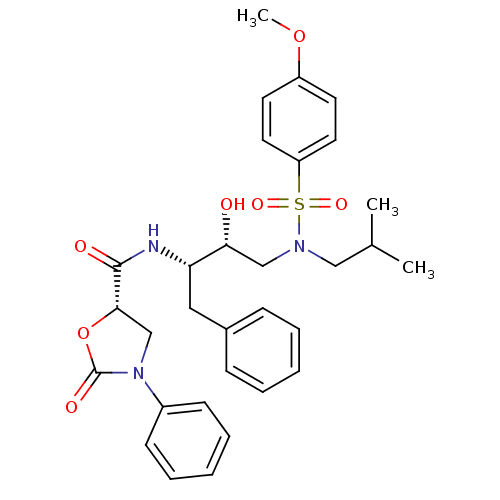
BDBM12884 (5S)-N-[(1S,2R)-1-Benzyl-2-hydroxy-3-[isobutyl[(4-methoxy-phenyl)sulfonyl]amino]propyl]-2-oxo-3-phenyloxazolidine-5-carboxamide::(5S)-N-[(2S,3R)-3-hydroxy-4-[(4-methoxybenzene)(2-methylpropyl)sulfonamido]-1-phenylbutan-2-yl]-2-oxo-3-phenyl-1,3-oxazolidine-5-carboxamide::N-Aryl-oxazolidinone-5-carboxamide Analogue 21a
SMILES COc1ccc(cc1)S(=O)(=O)N(CC(C)C)C[C@@H](O)[C@H](Cc1ccccc1)NC(=O)[C@@H]1CN(C(=O)O1)c1ccccc1
InChI Key InChIKey=IGYIMSLYOKOAJR-NHKHRBQYSA-N
Data 4 KI
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 4 hits for monomerid = 12884
Found 4 hits for monomerid = 12884
TargetDimer of Gag-Pol polyprotein [491-589,Q496K](Human immunodeficiency virus type 1)
University of Massachusetts Medical School
University of Massachusetts Medical School
Affinity DataKi: 0.100nMAssay Description:HIV-1 protease inhibitor activities were determined by the fluorescence resonance energy transfer (FRET) method. The energy transfer donor (EDANS) an...More data for this Ligand-Target Pair
TargetProtease(Human immunodeficiency virus 1 (HIV-1))
University Of Massachusetts Medical School
Curated by ChEMBL
University Of Massachusetts Medical School
Curated by ChEMBL
Affinity DataKi: 0.100nMAssay Description:Inhibition of wild type HIV1 protease by FRETMore data for this Ligand-Target Pair
Affinity DataKi: 0.100nMAssay Description:Inhibition of HIV1 protease Q7K mutant by FRET methodMore data for this Ligand-Target Pair
Affinity DataKi: 0.100nMAssay Description:Inhibition of HIV1 protease Q7K mutant by FRET methodMore data for this Ligand-Target Pair