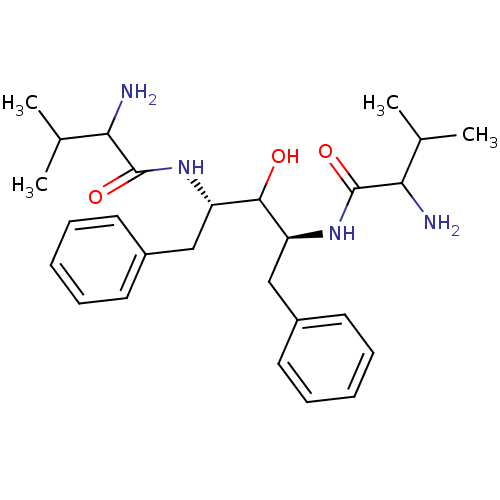
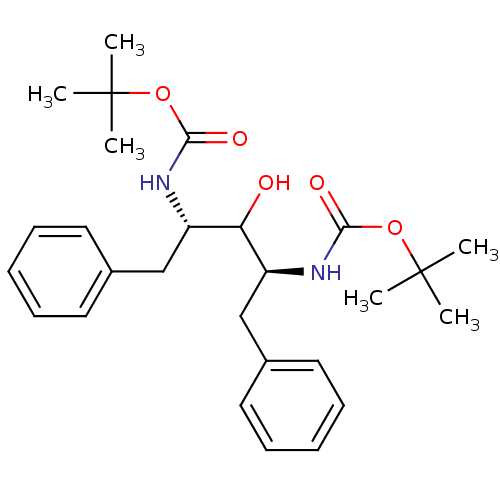
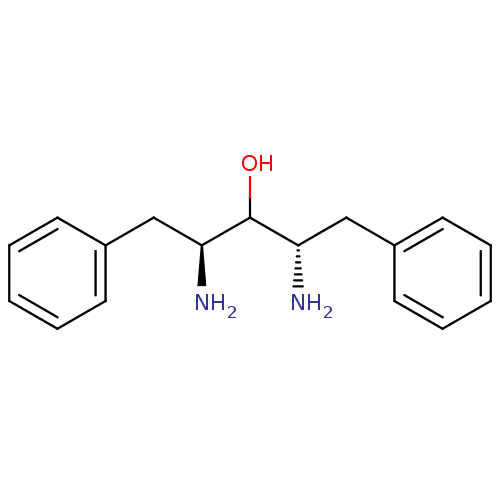
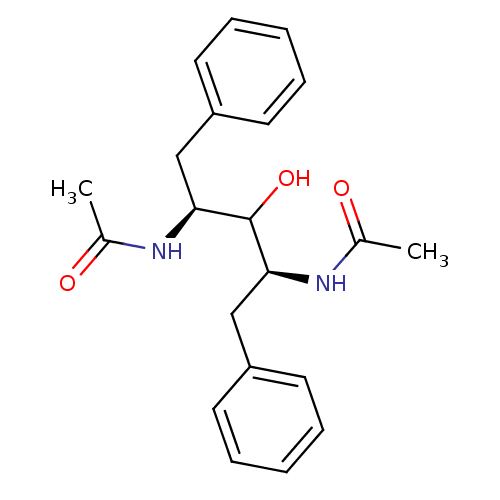
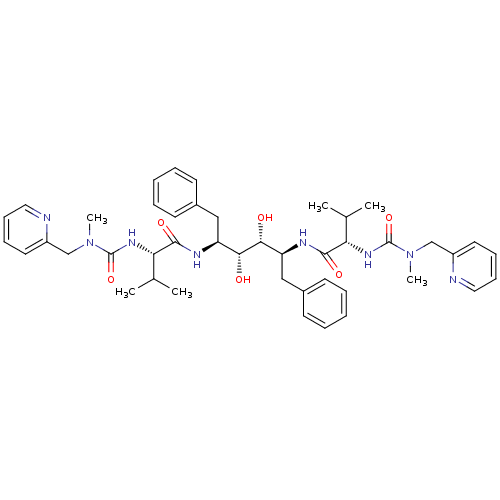
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Abbott Laboratories
Curated by ChEMBL
Abbott Laboratories
Curated by ChEMBL
Affinity DataKi: 0.0900nMAssay Description:Compound was evaluated for the inhibitory activity against HIV-1 proteaseMore data for this Ligand-Target Pair
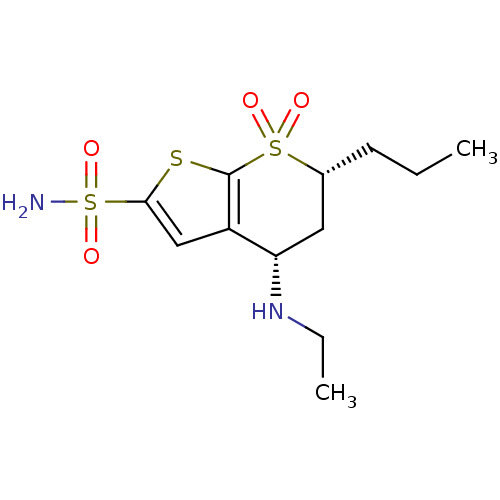
Affinity DataKi: 0.140nMAssay Description:Compound was evaluated for the inhibitory constant towards Human Carbonic anhydrase II (HCA II)More data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Abbott Laboratories
Curated by ChEMBL
Abbott Laboratories
Curated by ChEMBL
Affinity DataKi: 0.150nMAssay Description:Compound was evaluated for the inhibitory activity against HIV-1 proteaseMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Abbott Laboratories
Curated by ChEMBL
Abbott Laboratories
Curated by ChEMBL
Affinity DataKi: 0.160nMAssay Description:Compound was evaluated for the inhibitory activity against HIV-1 proteaseMore data for this Ligand-Target Pair
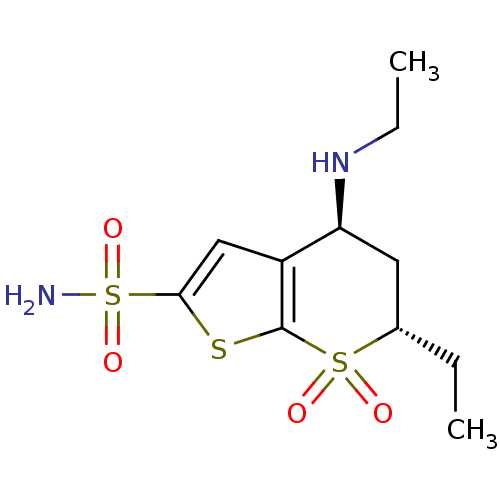
Affinity DataKi: 0.170nMAssay Description:Compound was evaluated for the inhibitory constant towards Human Carbonic anhydrase II (HCA II)More data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Abbott Laboratories
Curated by ChEMBL
Abbott Laboratories
Curated by ChEMBL
Affinity DataKi: 0.180nMAssay Description:Compound was evaluated for the inhibitory activity against HIV-1 proteaseMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Abbott Laboratories
Curated by ChEMBL
Abbott Laboratories
Curated by ChEMBL
Affinity DataKi: 0.190nMAssay Description:Compound was evaluated for the inhibitory activity against HIV-1 proteaseMore data for this Ligand-Target Pair
Affinity DataKi: 0.300nMAssay Description:Compound was evaluated for the inhibitory constant towards Human Carbonic anhydrase II (HCA II)More data for this Ligand-Target Pair
Affinity DataKi: 0.370nMAssay Description:Compound was evaluated for the inhibitory constant towards Human Carbonic anhydrase II (HCA II)More data for this Ligand-Target Pair
Affinity DataKi: 0.610nMAssay Description:Compound was evaluated for the affinity towards Human Carbonic anhydrase II (HCA II)More data for this Ligand-Target Pair
