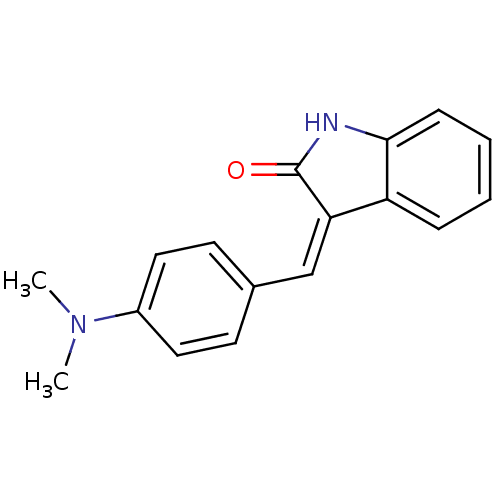
BDBM17747 (3Z)-3-{[4-(dimethylamino)phenyl]methylidene}-2,3-dihydro-1H-indol-2-one::CHEMBL328710::SU 4312::SU4312
SMILES CN(C)c1ccc(\C=C2/C(=O)Nc3ccccc23)cc1
InChI Key InChIKey=UAKWLVYMKBWHMX-PTNGSMBKSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 24 hits for monomerid = 17747
Found 24 hits for monomerid = 17747
Affinity DataKi: 84nMAssay Description:Displacement of Thio-T from recombinant alpha-synuclein (unknown origin) expressed in Escherichia coli after 1.5 hrs by fluorescence assayMore data for this Ligand-Target Pair
Affinity DataKi: 92nMAssay Description:Displacement of Thio-T from amyloid beta (1 to 42) (unknown origin) expressed in Escherichia coli after 1.5 hrs by fluorescence assayMore data for this Ligand-Target Pair
TargetMicrotubule-associated protein tau(Homo sapiens (Human))
Washington University
Curated by ChEMBL
Washington University
Curated by ChEMBL
Affinity DataKi: 262nMAssay Description:Displacement of Thio-T from human recombinant tau (243 to 375) expressed in Escherichia coli BL21(DE3)RIL after 1.5 hrs by fluorescence assayMore data for this Ligand-Target Pair
Affinity DataIC50: 1.94E+4nMAssay Description:Test concentration required to achieve 50% inhibition of tyrosine phosphorylation on human Platelet-derived growth factor receptor beta (PDGF RTK).More data for this Ligand-Target Pair
Affinity DataIC50: 800nMAssay Description:Concentration required to achieve 50% inhibition of tyrosine phosphorylation on murine VEGF receptor (FLK-1 RTK).More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Concentration required to achieve 50% inhibition of tyrosine phosphorylation on human Her-2 receptor tyrosine kinase (HER-2 RTK)More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Concentration required to achieve 50% inhibition of tyrosine phosphorylation on human Epidermal growth factor receptor (EGF RTK).More data for this Ligand-Target Pair
Affinity DataIC50: >1.00E+5nMAssay Description:Concentration required to achieve 50% inhibition of tyrosine phosphorylation on human Insulin-like growth factor I receptorMore data for this Ligand-Target Pair
Affinity DataIC50: 3.70E+3nMAssay Description:Inhibition of PDGFRbeta phosphorylation in human SF539 cells after 10 mins by FLISAMore data for this Ligand-Target Pair
Affinity DataIC50: 3.75E+3nMAssay Description:Inhibition of PDGFRbeta tyrosine kinase activity in PDGF-BB-stimulated human SF-539 cells after 60 mins by ELISAMore data for this Ligand-Target Pair
Affinity DataIC50: 3.70E+3nMAssay Description:Inhibition of PDGFRbeta in human SF539 cells assessed as inhibition of PDGFR-BB-induced tyrosine phosphorylation incubated for 60 mins prior to EGF-a...More data for this Ligand-Target Pair
TargetProto-oncogene tyrosine-protein kinase receptor Ret(Homo sapiens (Human))
University Of Milano-Bicocca
Curated by ChEMBL
University Of Milano-Bicocca
Curated by ChEMBL
Affinity DataIC50: >1.00E+5nMAssay Description:Inhibition of human recombinant His-tagged RET expressed in Sf9 insect cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 3.75E+3nMAssay Description:Inhibition of PDGFRbeta in human SF539 cells by phosphotyrosine cell-based ELISAMore data for this Ligand-Target Pair
