Reaction Details Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Prothrombin
Ligand
BDBM50129978
Substrate
n/a
Meas. Tech.
ChEBML_207995
IC50
190±n/a nM
Citation
 Ries, UJ; Priepke, HW; Hauel, NH; Handschuh, S; Mihm, G; Stassen, JM; Wienen, W; Nar, H Heterocyclic thrombin inhibitors. Part 2: quinoxalinone derivatives as novel, potent antithrombotic agents. Bioorg Med Chem Lett 13:2297-302 (2003) [PubMed] Article
Ries, UJ; Priepke, HW; Hauel, NH; Handschuh, S; Mihm, G; Stassen, JM; Wienen, W; Nar, H Heterocyclic thrombin inhibitors. Part 2: quinoxalinone derivatives as novel, potent antithrombotic agents. Bioorg Med Chem Lett 13:2297-302 (2003) [PubMed] Article More Info.:
Target
Name:
Prothrombin
Synonyms:
Activation peptide fragment 1 | Activation peptide fragment 2 | Coagulation factor II | F2 | Prothrombin precursor | THRB_HUMAN | Thrombin heavy chain | Thrombin light chain
Type:
Protein
Mol. Mass.:
70029.57
Organism:
Homo sapiens (Human)
Description:
P00734
Residue:
622
Sequence:
MAHVRGLQLPGCLALAALCSLVHSQHVFLAPQQARSLLQRVRRANTFLEEVRKGNLERECVEETCSYEEAFEALESSTATDVFWAKYTACETARTPRDKLAACLEGNCAEGLGTNYRGHVNITRSGIECQLWRSRYPHKPEINSTTHPGADLQENFCRNPDSSTTGPWCYTTDPTVRRQECSIPVCGQDQVTVAMTPRSEGSSVNLSPPLEQCVPDRGQQYQGRLAVTTHGLPCLAWASAQAKALSKHQDFNSAVQLVENFCRNPDGDEEGVWCYVAGKPGDFGYCDLNYCEEAVEEETGDGLDEDSDRAIEGRTATSEYQTFFNPRTFGSGEADCGLRPLFEKKSLEDKTERELLESYIDGRIVEGSDAEIGMSPWQVMLFRKSPQELLCGASLISDRWVLTAAHCLLYPPWDKNFTENDLLVRIGKHSRTRYERNIEKISMLEKIYIHPRYNWRENLDRDIALMKLKKPVAFSDYIHPVCLPDRETAASLLQAGYKGRVTGWGNLKETWTANVGKGQPSVLQVVNLPIVERPVCKDSTRIRITDNMFCAGYKPDEGKRGDACEGDSGGPFVMKSPFNNRWYQMGIVSWGEGCDRDGKYGFYTHVFRLKKWIQKVIDQFGE
Inhibitor
Name:
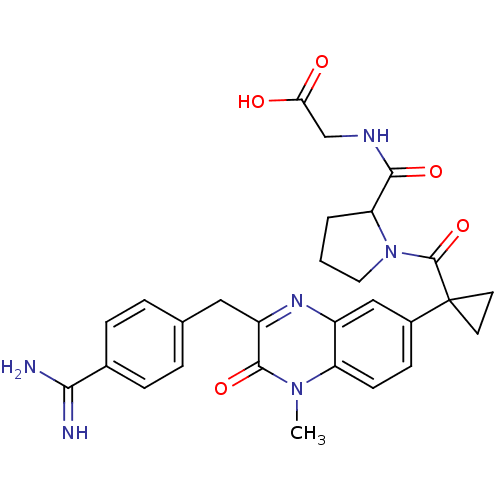
BDBM50129978
Synonyms:
CHEMBL76068 | [(1-{1-[3-(4-Carbamimidoyl-benzyl)-1-methyl-2-oxo-1,2-dihydro-quinoxalin-6-yl]-cyclopropanecarbonyl}-pyrrolidine-2-carbonyl)-amino]-acetic acid
Type:
Small organic molecule
Emp. Form.:
C28H30N6O5
Mol. Mass.:
530.575
SMILES:
Cn1c2ccc(cc2nc(Cc2ccc(cc2)C(N)=N)c1=O)C1(CC1)C(=O)N1CCCC1C(=O)NCC(O)=O