Affinity DataKi: 1nMAssay Description:Compound was evaluated for the inhibitory constant towards Human Carbonic anhydrase II (HCA II)More data for this Ligand-Target Pair
Affinity DataKi: 1.5nMAssay Description:Compound was evaluated for the inhibitory constant towards Human Carbonic anhydrase II (HCA II)More data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Abbott Laboratories
Curated by ChEMBL
Abbott Laboratories
Curated by ChEMBL
Affinity DataKi: 1.70nMAssay Description:Compound was evaluated for the inhibitory activity against HIV-1 proteaseMore data for this Ligand-Target Pair
Affinity DataKi: 1.90nMAssay Description:Compound was evaluated for the inhibitory constant towards Human Carbonic anhydrase II (HCA II)More data for this Ligand-Target Pair
Affinity DataKi: 2nMAssay Description:Compound was evaluated for the inhibitory constant of human thymidylate synthaseMore data for this Ligand-Target Pair
Affinity DataKi: 2nMAssay Description:Compound was evaluated for the affinity towards Human Carbonic anhydrase II (HCA II)More data for this Ligand-Target Pair
Affinity DataKi: 2nMAssay Description:Compound was evaluated for the inhibitory constant of human thymidylate synthaseMore data for this Ligand-Target Pair
Affinity DataKi: 15nMAssay Description:Compound was evaluated for the affinity towards Human Carbonic anhydrase II (HCA II)More data for this Ligand-Target Pair
Affinity DataKi: 17nMAssay Description:Compound was evaluated for the inhibitory constant towards Human Carbonic anhydrase II (HCA II)More data for this Ligand-Target Pair
Affinity DataKi: 34nMAssay Description:Compound was evaluated for the inhibitory constant of human thymidylate synthaseMore data for this Ligand-Target Pair
Affinity DataKi: 71nMAssay Description:Compound was evaluated for the affinity towards Human Carbonic anhydrase II (HCA II)More data for this Ligand-Target Pair
Affinity DataKi: 140nMAssay Description:Compound was evaluated for the inhibitory constant towards Human Carbonic anhydrase II (HCA II)More data for this Ligand-Target Pair
Affinity DataKi: 1.60E+3nMAssay Description:Compound was evaluated for the inhibitory constant of human thymidylate synthaseMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Abbott Laboratories
Curated by ChEMBL
Abbott Laboratories
Curated by ChEMBL
Affinity DataIC50: 0.220nMAssay Description:Inhibition of HIV-1 proteaseMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Abbott Laboratories
