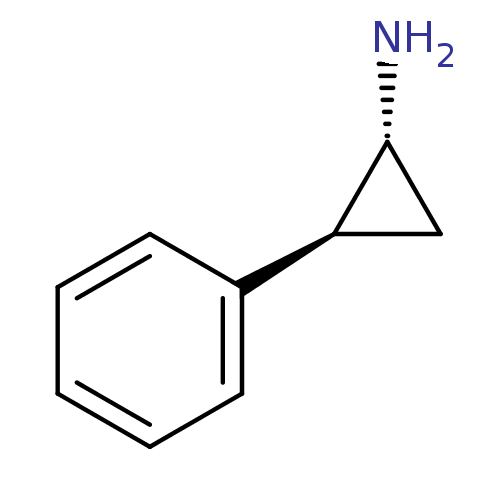
BDBM50240772 (1R,2S)-(-)-2-phenylcyclopropylamine::(1R,2S)-2-phenylcyclopropanamine::(tranylcypromine)2-Phenyl-cyclopropylamine::2-Phenyl-cyclopropylamine::CHEMBL1179::Parnate::TRANYLCYPROMINE::TRANYLCYPROMINE HYDROCHLORIDE::US10836743, Compound TCP::US8993808, Tranylcypromine::US9180183, Tranylcypromine::cid_2723716::rel-Tranylcypromine
SMILES N[C@@H]1C[C@H]1c1ccccc1
InChI Key InChIKey=AELCINSCMGFISI-DTWKUNHWSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 15 hits for monomerid = 50240772
Found 15 hits for monomerid = 50240772
Affinity DataKi: 26nMAssay Description:Inhibition of human recombinant soluble MAO-B expressed in Pichia pastoris incubated for 30 mins prior to substrate addition measured after 60 mins b...More data for this Ligand-Target Pair
Affinity DataKi: 130nMAssay Description:Mixed inhibition of CYP2A6 (unknown origin)More data for this Ligand-Target Pair
TargetSigma non-opioid intracellular receptor 1(Cavia porcellus (Guinea pig))
Universit£T M£Nster
Curated by ChEMBL
Universit£T M£Nster
Curated by ChEMBL
Affinity DataKi: 1.80E+3nMAssay Description:Displacement of [3H]-(+)-pentazocine from sigma1 receptor in guinea pig brain membrane incubated for 120 mins by solid scintillation counting methodMore data for this Ligand-Target Pair
Affinity DataKi: 6.20E+3nMAssay Description:Displacement of [3H]-di-o-tolylguanidine from Sigma 2 receptor in rat liver membrane incubated for 120 mins by solid scintillation counting methodMore data for this Ligand-Target Pair
Affinity DataKi: 6.50E+3nMAssay Description:Competitive inhibition of CYP2A13 (unknown origin)More data for this Ligand-Target Pair
Affinity DataKi: 1.28E+4nMAssay Description:Displacement of [3H]R-(+)-7-OHDPAT from dopamine D3 receptor in Sprague-Dawley rat ventral striatum after 90 minsMore data for this Ligand-Target Pair
Affinity DataKi: 1.60E+4nMAssay Description:Inhibition of human MAO-B expressed in baculovirus infected BTI insect cells using tyramine as substrate by 4-AAP/3,5-DCHBS and amplex red dye based ...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
University Of Southampton
Curated by ChEMBL
University Of Southampton
Curated by ChEMBL
Affinity DataKi: 2.10E+4nMAssay Description:Inhibition of human LSD1More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
University Of Southampton
Curated by ChEMBL
University Of Southampton
Curated by ChEMBL
Affinity DataKi: 2.50E+4nMAssay Description:Inhibition of human recombinant LSD1 using H3K4me2 as substrate preincubated for 15 mins followed by substrate addition and measured after 30 mins by...More data for this Ligand-Target Pair
TargetAmine oxidase [flavin-containing] A(Homo sapiens (Human))
Key Laboratory Of Henan Provinc
Curated by ChEMBL
Key Laboratory Of Henan Provinc
Curated by ChEMBL
Affinity DataKi: 1.02E+5nMAssay Description:Inhibition of human MAO-A expressed in baculovirus infected BTI insect cells using tyramine as substrate by 4-AAP/3,5-DCHBS and amplex red dye based ...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
University Of Southampton
Curated by ChEMBL
University Of Southampton
Curated by ChEMBL
Affinity DataKi: 2.43E+5nMAssay Description:Inhibition of N-terminal 6histidine-tagged LSD1 (unknown origin) expressed in Escherichia coli BL21(DE3) cells assessed as reduction in Peroxide prod...More data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
University Of Southampton
Curated by ChEMBL
University Of Southampton
Curated by ChEMBL
Affinity DataKi: 2.43E+5nMAssay Description:Binding affinity to LSD1 (unknown origin) assessed as inhibition constantMore data for this Ligand-Target Pair
TargetLysine-specific histone demethylase 1A(Homo sapiens (Human))
University Of Southampton
Curated by ChEMBL
University Of Southampton
Curated by ChEMBL
Affinity DataKi: 3.57E+5nMAssay Description:Irreversible inhibition of LSD1More data for this Ligand-Target Pair