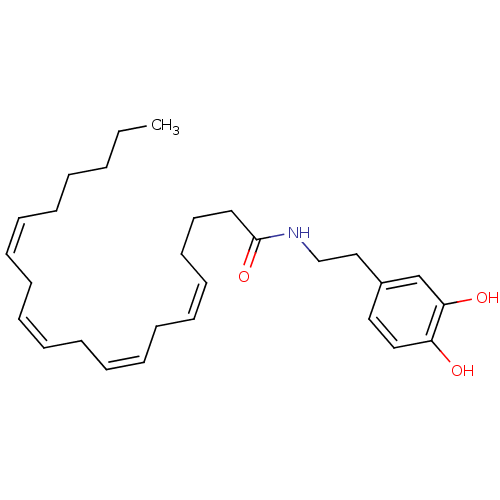
BDBM20462 (5Z,8Z,11Z,14Z)-N-[2-(3,4-dihydroxyphenyl)ethyl]icosa-5,8,11,14-tetraenamide::Arachidonoyl dopamine::CHEMBL138921::N-arachidonoyl-dopamine (NADA)
SMILES CCCCC\C=C/C\C=C/C\C=C/C\C=C/CCCC(=O)NCCc1ccc(O)c(O)c1
InChI Key InChIKey=MVVPIAAVGAWJNQ-DOFZRALJSA-N
Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB
 Found 6 hits for monomerid = 20462
Found 6 hits for monomerid = 20462
Affinity DataKi: 250nMAssay Description:Binding affinity for cannabinoid receptor 1More data for this Ligand-Target Pair
Affinity DataKi: 250nMAssay Description:Displacement of [3H]SR141716A from rat brain CB1 receptor by liquid scintillation spectrophotometryMore data for this Ligand-Target Pair
Affinity DataKi: 380nMAssay Description:Concentration required to displace 0.4 nM [3H]-SR-141,716A from CB1 receptor in rat brain preparations in the presence of 0.1 mM phenylmethyl sulphon...More data for this Ligand-Target Pair
TargetTransient receptor potential cation channel subfamily V member 1(Homo sapiens (Human))
Abbott Laboratories
Abbott Laboratories
Affinity DataKi: >6.31E+3nM ΔG°: >-7.09kcal/mole EC50: 1.48E+3nMpH: 7.4 T: 2°CAssay Description:The hTRPV1-expressing CHO cell membranes were incubated with [3H]A-778317 and test compounds to establish equilibrium. After incubation was terminate...More data for this Ligand-Target Pair
Affinity DataKi: 1.00E+4nMAssay Description:Displacement of [3H]Win55212-2 from CB2 receptor of rat spleen by liquid scintillation countingMore data for this Ligand-Target Pair
TargetTransient receptor potential cation channel subfamily V member 1(Homo sapiens (Human))
Abbott Laboratories
Abbott Laboratories
Affinity DataEC50: 50nMAssay Description:Activation of human VR1 expressed in HEK293 cells assessed as stimulation of Ca2+ influx by Fluo-3 staining-based fluorescence assayMore data for this Ligand-Target Pair