Affinity DataIC50: 3.75E+3nMAssay Description:Inhibition of PDGFRbeta expressed in human A431 cellsMore data for this Ligand-Target Pair
Affinity DataIC50: 3.75E+3nMAssay Description:Inhibition of PDGFRbeta in human SF539 cells pretreated for 60 mins measured after 1 hr by ELISAMore data for this Ligand-Target Pair
Affinity DataIC50: 3.70E+3nMAssay Description:Inhibition of PDGF-BB-induced PDGFR-beta phosphorylation in human SF-539 cells preincubated for 60 mins followed by PDGF-BB induction measured after ...More data for this Ligand-Target Pair
TargetLuciferin 4-monooxygenase(Photinus pyralis)
National Human Genome Research Institute
Curated by ChEMBL
National Human Genome Research Institute
Curated by ChEMBL
Affinity DataIC50: 2.50E+3nMAssay Description:Inhibition of Photinus pyralis luciferase by PK-Light assayMore data for this Ligand-Target Pair
TargetLuciferin 4-monooxygenase(Photinus pyralis)
National Human Genome Research Institute
Curated by ChEMBL
National Human Genome Research Institute
Curated by ChEMBL
Affinity DataIC50: 4.50E+3nMAssay Description:Inhibition of Photinus pyralis luciferase by Easy lite assayMore data for this Ligand-Target Pair
TargetLuciferin 4-monooxygenase(Photinus pyralis)
National Human Genome Research Institute
Curated by ChEMBL
National Human Genome Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.42E+4nMAssay Description:Inhibition of Photinus pyralis luciferase by Steady-Glo reporter gene assayMore data for this Ligand-Target Pair
TargetLuciferin 4-monooxygenase(Photinus pyralis)
National Human Genome Research Institute
Curated by ChEMBL
National Human Genome Research Institute
Curated by ChEMBL
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of Photinus pyralis luciferase by BrightGlo reporter gene assayMore data for this Ligand-Target Pair
TargetMacrophage colony-stimulating factor 1 receptor [538-678,753-922](Human Sapiens (Human))
Johnson & Johnson Pharmaceutical
Johnson & Johnson Pharmaceutical
Affinity DataIC50: 3.90E+3nMpH: 7.5 T: 2°CAssay Description:The full-length cFMS cytoplasmic domain (FMS.538-972.6His) or chimera was incubated with compound in reaction buffer. Control reactions were run in e...More data for this Ligand-Target Pair
TargetLuciferin 4-monooxygenase(Photinus pyralis)
National Human Genome Research Institute
Curated by ChEMBL
National Human Genome Research Institute
Curated by ChEMBL
Affinity DataIC50: 800nMAssay Description:Inhibition of Photinus pyralis luciferaseMore data for this Ligand-Target Pair
Affinity DataIC50: 4.70E+3nMpH: 7.5 T: 2°CAssay Description:The full-length cFMS cytoplasmic domain (FMS.538-972.6His) or chimera was incubated with compound in reaction buffer. Control reactions were run in e...More data for this Ligand-Target Pair
TargetMacrophage colony-stimulating factor 1 receptor [538-678,753-922](Homo sapiens (Human))
Johnson & Johnson Pharmaceutical
Johnson & Johnson Pharmaceutical
Affinity DataIC50: 4.60E+3nMpH: 7.5 T: 2°CAssay Description:The full-length cFMS cytoplasmic domain (FMS.538-972.6His) or chimera was incubated with compound in reaction buffer. Control reactions were run in e...More data for this Ligand-Target Pair