Curated by ChEMBL
Abbott Laboratories
Curated by ChEMBL
Affinity DataIC50: 0.220nMAssay Description:Inhibition of HIV-1 proteaseMore data for this Ligand-Target Pair
Affinity DataIC50: 0.230nMAssay Description:Compound was evaluated for the inhibitory activity against Human Carbonic anhydrase II (HCA II)More data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Abbott Laboratories
Curated by ChEMBL
Abbott Laboratories
Curated by ChEMBL
Affinity DataIC50: 0.380nMAssay Description:Inhibition of HIV-1 proteaseMore data for this Ligand-Target Pair
Affinity DataIC50: 0.540nMAssay Description:Compound was evaluated for the inhibitory activity against Human Carbonic anhydrase II (HCA II)More data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Abbott Laboratories
Curated by ChEMBL
Abbott Laboratories
Curated by ChEMBL
Affinity DataIC50: 3nMAssay Description:Inhibition of HIV-1 proteaseMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Abbott Laboratories
Curated by ChEMBL
Abbott Laboratories
Curated by ChEMBL
Affinity DataIC50: 10nMAssay Description:Inhibition of HIV-1 proteaseMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Abbott Laboratories
Curated by ChEMBL
Abbott Laboratories
Curated by ChEMBL
Affinity DataIC50: 12nMAssay Description:Inhibition of HIV-1 proteaseMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Abbott Laboratories
Curated by ChEMBL
Abbott Laboratories
Curated by ChEMBL
Affinity DataIC50: 12nMAssay Description:Inhibition of HIV-1 proteaseMore data for this Ligand-Target Pair
Affinity DataIC50: 30nMAssay Description:Compound was evaluated for the inhibitory activity against Human Carbonic anhydrase II (HCA II)More data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Abbott Laboratories
Curated by ChEMBL
Abbott Laboratories
Curated by ChEMBL
Affinity DataIC50: 40nMAssay Description:Inhibition of HIV-1 proteaseMore data for this Ligand-Target Pair
Affinity DataIC50: 45nMAssay Description:Compound was evaluated for the inhibitory activity against Human Carbonic anhydrase II (HCA II)More data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Abbott Laboratories
Curated by ChEMBL
Abbott Laboratories
Curated by ChEMBL
Affinity DataIC50: 280nMAssay Description:Inhibition of HIV-1 proteaseMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Abbott Laboratories
Curated by ChEMBL
Abbott Laboratories
Curated by ChEMBL
Affinity DataIC50: 590nMAssay Description:Inhibition of HIV-1 proteaseMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Abbott Laboratories
Curated by ChEMBL
Abbott Laboratories
Curated by ChEMBL
Affinity DataIC50: 3.00E+3nMAssay Description:Inhibition of HIV-1 proteaseMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Abbott Laboratories
Curated by ChEMBL
Abbott Laboratories
Curated by ChEMBL
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of HIV-1 proteaseMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Abbott Laboratories
Curated by ChEMBL
Abbott Laboratories
Curated by ChEMBL
Affinity DataIC50: >1.00E+4nMAssay Description:Inhibition of HIV-1 proteaseMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Abbott Laboratories
Curated by ChEMBL
Abbott Laboratories
Curated by ChEMBL
Affinity DataEC50: 280nMAssay Description:Effective concentration of compound for inhibition of HIV 1 protease was determinedMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Abbott Laboratories
Curated by ChEMBL
Abbott Laboratories
Curated by ChEMBL
Affinity DataEC50: 70nMAssay Description:Effective concentration of compound for inhibition of HIV 1 protease was determinedMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Abbott Laboratories
Curated by ChEMBL
Abbott Laboratories
Curated by ChEMBL
Affinity DataEC50: 60nMAssay Description:Effective concentration of compound for inhibition of HIV 1 protease was determinedMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Abbott Laboratories
Curated by ChEMBL
Abbott Laboratories
Curated by ChEMBL
Affinity DataEC50: 120nMAssay Description:Effective concentration of compound for inhibition of HIV 1 protease was determinedMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Abbott Laboratories
Curated by ChEMBL
Abbott Laboratories
Curated by ChEMBL
Affinity DataEC50: 50nMAssay Description:Effective concentration of compound for inhibition of HIV 1 protease was determinedMore data for this Ligand-Target Pair
TargetGag-Pol polyprotein [489-587](Human immunodeficiency virus type 1)
Abbott Laboratories
Curated by ChEMBL
Abbott Laboratories
Curated by ChEMBL
Affinity DataEC50: 20nMAssay Description:Effective concentration of compound for inhibition of HIV 1 protease was determinedMore data for this Ligand-Target Pair

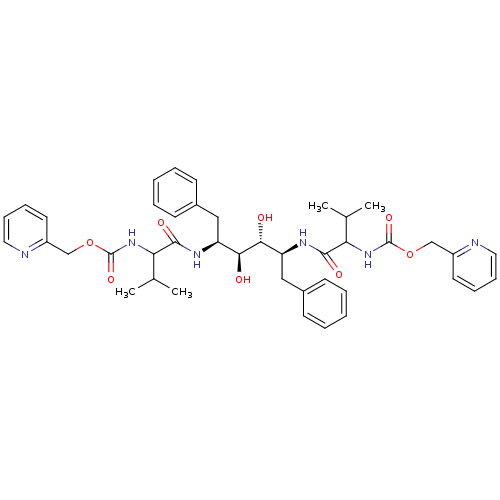
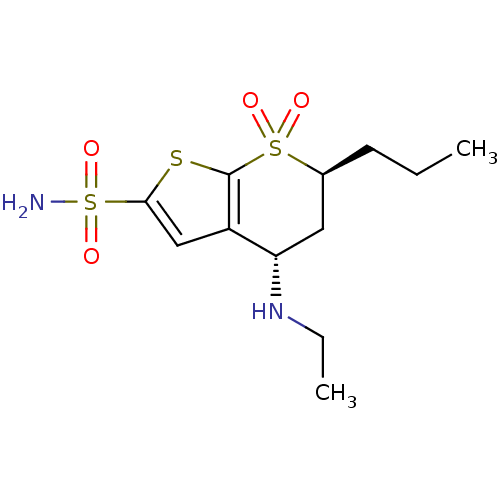
 3D Structure (crystal)
3D Structure (crystal)